Clear Sky Science · en
Utilizing of KNO3, KCl, and NaCl as phase change materials within the refrigerator for energy saving
Why your fridge’s hidden cold matters
Home refrigerators quietly run day and night, often in hot kitchens, and together they use a surprising share of household electricity. This study explores a simple idea with big potential: slipping a slim pack of special cooling material under the freezer so the fridge can store cold and coast through temperature swings. By making better use of stored cold, the fridge can stay steadier inside, ride out short power cuts, and shave money off the electric bill without changing how we use it.
Storing cold like a battery
The researchers focused on “phase change” materials, substances that absorb or release large amounts of heat as they melt and freeze, much like ice does when it turns to water. Instead of redesigning the refrigerator, they built a flat stainless-steel container that fits under the freezer’s evaporator coils without stealing shelf space. They filled this pack with water alone or with water containing small amounts (10 percent by weight) of familiar salts: potassium nitrate (KNO3), sodium chloride (NaCl), and potassium chloride (KCl). These mixtures were chosen because they freeze and melt a few degrees below zero, close to the normal freezer temperature, so they can quietly store and release cold as the fridge cycles on and off.
Real-world testing in a hot room
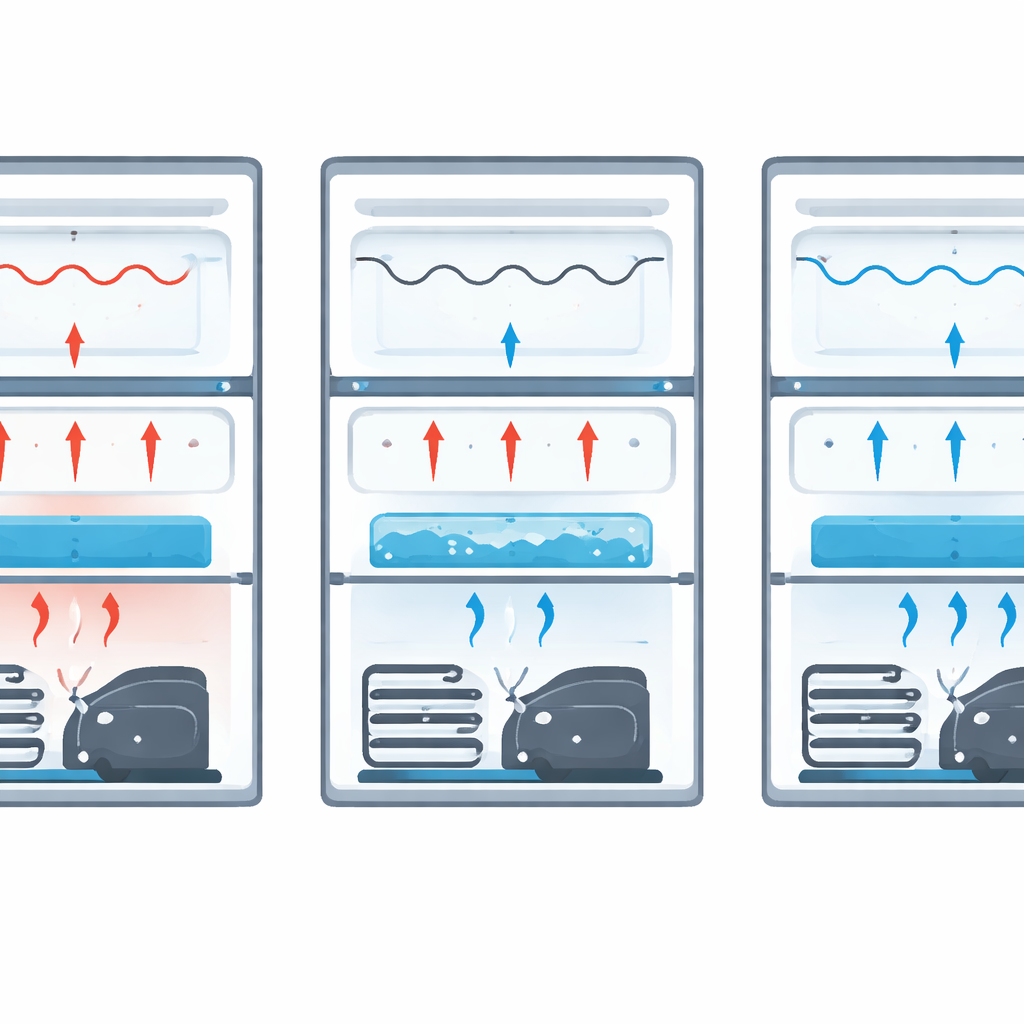
To see what difference this hidden cold store could make, the team tested a standard 289-liter single-door refrigerator running on a common refrigerant (R-134a). They placed it in a well-controlled chamber kept at 32 °C and moderate humidity, mimicking a hot summer kitchen. Precision sensors tracked temperatures at many points: inside the freezer, food compartment, vegetable drawer, door, and on the surfaces of the condenser and compressor. They also measured power use and pressures in the cooling circuit. Eight scenarios were compared: no phase change material, water alone, and the three salt–water mixtures, each tried at two volumes (1 liter and 1.5 liters) in the container.
Cooler, steadier food and gentler hardware
The phase change packs clearly calmed down temperature swings. In the freezer, all salt mixtures kept the air colder and more stable than running without a pack, while the rest of the fridge dropped by about 1–3 °C on average. The vegetable drawer cooled by up to roughly 22 percent relative to the baseline, which is helpful for keeping produce fresh. Even though the pack sat beneath the freezer, it indirectly cooled and smoothed temperatures in the main compartment and at the door by absorbing bursts of heat and then releasing cold slowly. At the same time, the midpoint of the condenser and the surface of the compressor ran a few degrees cooler, signs that the machine was working less hard and under milder thermal stress.
Saving energy and riding out power cuts
Because the phase change materials soaked up part of the cooling load, the compressor could shut off more often. In the best case, using 1 liter of the KNO3 mixture cut the compressor’s operating time over a day by 7.1 percent. This translated into an 8.6 percent drop in electrical energy use compared with the fridge running without any pack. The KCl and NaCl mixtures also saved energy, though slightly less, while water alone offered only modest gains. During a simulated one-hour power outage, fridges with packs kept the freezer below zero for about half an hour and held the fridge compartment up to 2 °C cooler than the unmodified unit, buying extra time before food warmed.
Which mixture works best and why it matters
Among all tested options, the 1-liter KNO3 solution provided the best overall balance: it increased the refrigerator’s efficiency rating (its coefficient of performance) by about 12 percent and delivered the largest energy savings. Larger 1.5-liter packs helped keep compartment temperatures even more uniform but did not improve power use as much, because extra material can become a thermal burden once fully melted or frozen. For a layperson, the takeaway is straightforward: a thin, well-chosen cold-storage pack integrated near the freezer can make an ordinary fridge cooler inside, kinder to food during outages, and cheaper to run. The work suggests that manufacturers could incorporate such packs using inexpensive salts to reduce household energy demand and support broader energy-saving and climate goals.
Citation: Samir, S., Salem, M., Mohamed, A.S.A. et al. Utilizing of KNO3, KCl, and NaCl as phase change materials within the refrigerator for energy saving. Sci Rep 16, 9815 (2026). https://doi.org/10.1038/s41598-026-41662-5
Keywords: energy efficient refrigerators, phase change materials, thermal storage, household cooling, electricity savings