Clear Sky Science · en
Rapid microwave assisted RAFT synthesis of amphiphilic HEMA-co-AMPS copolymers for high performance Cu2+ and Cr6+ removal from water
Why cleaning up metal-polluted water matters
Across the globe, rivers and groundwater are picking up invisible hitchhikers from industry: metal ions such as copper and chromium. In small amounts, some metals are harmless or even useful; in higher doses, they can damage the brain, liver, and kidneys, and may cause cancer. Conventional treatment plants struggle to remove these pollutants efficiently, especially when metal levels are low. This paper explores a new, fast-made plastic-like material that can grab and hold toxic copper and chromium from water, offering a promising tool for safer drinking water and cleaner wastewater.
A new sponge built from smart building blocks
The researchers designed a tailor-made polymer, a long-chain molecule similar to those in everyday plastics, but decorated with chemical groups that strongly attract metal ions. It is built from two smaller units: one that likes water and carries hydroxyl groups, and another that carries powerful sulfonate groups that become negatively charged in water. Together they form an "amphiphilic" copolymer—part water-friendly, part highly charged—so it can disperse well in water while presenting many active spots where metal ions can stick. This dual nature is key to drawing in both positively charged copper (Cu²⁺) and negatively charged chromium species (Cr⁶⁺).
Speeding up chemistry with microwaves
Making such a precise polymer normally takes time and energy. The team turned to a controlled method called RAFT polymerization, then supercharged it using microwave heating. Instead of relying on slow, uneven warming from the outside of a flask, microwaves heat the entire liquid more uniformly and quickly. Within 10 to 40 minutes, at modest temperatures, the monomers link into chains while a special control agent keeps the chains uniform and prevents unwanted branching. Tests using infrared spectroscopy, electron microscopy, and heat analysis showed that the resulting copolymer has the desired mix of functional groups, forms smooth and uniform particles, and is stable enough to survive repeated use.
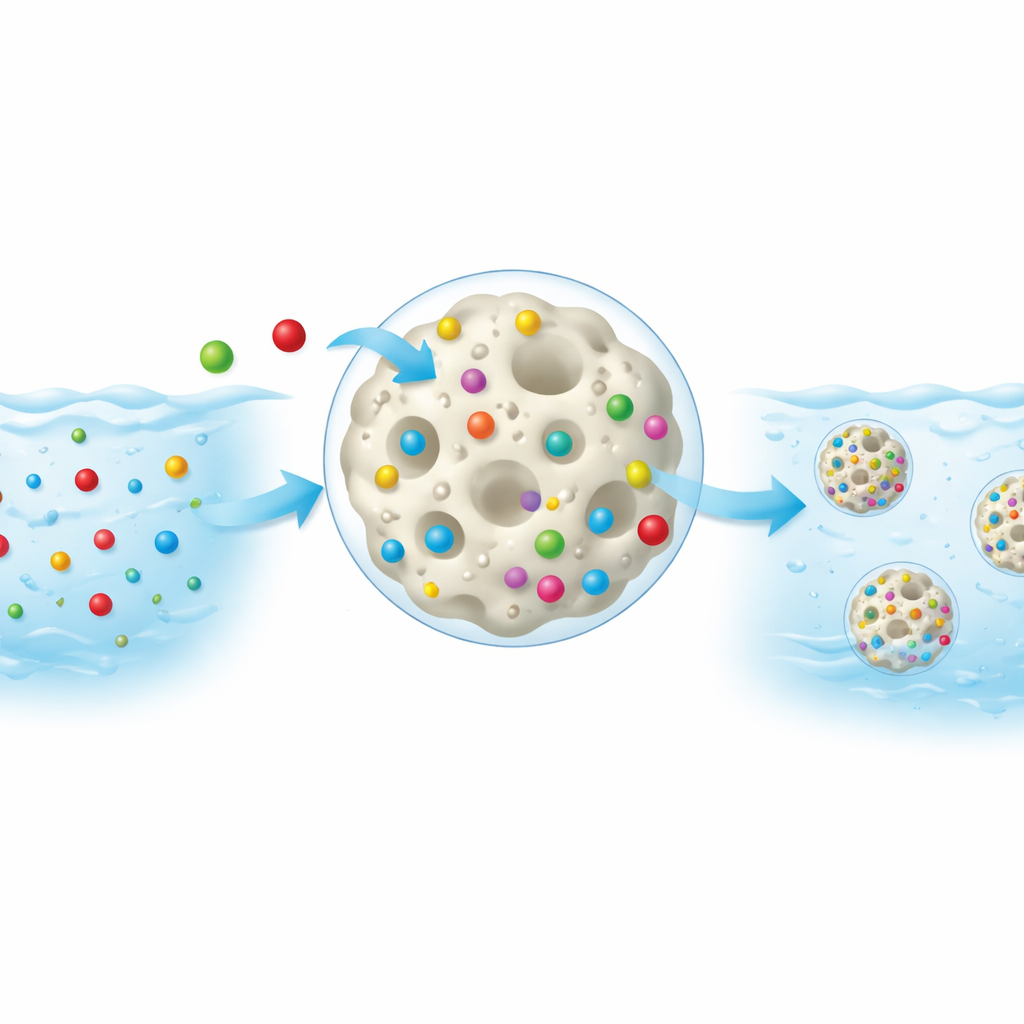
How the metal-grabbing process works
When the copolymer is mixed into contaminated water, its charged and polar groups act like tiny magnets for metal ions. For copper, which carries a positive charge, the attraction is mainly electrostatic: Cu²⁺ ions are drawn to the negatively charged sulfonate groups, and then further anchored by subtle coordination to nearby oxygen atoms. For chromium, which usually appears as negatively charged oxyanions, the situation is different. Because both the polymer surface and chromium species are negatively charged at many pH values, simple attraction is weaker. Instead, chromium is taken up through hydrogen bonding, surface complexation, and being physically trapped within the polymer network, especially under more acidic conditions where some groups on the polymer become protonated and more welcoming to these anions.
Performance in real-world-like conditions
In batch tests that mimic treatment tanks, the new material quickly soaked up metals from water containing a range of starting concentrations. Within one to three hours, the polymer reached near-maximum loading: about 165 milligrams of copper and 115 milligrams of chromium per gram of polymer. The uptake followed kinetic patterns typical of strong, chemically anchored binding rather than weak, easily reversible sticking. Models of how the metals arrange on the surface suggest that copper forms an orderly single layer on relatively uniform sites, while chromium binds more irregularly to a variety of sites and depths. Importantly, the process is naturally favorable: copper uptake becomes stronger at higher temperatures, while chromium binding releases heat and works best at somewhat lower temperatures.
Built to be used again and again
For any realistic water treatment technology, reusability is crucial. The team cycled their copolymer through five rounds of loading with metals and then regenerating it. After these cycles, the material still held more than 87 percent of its original adsorption capacity for both copper and chromium. Imaging showed that, after binding metals, the initially smooth, spherical particles tend to clump into rougher clusters, consistent with metal ions bridging different polymer particles together. Yet the main backbone of the material remains intact, and only a modest decline in performance appears, likely due to a few blocked sites that are not fully cleared during regeneration.
What this means for cleaner water
To a non-specialist, the take-home message is that the authors have built a kind of reusable, custom-designed sponge that efficiently pulls both positively and negatively charged metal pollutants out of water. By using microwaves to drive a carefully controlled polymerization process, they can make this sponge quickly, with less energy and more consistent quality than traditional routes. The material works fast, holds a large amount of both copper and chromium, and survives multiple cleaning cycles with little loss of effectiveness. This combination of smart chemistry, energy-efficient manufacture, and strong performance points toward more practical and sustainable tools for protecting waterways and drinking water from heavy metal contamination.
Citation: Gaffer, A., Ebada, A. & Alawady, A.R. Rapid microwave assisted RAFT synthesis of amphiphilic HEMA-co-AMPS copolymers for high performance Cu2+ and Cr6+ removal from water. Sci Rep 16, 10942 (2026). https://doi.org/10.1038/s41598-026-41634-9
Keywords: heavy metal removal, water treatment, polymer adsorbent, microwave-assisted synthesis, copper and chromium pollution