Clear Sky Science · en
Evaluation of phytochemicals from Tamarindus indica as a potential catechol-O-methyltransferase (COMT) inhibitor: an in-silico approach for Parkinson’s disease
Why a sour fruit matters for brain health
Parkinson’s disease robs people of smooth movement, clear speech, and often much more, yet current medicines can lose their punch and cause serious side effects over time. This study explores whether natural chemicals from the familiar tamarind tree, often used in cooking and traditional remedies, might help existing Parkinson’s drugs work better and more safely. Using advanced computer simulations instead of lab animals or human volunteers, the researchers hunted for tamarind molecules that could protect a key brain chemical called dopamine by blocking an enzyme that breaks it down.
The problem of fading Parkinson’s medicine
People with Parkinson’s disease gradually lose brain cells that make dopamine, a messenger that helps control movement, mood, and thinking. The main drug, levodopa, is converted into dopamine in the brain, but it is also quickly broken down in the body, so its benefits can wear off between doses, leading to “on–off” periods where symptoms surge back. One of the main culprits is an enzyme called catechol-O-methyltransferase, or COMT, which chemically modifies dopamine and related molecules. Doctors already prescribe synthetic COMT blockers alongside levodopa to keep dopamine around longer, but some of these drugs can stress the liver or cause other unwanted effects, limiting how long and how widely they can be used.
Turning to tamarind for gentler helpers
To search for gentler alternatives, the team focused on Tamarindus indica, the tamarind tree, known for its tangy pulp and long history in traditional medicine. From a public database of Indian medicinal plants, they collected information on 170 distinct tamarind chemicals. Using specialized software, they built a high‑resolution 3D model of human COMT based on its known crystal structure and virtually “docked” each plant compound into the enzyme’s active site, the pocket where it grabs dopamine. This allowed them to predict how tightly each molecule might bind and what kinds of contacts—such as hydrogen bonds and attraction to metal ions—would hold it in place compared with existing drugs like entacapone, tolcapone, and opicapone.
A standout plant molecule in the crowd
One compound, gallacetophenone, rose to the top. In the docking tests, it latched onto COMT with a strength similar to or better than some approved drugs, while forming several distinctive stabilizing contacts inside the enzyme’s pocket. Importantly, computer-based checks of “drug‑likeness” suggested that gallacetophenone is small and balanced enough in its water‑loving and fat‑loving character to be absorbed when taken by mouth and to cross the blood–brain barrier, which is essential for acting in the brain. Safety prediction tools indicated a relatively low likelihood of common toxic effects, putting it on at least comparable footing with current COMT inhibitors and in some respects looking safer.
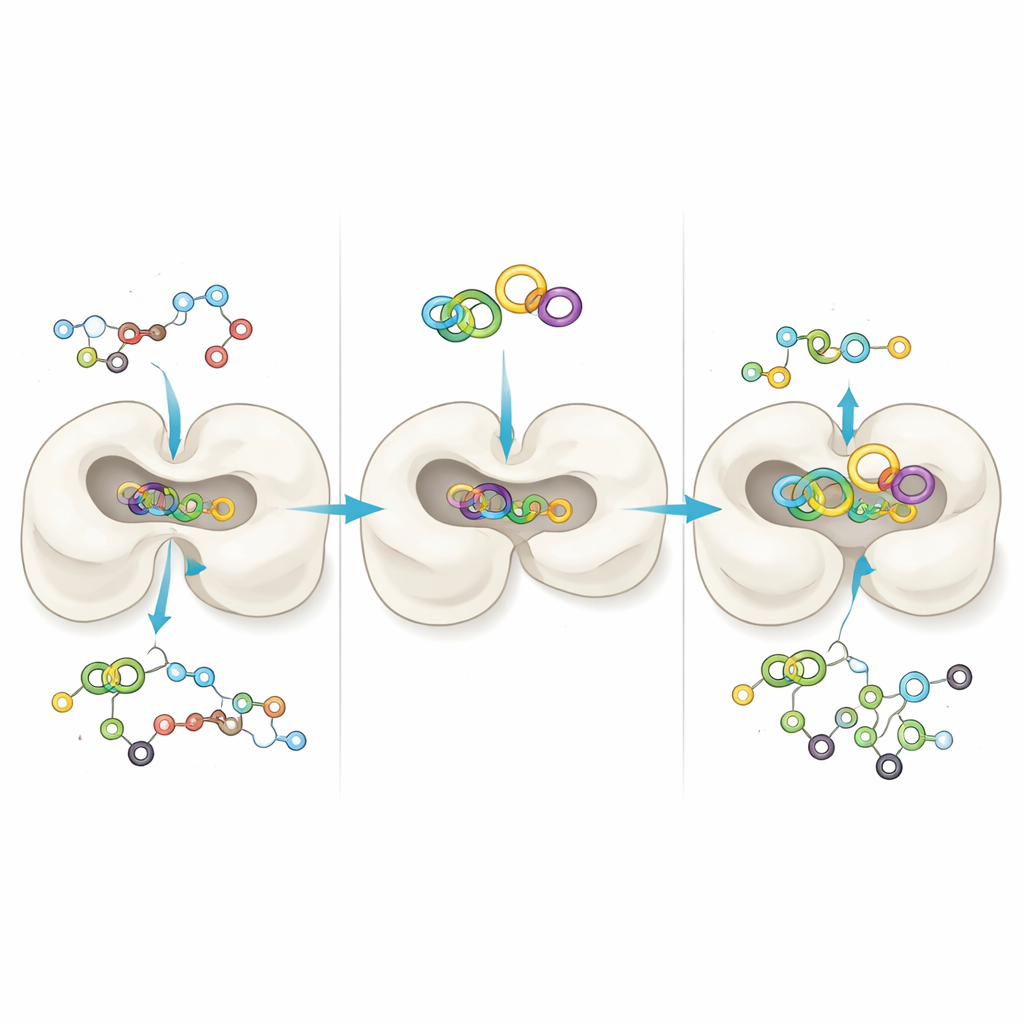
Watching the enzyme and plant molecule move together
Docking studies give a snapshot, but real proteins flex and breathe. To see how stable the match between COMT and gallacetophenone might be over time, the researchers ran long molecular dynamics simulations—virtual movies that follow every atom in the enzyme and the bound compound over hundreds of nanoseconds. They compared COMT alone (or with a standard reference molecule) to COMT bound to gallacetophenone. Measures of movement and shape, such as how much the protein structure drifted, how compact it stayed, and how its surface interacted with water, all showed that the plant compound formed a steady, well‑fitted complex. Additional energy calculations and statistical analyses of the motions suggested that gallacetophenone helps “lock” COMT into a stable form, reinforcing the idea that it could be a strong and reliable blocker.
What this could mean for future Parkinson’s care
For non‑specialists, the key message is that a naturally occurring molecule from tamarind shows strong computer‑based signs of being able to safely tame an enzyme that shortens the action of Parkinson’s medicine. By binding tightly to COMT and appearing stable and well‑tolerated in simulations, gallacetophenone emerges as a promising starting point for a new class of supportive drugs designed to keep dopamine—and levodopa—working longer in the brain. However, these results are still predictions on a screen, not proof in patients. The compound must now be tested in test tubes, cells, and animal models, and eventually in clinical trials, before anyone can know whether this tamarind‑derived candidate can truly make life better for people living with Parkinson’s disease.
Citation: Shenoy, A.G., John, A., Ravi, V. et al. Evaluation of phytochemicals from Tamarindus indica as a potential catechol-O-methyltransferase (COMT) inhibitor: an in-silico approach for Parkinson’s disease. Sci Rep 16, 14227 (2026). https://doi.org/10.1038/s41598-026-41470-x
Keywords: Parkinson’s disease, dopamine, COMT inhibitors, tamarind phytochemicals, drug discovery