Clear Sky Science · en
Micronucleus quantification from whole-slide haematology images using AI serves as a translatable pharmacodynamic biomarker for DNA damage response inhibitors
Why tiny dots in blood can tell big stories
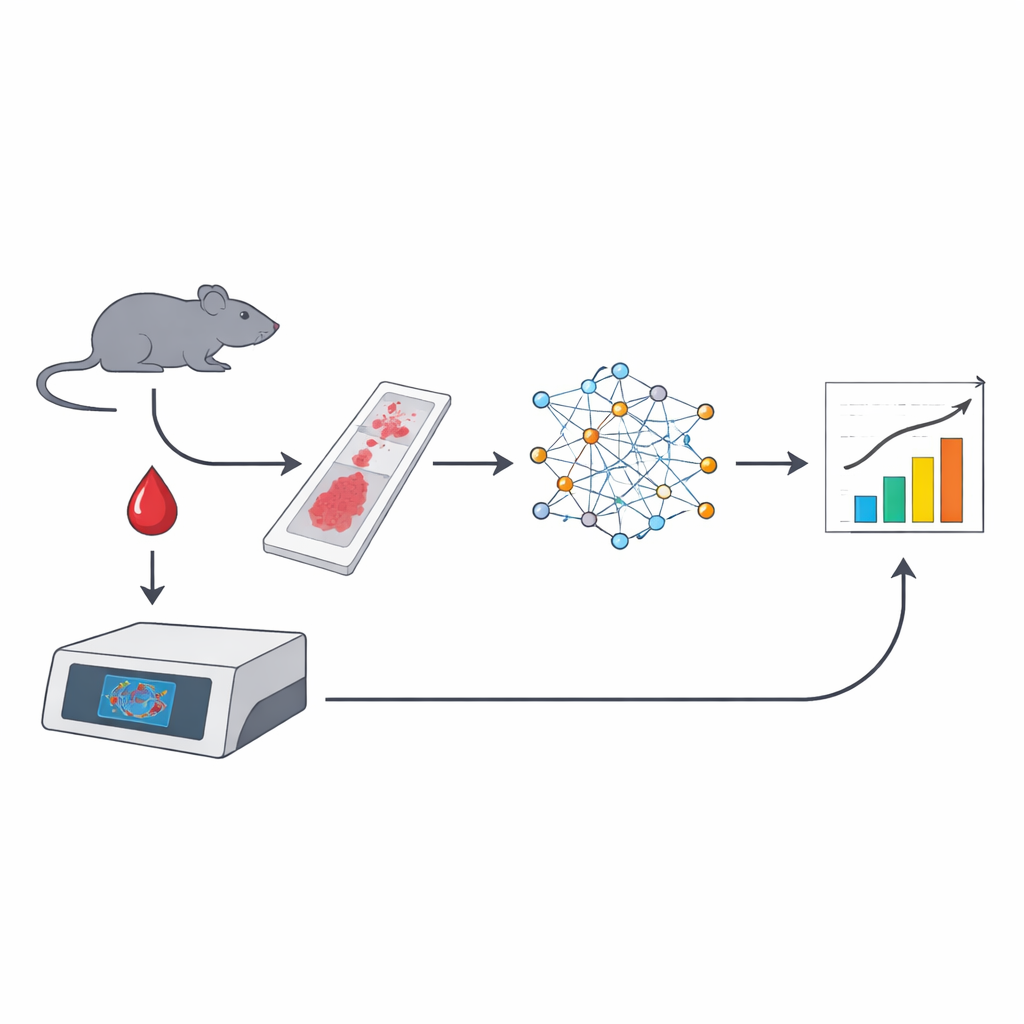
Doctors and scientists are always looking for simple ways to see whether a cancer drug is doing what it is supposed to do inside the body. This study shows that tiny specks inside red blood cells, called micronuclei, can act like natural trackers of DNA damage caused by certain cancer treatments. By combining microscope-style whole-slide imaging with artificial intelligence (AI), the researchers turned a few drops of blood into a rich, automated readout of how strongly DNA-targeting drugs are working.
Small islands of DNA in red blood cells
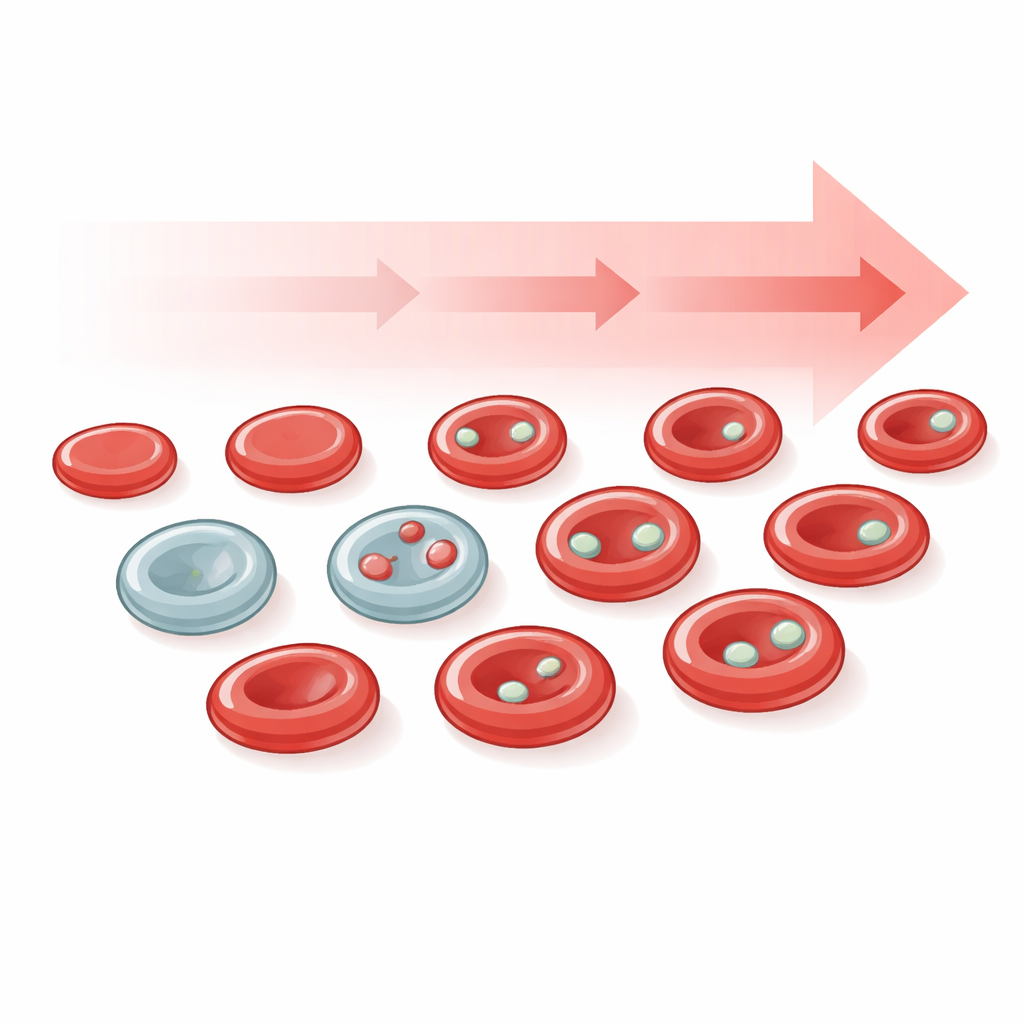
Micronuclei are little bubbles of DNA that sit apart from the main nucleus of a cell. They form when chromosomes break or are mis-sorted during cell division, events that are common in cancer and after exposure to DNA-damaging agents. In most tissues, spotting real micronuclei under the microscope is tricky because thin tissue slices can fool the eye. Red blood cells offer a workaround: as they mature, they normally spit out their main nucleus, but any micronuclei that formed earlier can be left behind as tiny, well-defined dots. These long-known structures, historically called Howell–Jolly bodies, are thus convenient, genuine micronuclei that circulate in the bloodstream and can reveal how much DNA damage has recently occurred.
Turning blood smears into data with AI
The team built an automated pipeline that starts with standard blood smears from mice, stained either with a fluorescent DNA dye or with common May–Grünwald Giemsa dyes used in hospital laboratories. Whole slides were scanned at high resolution, and supervised deep-learning models were trained to recognise red blood cells, pick out micronuclei, and distinguish immature reticulocytes from mature erythrocytes based on subtle differences in size and colour. From a single image, the system could analyse more than 100,000 cells, flag those carrying micronuclei and even count cells that contained more than one micronucleus—something conventional flow cytometry cannot easily do.
Connecting tiny changes in blood to drug strength
The researchers then asked whether the frequency of micronucleus-positive red blood cells tracked with exposure to DNA-damaging chemotherapy and to drugs that block the DNA damage response, such as PARP inhibitors. In mice treated with cisplatin or paclitaxel, the AI-based counts captured expected rises in micronucleated cells. In tumour-bearing mice treated with the PARP inhibitors olaparib or saruparib, higher doses produced larger increases in micronucleus-carrying cells and stronger tumour shrinkage. Over several weeks of daily dosing, small serial blood samples (only about 5 microlitres each) allowed the team to follow how micronuclei slowly accumulated and then declined after treatment stopped. When they compared their slide-based method with standard flow cytometry, the two approaches agreed closely, showing that the new workflow is both accurate and robust.
Zooming in on young blood cells for sharper signals
Because young red blood cells (reticulocytes) are especially likely to contain fresh micronuclei, the investigators trained their algorithms to split the red cell population into immature and mature cells on routine stained slides. This refinement revealed that micronuclei were far more enriched and more dynamic in reticulocytes than in older cells, providing a more sensitive and rapidly changing signal of drug activity. The whole-slide images also made it possible to monitor sample quality itself—for example, detecting red cells that had become misshapen after poor storage—so that substandard slides could be flagged before analysis, an important step for eventual clinical use.
What this could mean for future cancer care
The study demonstrates that a tiny blood sample, a standard slide stain and an AI-powered imaging pipeline can together deliver a scalable, minimally invasive measure of how strongly DNA-targeting drugs are affecting the body. While the work was done in mice and red blood cells are only a stand-in for what happens inside tumours, the strong link between micronucleus signals and tumour response suggests this approach could become a practical tool in drug development and, eventually, in clinical trials. In simple terms, counting these microscopic DNA "dots" in blood may help researchers and clinicians tune doses, compare new medicines and follow treatment effects over time—without repeated, invasive biopsies.
Citation: Yong, K.H.R., Robak, W.S., Mulderrig, L. et al. Micronucleus quantification from whole-slide haematology images using AI serves as a translatable pharmacodynamic biomarker for DNA damage response inhibitors. Sci Rep 16, 11437 (2026). https://doi.org/10.1038/s41598-026-41458-7
Keywords: micronuclei, red blood cells, PARP inhibitors, digital pathology, DNA damage response