Clear Sky Science · en
Efficient adsorption of perfluorooctane sulfonate and perfluorobutane sulfonic acid from aqueous solution onto amine-functionalized diatomite granules
Why stubborn chemicals in water matter
Many everyday products—from nonstick pans and takeout boxes to firefighting foams—leave behind a class of industrial compounds often called “forever chemicals.” These substances, known as PFAS, do not easily break down in nature and can build up in our bodies, where they have been linked to cancer, immune problems, and developmental issues. The study summarized here explores a simple, low-cost filter material made from fossilized algae that can strip two common PFAS chemicals out of water with high efficiency and be reused many times.
A new twist on a natural filter
The researchers focused on diatomaceous earth, a powder formed from the fossil shells of microscopic algae. On its own, this material is cheap, abundant, porous, and already used in some filters, but it does not grab PFAS strongly enough. To improve its performance, the team formed the powder into small granules and coated their surfaces with a molecule containing nitrogen-rich groups that can carry positive charges. These treated granules, called APTES-DE, provide many active spots that can attract the negatively charged heads of PFAS molecules while still preserving the porous structure that allows water to flow through.
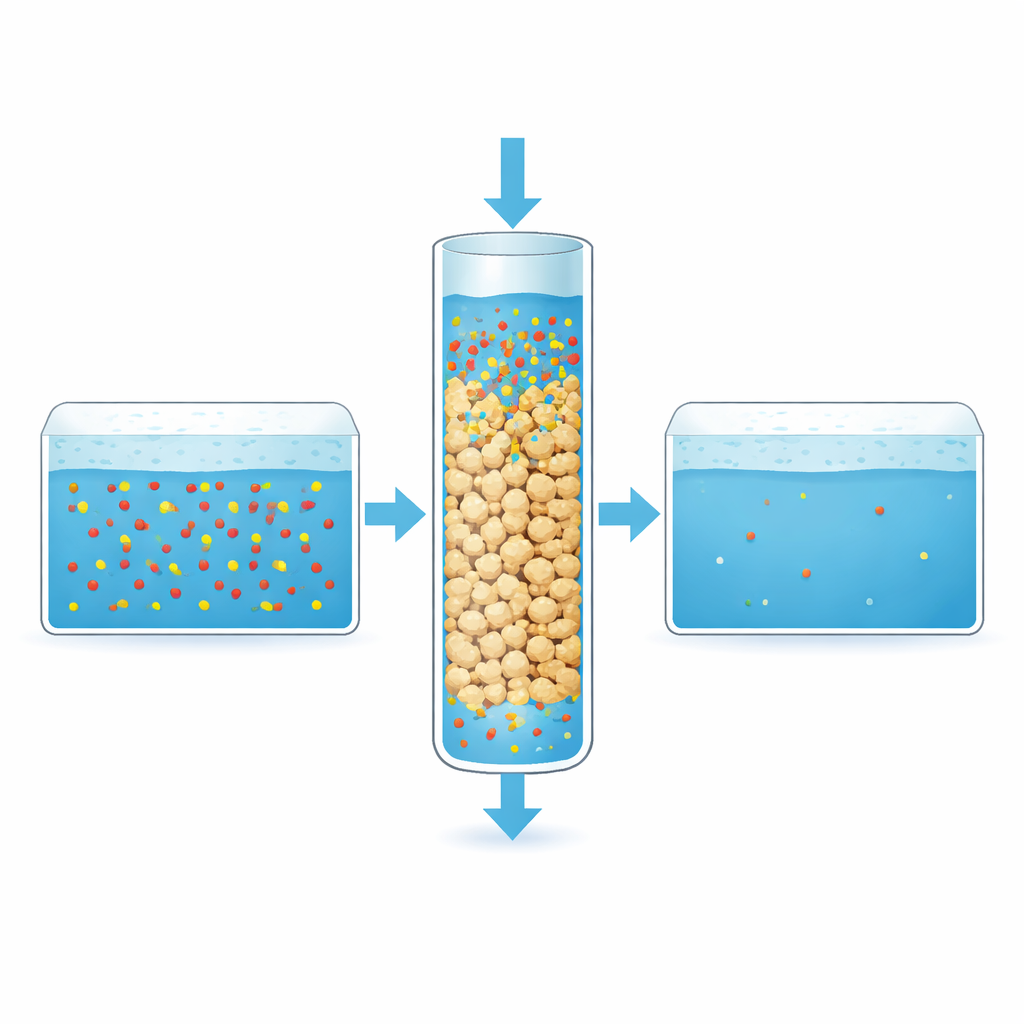
Putting the granules to the test
The scientists examined the modified granules with several imaging and surface-analysis tools to confirm that the new coating was present and that the internal pores were still accessible. They then tested how well APTES-DE could remove two widely detected PFAS compounds from water: PFOS, which has a longer fluorinated chain, and PFBS, a shorter version. In stirred batch experiments, the granules removed up to about 92% of PFOS and 90% of PFBS under optimized conditions. The data showed that PFAS formed multiple layers on the irregular surface rather than a single, uniform coating, consistent with a patchy landscape of strong and weak binding sites across each grain.
How the removal process works
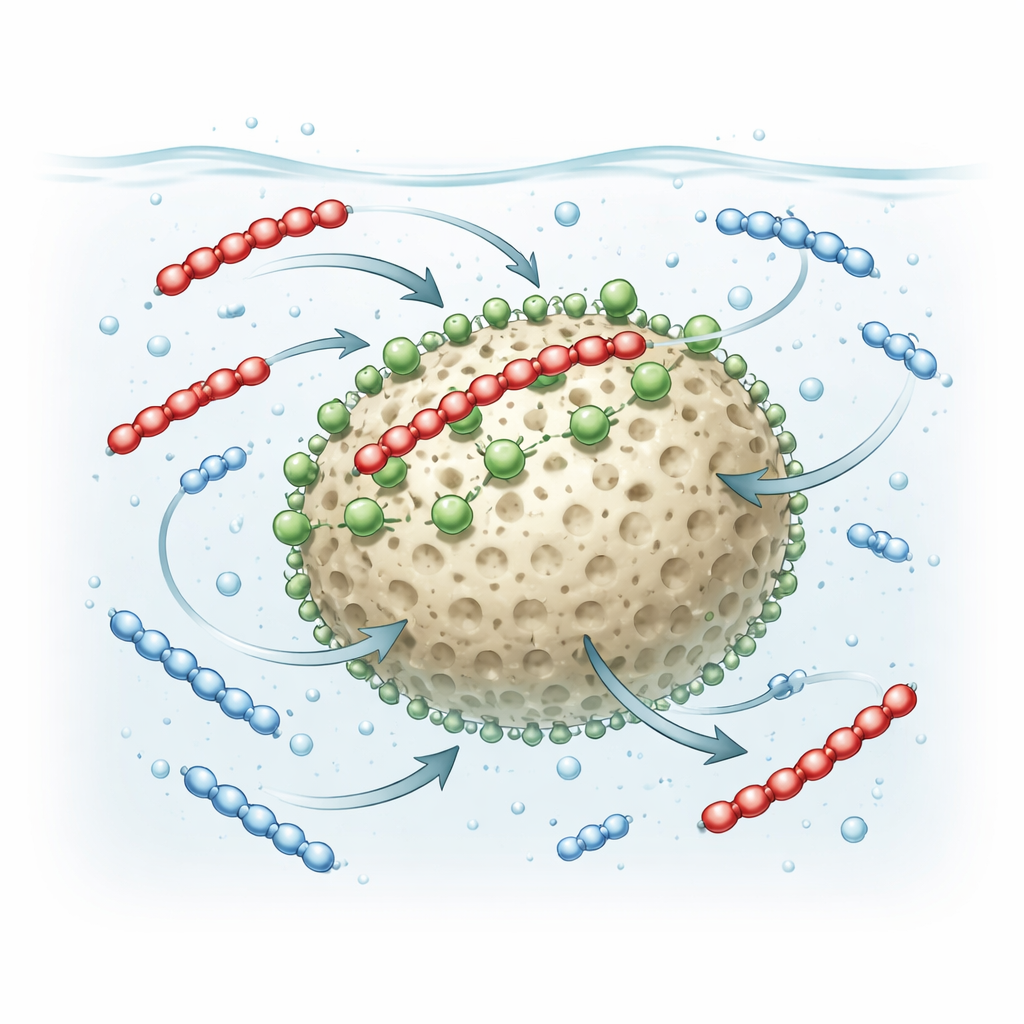
By varying contact time, concentration, water acidity, and flow conditions, the team teased apart the main forces behind PFAS capture. At mildly acidic to neutral pH, the nitrogen groups on the granules become positively charged, pulling in the negatively charged PFAS heads through electrostatic attraction. At the same time, the water-repelling fluorinated tails of PFAS are drawn to the relatively less watery microenvironment on the granule surface, adding a hydrophobic “stickiness.” Hydrogen bonds between PFAS and surface groups on the silica further stabilize these attachments. Together, these forces create a strong, cooperative grip, especially on longer-chain PFAS, which present more hydrophobic surface to latch onto the granules.
From lab beaker to flowing columns
Because real treatment plants use continuous flow rather than still tanks, the researchers packed the APTES-DE granules into transparent columns and pumped PFAS-contaminated water through them. Taller beds of granules held onto PFAS for longer times, delaying the moment when pollutants began to appear at the outlet and boosting overall removal. Slower flow rates also improved performance by giving PFAS more time to reach and bind to the granule surfaces. Throughout these trials, PFOS consistently attached more strongly and broke through later than PFBS, underlining how chain length influences capture in practical systems.
Reusing the filter again and again
For any filter to be useful at scale, it must be cleaned and reused without losing much capacity. Here, the saturated APTES-DE granules were rinsed with a mixture of alcohol and a mild alkaline solution, which loosened the PFAS and washed them away. After five cycles of adsorption and regeneration, the granules still removed more than 95% of their original PFOS capacity and about 96% for PFBS, showing only small declines. This durability, combined with high removal efficiencies and the use of a naturally abundant starting material, suggests that amine-modified diatomaceous earth could become a practical and more sustainable option for stripping stubborn PFAS pollutants from contaminated waters.
Citation: Bano, A., Prasad, B., Dave, H. et al. Efficient adsorption of perfluorooctane sulfonate and perfluorobutane sulfonic acid from aqueous solution onto amine-functionalized diatomite granules. Sci Rep 16, 10368 (2026). https://doi.org/10.1038/s41598-026-41388-4
Keywords: PFAS removal, water purification, diatomaceous earth, adsorption, forever chemicals