Clear Sky Science · en
Linking cortical morphology and neurophysiological dynamics in Parkinson’s disease
Why Brain Shape Matters in Parkinson’s
Parkinson’s disease is usually described as a problem of brain chemistry, especially the loss of the messenger dopamine. But chemicals are only part of the story. This study asks a deeper question: how does the physical shape of the brain’s surface relate to the electrical rhythms that go wrong in Parkinson’s? By linking structure and activity in living patients during surgery, the researchers aim to uncover new clues that could improve diagnosis and guide brain‑stimulation therapies.
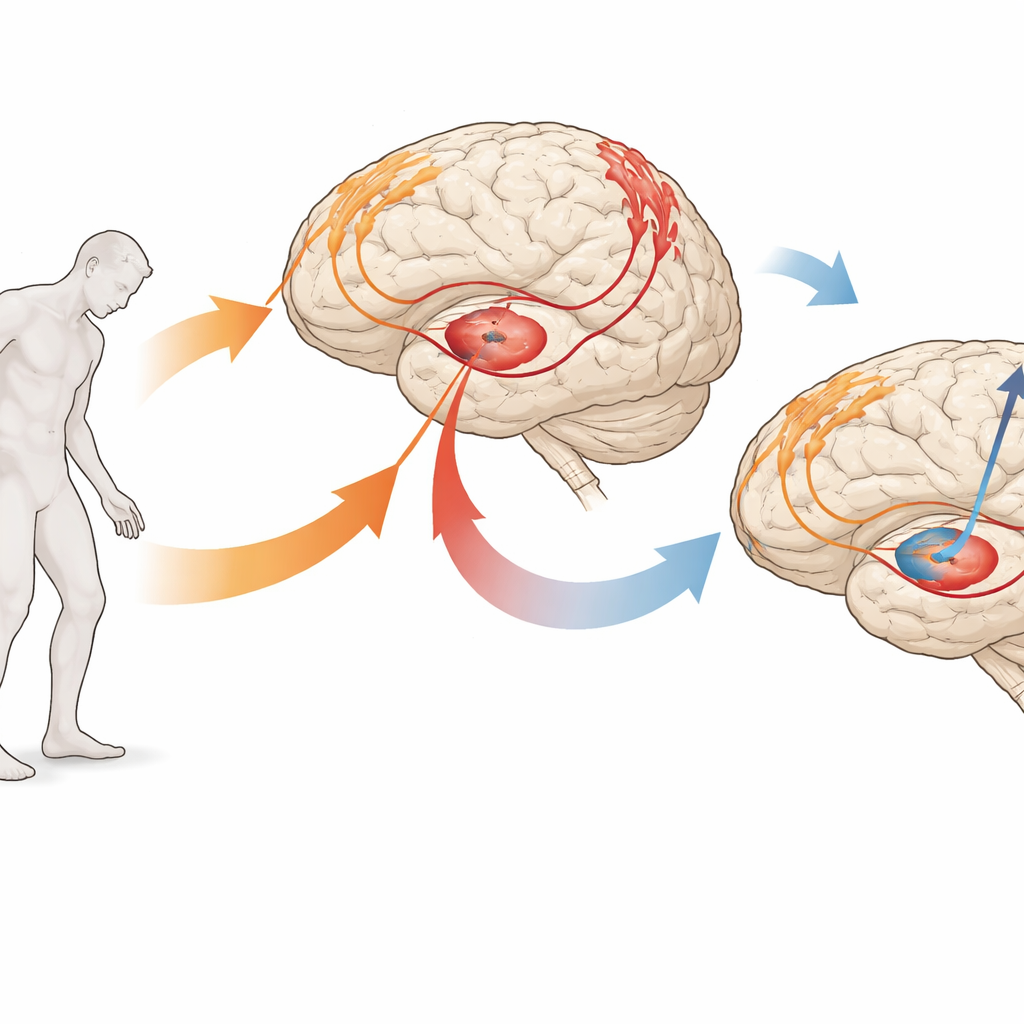
Looking at the Brain from Two Angles
The team studied 50 people with Parkinson’s who were undergoing deep brain stimulation surgery. Before surgery, each volunteer received detailed MRI scans that allowed the scientists to measure the thickness, surface area, and volume of key regions on the brain’s outer layer involved in movement and touch. During surgery, while patients were awake, the researchers recorded tiny electrical signals from the motor areas on the brain’s surface and from a deep structure called the globus pallidus internus, a common stimulation target. These electrical traces capture the brain’s natural rhythms, including brief "bursts" of activity in different frequency bands often linked to Parkinson’s symptoms.
From Simple Links to Hidden Patterns
At first, the scientists used straightforward pairwise comparisons, asking whether any single structural measure was directly tied to a single electrical feature. They did find some meaningful links—for example, how long bursts lasted or how strongly areas synchronized sometimes tracked the size or thickness of nearby cortex. But overall, this approach yielded a patchy, fragmented picture. Parkinson’s involves many interconnected changes happening at once, so the authors suspected that looking at one measure at a time was missing the bigger story.
Uncovering a Shared Brain Signature
To capture that bigger story, the team turned to a multivariate method called sparse partial least squares, which looks for combinations of structural and electrical features that move together across patients. This analysis revealed a strong hidden dimension that tied the two worlds together. On the structural side, this latent pattern was dominated by thinning of sensorimotor regions—areas that help process touch and plan and control movement. On the electrical side, it was driven by complex changes in burst dynamics in the alpha, low‑beta, and gamma ranges, including how often bursts occurred, how long they lasted, and how strong they were in both cortex and globus pallidus. Patients whose sensorimotor cortex was thinner tended to show a characteristic shift in these rhythmic bursts.
Beyond Age and Disease Stage
One concern was that this shared pattern might simply reflect aging or how long someone had lived with Parkinson’s. The researchers tested this by mathematically removing the influence of age, years since diagnosis, and standard clinical scores of movement problems. Even after doing so, the link between structure and electrical activity remained strong. Interestingly, the overall latent pattern was not tightly tied to current symptom ratings, suggesting it may capture a more basic property of how the disease reshapes brain circuits rather than just a snapshot of symptom severity on a given day.
What This Means for Patients and Care
For a layperson, the main message is that Parkinson’s is not only a matter of lost brain chemicals or abnormal rhythms, and not only a matter of brain shrinkage—it is about how these two aspects are woven together. This study shows that thinning in specific movement‑related regions of the cortex goes hand in hand with distinctive changes in the brain’s electrical bursts, forming a stable structural–functional signature of the disease. In the future, combining MRI measures of brain shape with recordings of brain rhythms could help doctors detect Parkinson’s earlier, monitor its progression more precisely, and fine‑tune deep brain stimulation or other treatments to each person’s unique brain wiring.
Citation: Mirpour, K., Alijanpourotaghsara, A. & Pouratian, N. Linking cortical morphology and neurophysiological dynamics in Parkinson’s disease. Sci Rep 16, 12164 (2026). https://doi.org/10.1038/s41598-026-41274-z
Keywords: Parkinson’s disease, brain structure, brain rhythms, deep brain stimulation, neural biomarkers