Clear Sky Science · en
Corrosion inhibition mechanism of a functionalized schiff base–derived quaternary ammonium salt for carbon steel in 1 M HCl: electrochemical, adsorption, and theoretical studies
Why Protecting Everyday Steel Matters
From oil pipelines deep underground to storage tanks at refineries, much of our energy infrastructure relies on a simple workhorse material: carbon steel. Yet this metal has an Achilles’ heel—when it meets strong acids, it can rust away quickly, risking leaks, failures, and costly shutdowns. This study explores a newly designed chemical additive that can be mixed into acidic cleaning solutions to form an invisible shield on steel, dramatically slowing its decay while equipment is being cleaned.
A Silent Threat Inside Industrial Pipes
In the petroleum industry, operators often clean pipes, tanks, and heat exchangers with hydrochloric acid to remove crusty mineral deposits. While this acid wash restores flow, it also attacks the steel itself, eating away the metal and creating pits and cracks. Replacing corroded sections is expensive and disruptive, and in the worst cases corrosion can lead to leaks or catastrophic failures. To reduce this damage, companies add corrosion inhibitors—molecules that stick to the metal and act like a raincoat, keeping the acid at bay. The challenge is to design inhibitors that are powerful, work at low doses, remain effective at high temperatures, and are reasonably safe and easy to make.
A Tailor-Made Chemical Shield
The research team created a new inhibitor called Q-Ar, built from a family of compounds known as Schiff bases and then converted into a positively charged quaternary ammonium salt. This architecture gives Q-Ar many “sticky” sites—nitrogen and oxygen atoms and flat ring systems—that can latch onto steel. Lab tests confirmed the molecule’s structure, and the scientists dissolved small amounts of Q-Ar (as little as a few parts per million) into 1 molar hydrochloric acid, a strong acid similar to those used in industrial cleaning. They then exposed carbon steel samples to this acid with and without Q-Ar and measured how fast the metal dissolved using sensitive electrochemical techniques.
How the Protective Film Performs
When no inhibitor was present, the steel rapidly lost material as the acid stripped away atoms from its surface. With Q-Ar added, both the metal’s tendency to dissolve and the reaction that releases hydrogen gas were strongly suppressed. At a concentration of just 35 parts per million, Q-Ar cut the corrosion rate by about 94 percent at room temperature. Electrical measurements showed that the steel’s resistance to charge transfer—a key step in corrosion—increased more than tenfold, while the apparent electrical “capacitance” of the interface dropped, a fingerprint of a thicker, more insulating film forming on the surface. Microscopy images supported this picture: steel left in acid alone became rough and covered with corrosion products, whereas steel treated with Q-Ar remained relatively smooth and clean after hours in acid, with fewer iron oxides detected.
Peering Into the Invisible Bonding
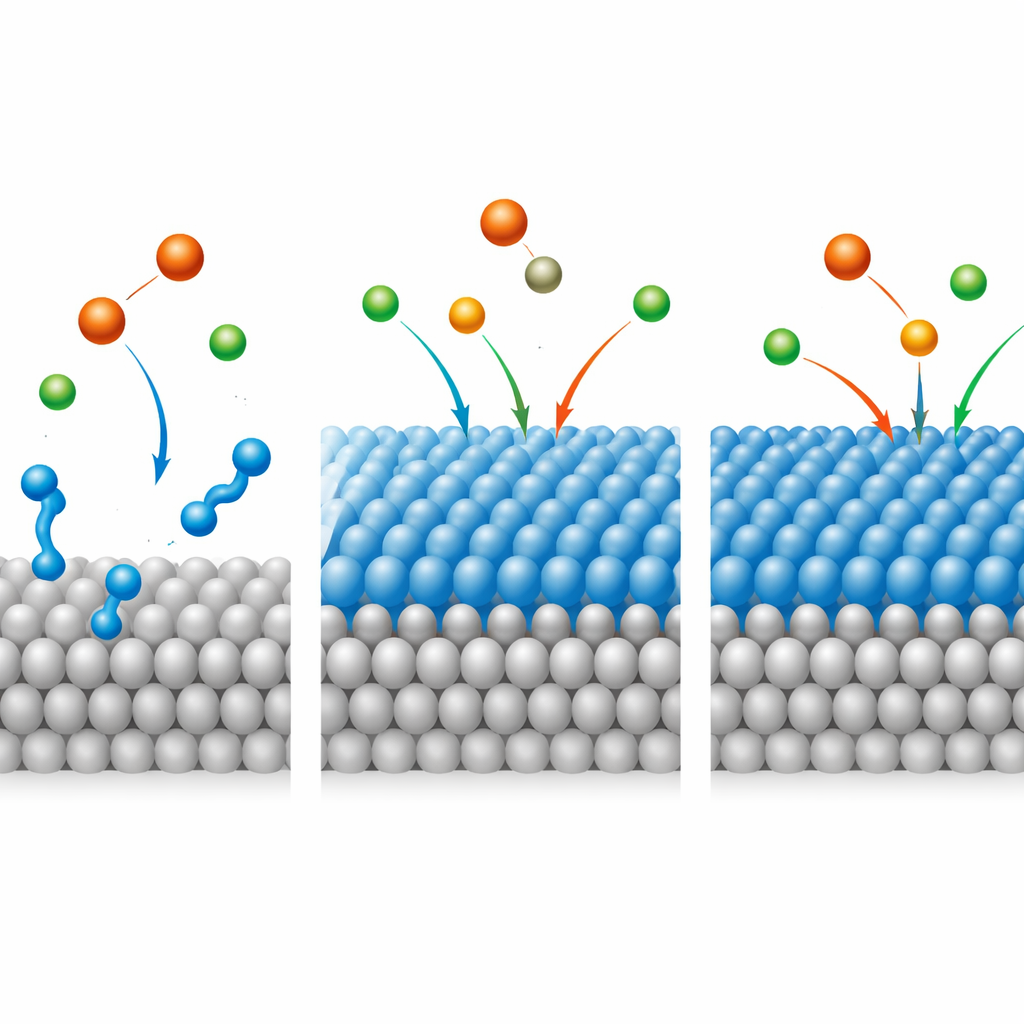
To understand why Q-Ar sticks so well, the researchers turned to computer modeling. Quantum chemical calculations revealed that the molecule has a small energy gap between its key electron orbitals, meaning it can readily share electrons with the iron atoms in steel. Simulations of Q-Ar lying flat on an idealized iron surface showed strong attraction energies and a snug, parallel orientation, ideal for building a dense protective layer. The analysis suggests that Q-Ar first attaches through electrostatic attraction—its positively charged centers are drawn to negatively charged sites near the steel—then strengthens this grip through true chemical bonding, where electrons are shared between the molecule and the metal. This mixed physical-and-chemical anchoring helps the film stay in place even as temperature rises and hydrogen bubbles form during acid attack.
What This Means for Real-World Use
Overall, the study shows that Q-Ar can form a tight, durable coating on carbon steel in harsh acidic environments, sharply slowing corrosion at very low doses. Because the inhibitor works on both main branches of the corrosion reaction and remains effective over higher temperatures and longer exposure times, it could help extend the life of pipelines and processing equipment during routine cleaning. While further work is needed to fully assess its environmental impact and performance in field conditions, the findings demonstrate how carefully designed molecules can act as molecular body armor for everyday metals, turning aggressive acids into more manageable tools rather than destructive forces.
Citation: Ahmed, M.I., Abd-El-Raouf, M., Migahed, M. et al. Corrosion inhibition mechanism of a functionalized schiff base–derived quaternary ammonium salt for carbon steel in 1 M HCl: electrochemical, adsorption, and theoretical studies. Sci Rep 16, 11618 (2026). https://doi.org/10.1038/s41598-026-41236-5
Keywords: carbon steel corrosion, acid cleaning, corrosion inhibitor, quaternary ammonium salt, surface adsorption