Clear Sky Science · en
A multihued sustainable appraisal of the electrochemical method for synchronized micro-estimation of the household drug Paracetamol with Aceclofenac or Dicyclomine
Why everyday pain pills need smart testing
Paracetamol, better known as a common pain and fever reliever, is one of the most widely taken medicines on the planet. It is often packed into combination tablets with other drugs to ease joint pain or stomach cramps. Making sure each tablet really contains the right tiny amounts of each ingredient is essential for safety and trust—but traditional testing methods can be slow, costly, and not very kind to the environment. This study introduces a quicker, cheaper, and greener way to check such tablets using electricity instead of large volumes of chemicals.
Everyday medicines under the microscope
The researchers focused on paracetamol combined with two partners frequently used in clinics: aceclofenac, an anti-inflammatory drug used for arthritis and joint pain, and dicyclomine, a drug that eases painful muscle spasms in the gut. In real life, these medications are sold together in a single tablet, so quality-control labs need methods that can measure both ingredients at once, even when they are present in very small amounts. Existing laboratory techniques often require expensive instruments, long analysis times, and large quantities of organic solvents, which generate chemical waste.
Measuring drugs with tiny electrical signals
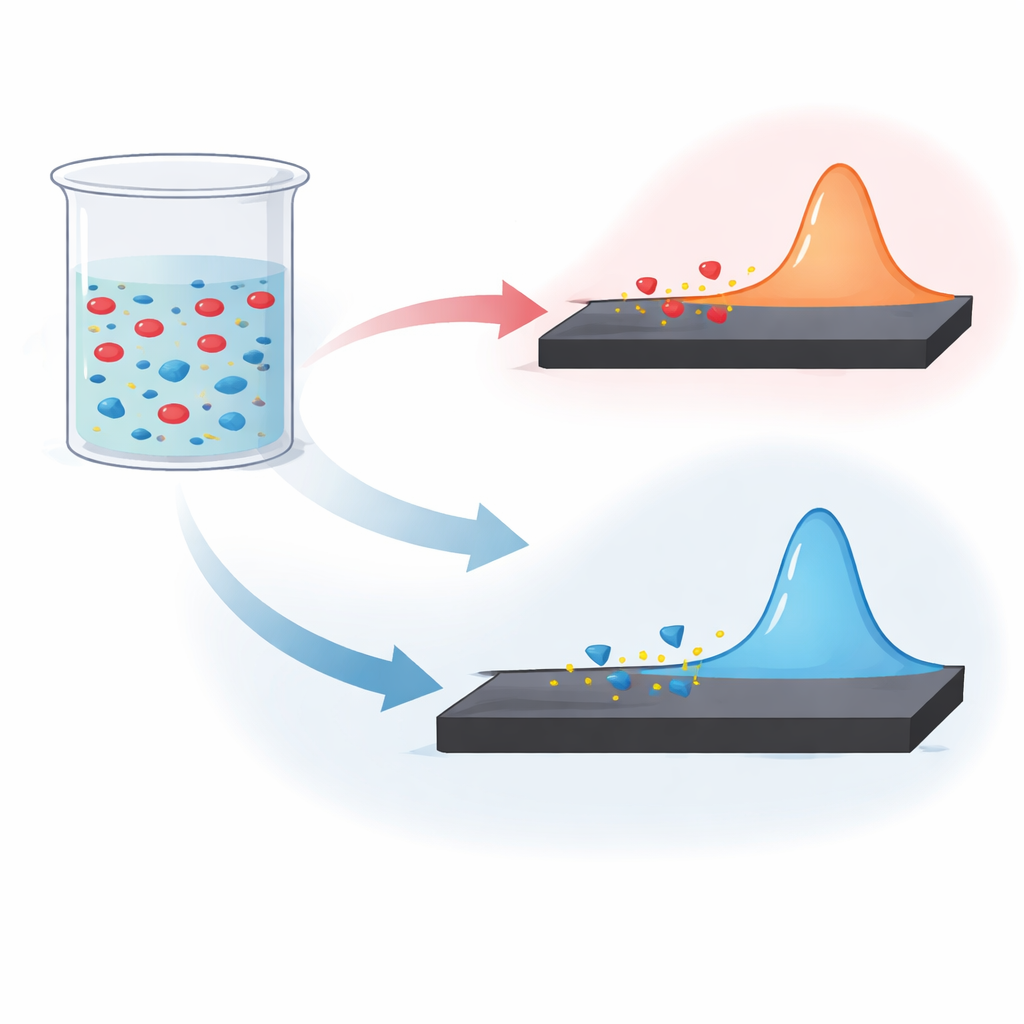
Instead of relying on large chromatography machines, the team used an electrochemical approach called differential pulse voltammetry. In simple terms, they dipped a small, unmodified piece of glassy carbon—acting as an electrical sensor—into a buffered water-based solution of the drug mixture and applied a series of tiny voltage pulses. Each drug in the mixture produces its own characteristic electrical “peak” when it loses electrons at the surface of the sensor. By tuning the acidity or basicity of the solution, they arranged conditions so that paracetamol and aceclofenac give separate peaks in an acidic solution, while paracetamol and dicyclomine separate cleanly in an alkaline one. This allows both ingredients in each pair to be measured simultaneously from the same run.
Fast, precise, and reliable results
The method was carefully optimized—adjusting solution pH, pulse size, timing, and scan settings—to maximize the size and separation of the electrical peaks. Under these conditions, the signal from each drug increased smoothly with concentration over a wide range, and the statistical “goodness of fit” was extremely high. The smallest detectable amounts were a fraction of a microgram per milliliter, showing very good sensitivity. When the researchers repeatedly tested standard solutions and laboratory-made tablets that mimic commercial products, the measurements were highly consistent from day to day, with errors and variability generally below two percent. Common tablet fillers did not interfere with the signals, meaning the method can analyze real dosage forms without extra purification steps.
Greener chemistry and global goals
Beyond accuracy, the authors set out to judge their method by how gentle it is on people and the planet. They compared it with established liquid chromatography and thin-layer chromatography techniques using several modern “greenness” and “sustainability” scoring tools. Because the new approach uses mainly water-based buffers, tiny sample volumes, no complex sensor coatings, and only a few minutes of instrument time per test, it earned higher scores for reduced chemical hazard, lower energy use, and better practicality. Using a recently proposed Need–Quality–Sustainability index, which links laboratory practices to the United Nations Sustainable Development Goals, the method scored highly for genuine need, technical quality, and contribution to more sustainable pharmaceutical analysis.
What this means for patients and the planet
For the lay reader, the take-home message is that the study showcases a way to check what is really inside combination pain tablets using a small, simple, and largely water-based electrical test instead of more resource-heavy techniques. The method can confirm in a matter of minutes that each pill contains the correct micro-quantities of paracetamol and its partner drug, while generating less waste and consuming fewer solvents and less energy. Although it is limited to ingredients that respond to electrical probing in water, this strategy offers a promising blueprint for routine, greener quality control of many everyday medicines—supporting both reliable healthcare and broader environmental and sustainability goals.
Citation: Ahmed, A.R., Ragab, M.A.A., Korany, M.A. et al. A multihued sustainable appraisal of the electrochemical method for synchronized micro-estimation of the household drug Paracetamol with Aceclofenac or Dicyclomine. Sci Rep 16, 9673 (2026). https://doi.org/10.1038/s41598-026-41215-w
Keywords: paracetamol combinations, electrochemical drug analysis, green analytical chemistry, pharmaceutical quality control, sustainable laboratory methods