Clear Sky Science · en
Personalized multi-agent reinforcement learning framework for adaptive chronic disease therapy management
Why smarter care for long-term illness matters
Millions of people live for years with conditions like diabetes, heart disease, and chronic kidney disease. Managing these illnesses is a daily balancing act involving medicines, diet, exercise, sleep, and stress. Doctors often rely on brief clinic visits and scattered records, which makes it hard to tailor treatment as a person’s life and body change. This paper explores how a new kind of privacy-respecting artificial intelligence could continuously learn from many patients at once while still giving highly personalized guidance to each individual.
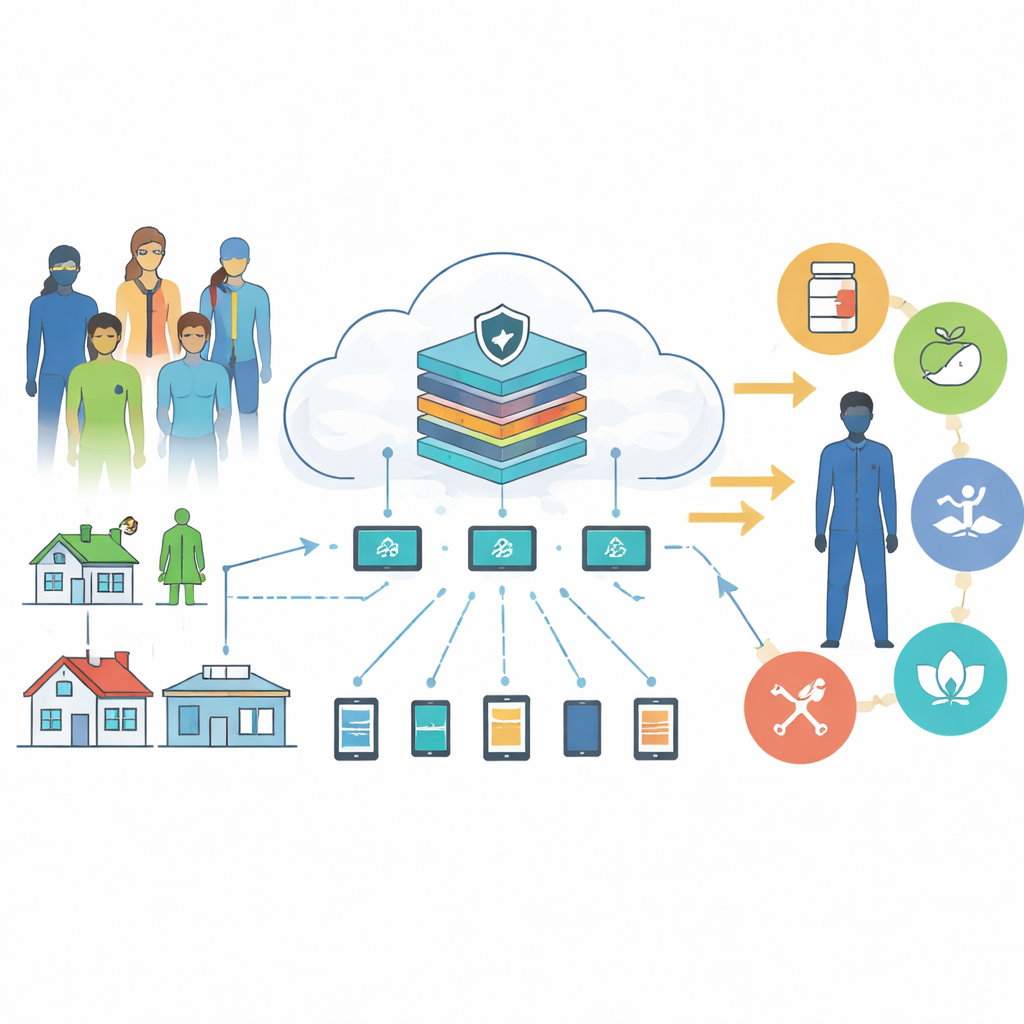
Many patients, many data streams, one shared brain
The authors start from a simple idea: people with chronic disease generate a constant stream of clues about their health—blood pressure readings, glucose levels, activity from wearables, clinic lab tests, and even sleep or mood logs. Today, most of that information sits in separate hospitals, apps, and devices. The proposed system links these sources through what is known as a federated learning network. Instead of sending raw patient data to a central server, each hospital or home device trains its own local prediction model and only shares the model’s parameter updates. A central computer then averages these updates into a stronger “global” model. This approach lets the system learn patterns from large, diverse populations while keeping personal records on-site, reducing privacy risk and communication cost.
A powerful predictor that understands patterns over time
To make sense of messy real-world health data, the framework uses a deep learning model that combines two strengths. One part, inspired by image-recognition networks, is good at pulling out important signals from complex inputs, such as combinations of lab results and sensor readings. The other part, borrowed from modern language models, is designed to track how things change over time—whether blood pressure is drifting up, for example, or how glucose responds across days. Together, this hybrid model turns each person’s history into a compact “embedding,” a numerical fingerprint that summarizes their current health state and future risk. In tests on two public datasets—one covering a wide range of chronic conditions and another focused on kidney disease—this predictor reached very high accuracy, around 98–99%, outperforming several recent artificial-intelligence tools.

Digital teammates that co-manage daily treatment
Prediction alone does not improve health; treatment decisions must also adapt over time. To address this, the authors add a personalized multi-agent reinforcement learning layer on top of the predictor. Instead of a single decision-maker, the system uses several cooperating software “agents,” each devoted to one aspect of care such as medication, diet, physical activity, or mental wellbeing. These agents watch the health fingerprint produced by the predictor, choose actions such as adjusting meal patterns or exercise intensity, and receive rewards when simulated outcomes improve—more stable glucose, healthier blood pressure, fewer side effects, and better quality-of-life indicators. Over many training episodes built from historical records, the agents learn coordinated strategies that favor long-term benefits rather than just short-term fixes.
Opening the black box for doctors and patients
Because medical decisions carry high stakes, the framework includes an explanation layer designed to show why the AI is reaching certain conclusions. The authors use a method that assigns each input feature—such as age, body weight, blood pressure, or activity level—a contribution score for a given prediction or treatment suggestion. Clinicians can view visual summaries that highlight which factors most strongly influenced a risk estimate or a recommended change in therapy. For example, the system might emphasize that recent blood pressure spikes and low activity are driving a higher risk score, and that this is why the activity agent proposes more walking while the medication agent avoids aggressive new drugs. This transparency aims to build trust, support shared decision-making, and help align AI suggestions with clinical guidelines.
What this means for people living with chronic illness
In experiments, the combined system proved more accurate, faster to learn good policies, and more communication-efficient than existing deep-learning or decision-making models used on their own. Importantly, it does this while keeping raw data close to where it is generated and by providing human-readable explanations of its choices. For patients, such a framework could eventually feel like an always-on, privacy-conscious digital coach that helps fine-tune treatment across medicines, meals, movement, and mental health, updating as their lives change. Although the current study relies on retrospective datasets and simulations, it points toward future health systems where advanced AI quietly supports doctors and patients in managing chronic disease more safely, personally, and effectively over the long term.
Citation: Ahmad, F., AlGhamdi, R. Personalized multi-agent reinforcement learning framework for adaptive chronic disease therapy management. Sci Rep 16, 11025 (2026). https://doi.org/10.1038/s41598-026-41093-2
Keywords: chronic disease management, personalized medicine, federated learning, reinforcement learning, explainable AI