Clear Sky Science · en
Hydroxyl radical production in liquid water by laser cavitation
Light That Tears Water Apart
Imagine using a tiny, invisible hammer made of light to smash water so hard that it creates powerful cleaning agents on the spot. This study explores exactly that idea. By firing short laser pulses into water, the researchers generate intense, microscopic bubbles that burst with such violence that water molecules are ripped apart, forming hydroxyl radicals—highly reactive particles that can break down many kinds of pollutants. The work shows how to tune this process to make it more efficient, hinting at new ways to clean wastewater or drive chemical reactions using light instead of added chemicals.
Why Tiny Bubbles Matter
Modern wastewater treatment increasingly relies on “advanced oxidation,” a family of methods that create aggressive oxidizing agents to destroy stubborn contaminants. One of the most important of these agents is the hydroxyl radical, which reacts quickly and non‑selectively with many organic molecules, ultimately turning them into carbon dioxide, water, and harmless salts. Traditionally, these radicals are produced using sound waves, flowing liquids, or chemical additives, each with its own limits in control and efficiency. Laser cavitation offers a fresh route: a laser pulse focuses inside water, causing an explosive breakdown that produces a bright plasma and a rapidly expanding bubble. When this bubble grows and then collapses, it creates extreme temperatures and pressures that can split water molecules and generate radicals.
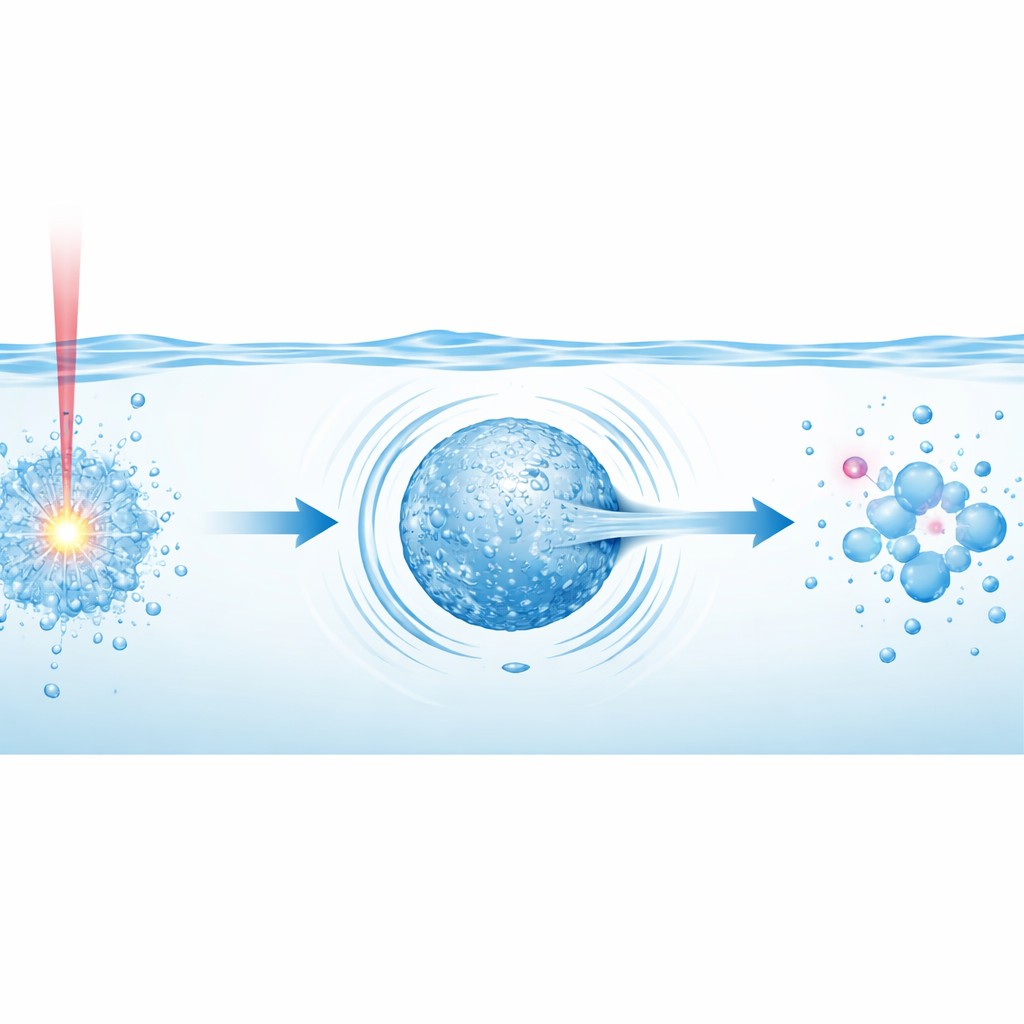
Following the Life of a Laser Bubble
The authors built a dedicated setup in which a pulsed Nd:YAG laser is focused into a small tube of dyed water placed above a black rubber surface that helps absorb energy. Using a high‑speed camera capturing hundreds of thousands of frames per second, they recorded the birth, growth, and collapse of single cavitation bubbles over three pulsation cycles. The first expansion–collapse cycle is the most violent: the water’s breakdown into plasma launches a laser shock wave, then the growing bubble stores energy that is suddenly released as it collapses, sending out a second shock wave and a fast, narrow water jet. Together, these mechanical impacts “tear” water molecules, producing hydroxyl radicals in a burst. Later pulsations are weaker, contributing much less to radical formation, so most useful chemistry happens in the very early moments after each laser shot.
Turning Color Loss into a Radical Meter
To measure how many radicals were formed, the team used a blue dye called methylene blue, which loses its color when attacked by hydroxyl radicals. By shining ultraviolet–visible light through the solution before and after laser treatment, they could see how much the color faded and, using a calibration curve, calculate how many radicals must have reacted. Systematic tests revealed that the dye concentration matters: too little dye and radicals mostly destroy each other; too much dye and it absorbs the laser light or interferes with the measurements. An intermediate concentration of 5 milligrams per liter struck the best balance, making the dye an effective chemical “sensor” for the radical output of the bubbles.
Finding the Sweet Spot for Bubble Power
The researchers then mapped how operating conditions influence radical production. Higher laser energy and higher pulse frequency both increased the total amount of radicals formed over an hour, because each shot generated stronger breakdown and larger, more energetic bubbles. However, when they accounted for how much laser energy was used, they found that lower energies and slower repetition rates were actually more efficient, producing more radicals per unit of input energy. Temperature also played a role: warming the water from 15 °C up to about 35–45 °C boosted radical yield, likely because bubbles grew larger and collapsed more violently, and molecules moved faster to meet the radicals. At still higher temperatures, cavitation became gentler and productivity dropped. Acidic water, especially similar to tap water rather than ultra‑pure water, and gentle stirring further aided radical production by providing more gas seeds for bubbles and keeping the reaction zone refreshed.
What This Could Mean for Cleaner Water
Overall, the study shows that carefully controlled laser pulses can generate substantial amounts of hydroxyl radicals directly in water, without adding chemical oxidants. Most radicals are created during the first, intense collapse of laser‑made bubbles, driven by the combination of laser shock waves, bubble‑collapse shock waves, and high‑speed water jets. By choosing moderate laser energies, low pulse frequencies, slightly warm and acidic tap water, and a flowing liquid, the authors achieved both high radical yields and good energy efficiency. While the technology is not yet ready for industrial cleanup on its own, it opens a promising path toward precisely tunable, contact‑free oxidation processes that could complement or enhance future water treatment and chemical manufacturing systems.
Citation: Zhou, X., Gu, J. Hydroxyl radical production in liquid water by laser cavitation. Sci Rep 16, 11251 (2026). https://doi.org/10.1038/s41598-026-41073-6
Keywords: laser cavitation, hydroxyl radicals, advanced oxidation, wastewater treatment, cavitation bubbles