Clear Sky Science · en
Enhanced photocatalytic and antibacterial performance of LaMnCeO3/TiO2 composites modified with Ti3C2Tx MXene for efficient methyl red degradation
Cleaning Colorful Water
Many factories release brightly colored dyes into rivers and lakes, where they can linger for a long time and harm ecosystems. Engineers are searching for ways to use light, rather than harsh chemicals, to break these dyes down into harmless substances. This paper describes a new light-activated material that strips a common red dye from water very efficiently, while leaving beneficial microbes largely unharmed—an attractive mix for real-world wastewater treatment plants.
A New Recipe for Light-Driven Cleanup
The researchers built a three-part "cleanup platform" by combining three advanced materials: titanium dioxide (a classic light-sensitive white powder), a perovskite oxide containing lanthanum, manganese, and cerium, and a sheet-like conductor called MXene. Together they form tiny composite particles a few billionths of a meter across, spread over thin, layered MXene sheets. Electron microscopy images show largely spherical particles anchored on these sheets, while elemental mapping confirms that all the key ingredients are evenly mixed. Measurements of crystal structure and surface chemistry indicate that the three components form a well-organized, porous network with many accessible nooks and crannies where reactions can occur.
Making the Most of Visible Light
On their own, many light-sensitive powders struggle because the electric charges they generate when illuminated quickly cancel each other out. By carefully adjusting how much lanthanum, manganese, and cerium they used, the team was able to tune how the composite absorbs light and moves charge. Optical tests revealed that different mixing ratios shift the energy gap of the material, changing how it responds to visible light. Electrical and glow-based measurements showed that when the particles sit on conductive MXene sheets, electrons can quickly flow away from their starting point instead of recombining with holes. This separation of charges is crucial, because it lets them meet oxygen and water at the surface and turn into aggressive short-lived oxidants.
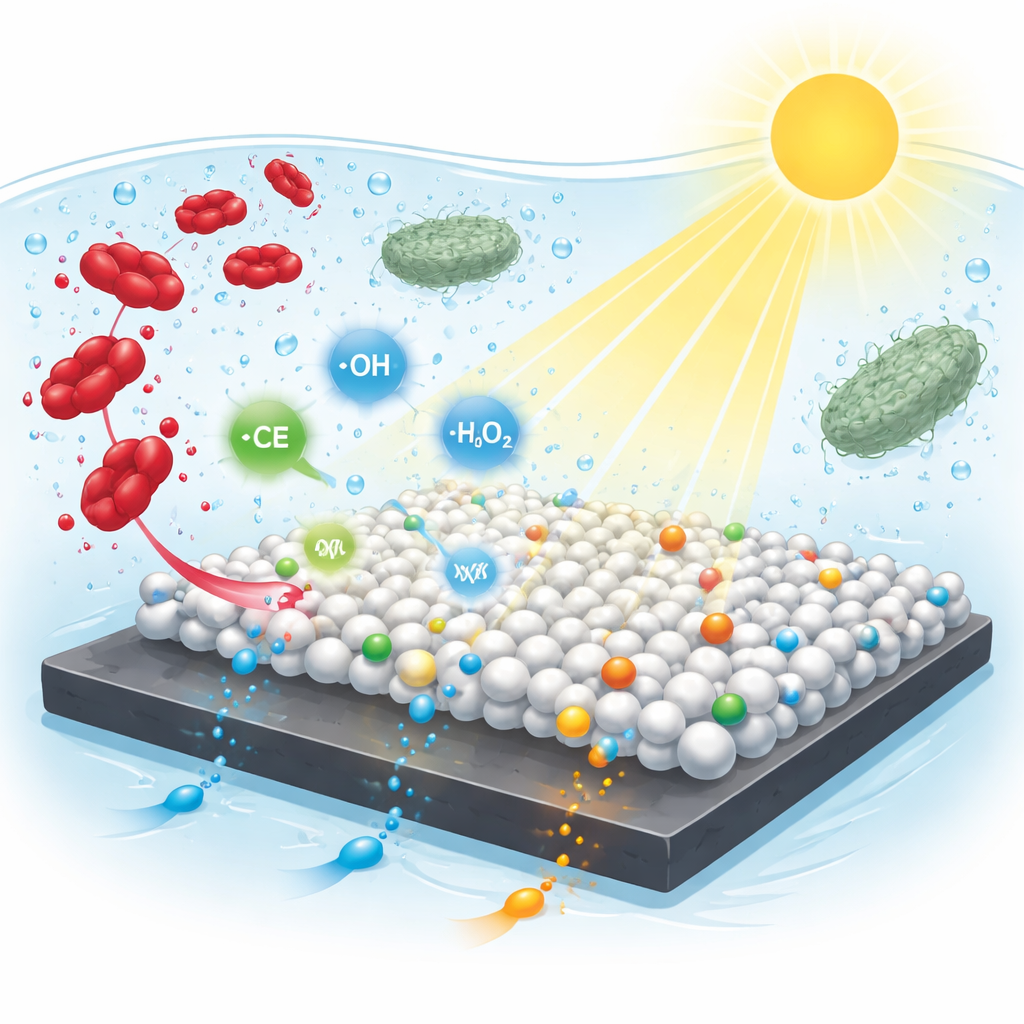
Breaking Down a Stubborn Red Dye
To test the new materials, the scientists focused on methyl red, a widely used laboratory dye and a stand-in for similar industrial colorants. When they shone visible light on water containing the dye and their nanocomposites, the red color faded rapidly. One version, labeled LMC-112, achieved the highest single-pass removal—over 95 percent under acidic conditions—while another, LMC-111, offered the best combination of efficiency and long-term stability. The team explored how acidity, temperature, dye concentration, and reaction time influence performance, and used statistical methods to identify operating conditions that maximize color removal while minimizing experimental effort. They also compared the three-part composite with each ingredient alone and found that the full combination consistently outperformed the separate parts, highlighting a strong synergistic effect.
How the Dye Gets Destroyed
Additional experiments probed which reactive particles actually do the work. By adding chemicals that selectively block different radicals, the researchers showed that hydroxyl radicals—highly reactive forms of oxygen—play the leading role, with superoxide and positive holes contributing to a lesser extent. In the proposed picture, visible light kicks electrons in the titanium dioxide and perovskite components into more energetic states. MXene then acts as a conductive highway, drawing electrons away and reducing oxygen at the surface, while the remaining holes help split water to generate hydroxyl radicals. These fleeting oxidants attack the dye’s nitrogen–nitrogen link and aromatic rings, turning the complex colored molecules into smaller, colorless fragments and eventually into carbon dioxide, water, and nitrate.
Gentle on Helpful Microbes
Many modern photocatalysts are designed to kill bacteria as well as degrade chemicals, but that can be a drawback in treatment systems that rely on healthy microbial communities. Here, tests against two model bacteria—one Gram-positive and one Gram-negative—showed essentially no antibacterial effect, even under light. No growth-free zones formed on culture plates, and liquid cultures continued to thrive in the presence of the nanocomposite. This biological neutrality suggests that the radical bursts near the catalyst surface are strong enough to chop up dye molecules but too localized or short-lived to seriously harm free-swimming microbes.
Promise for Real-World Water Treatment
Taken together, the results paint a picture of a sturdy, tunable material that uses visible light to scrub dyes from water, cycle after cycle, without acting as a broad-spectrum disinfectant. The carefully engineered blend of titanium dioxide, perovskite oxide, and MXene creates efficient charge separation and radical generation, while the mesoporous structure offers plenty of space for dye molecules to land and be attacked. For wastewater plants that must remove stubborn colorants yet preserve beneficial bacteria in downstream biological units, such a selective photocatalyst could become a valuable part of a greener treatment toolkit.
Citation: Parsafard, N., Riahi-Madvar, A. Enhanced photocatalytic and antibacterial performance of LaMnCeO3/TiO2 composites modified with Ti3C2Tx MXene for efficient methyl red degradation. Sci Rep 16, 12322 (2026). https://doi.org/10.1038/s41598-026-41059-4
Keywords: wastewater treatment, photocatalytic dye degradation, nanocomposite catalyst, visible light photocatalysis, environmental remediation