Clear Sky Science · en
Antimicrobial effects of novel Hermetia illucens peptides
Why tiny insect tools matter for our health
Antibiotic-resistant infections already kill millions of people each year, and many familiar drugs are slowly losing their power. This study explores an unexpected ally in the search for new treatments: the black soldier fly, an insect better known for recycling food waste. The researchers asked whether small natural molecules made by these insects could safely kill dangerous bacteria that infect humans.
Hunting germs with nature’s small weapons
All animals, including insects, rely on built-in chemical defenses to survive in a world full of microbes. Among the most important of these defenses are short chains of amino acids called antimicrobial peptides. They often work by punching holes in the outer shell of bacteria, killing them quickly. The team behind this study focused on such peptides from the black soldier fly, which thrives in microbe-rich environments and is thought to produce a particularly rich arsenal of germ-fighting molecules.
Picking the most promising candidates
Using computer analysis of previously mapped genetic information from the black soldier fly, the researchers narrowed a list of dozens of candidate peptides down to ten with the most promising predicted properties. These ten were then made in the lab and tested against a panel of microbes that frequently cause human disease, including two common Gram-positive bacteria (such as Staphylococcus aureus), two Gram-negative bacteria (such as Escherichia coli and Pseudomonas aeruginosa), and two types of disease-causing fungi. At the same time, the team checked whether these peptides damaged human cells or red blood cells, which would signal potential side effects.
One standout peptide with fast-killing power
Most of the peptides showed little or only selective activity, but one, named Hill_BB_C7176, clearly stood out. It was able to stop the growth of all four tested bacterial species, including hard-to-treat Gram-negative organisms, at low micromolar concentrations. Detailed time-course experiments revealed that once enough of this peptide was present, it did not just slow the bacteria down; it wiped them out within one to two hours, with no regrowth during the test period. At the same time, Hill_BB_C7176 showed no detectable toxicity toward human lung cells and only very weak effects on red blood cells at the highest tested doses, suggesting a favorable early safety profile.
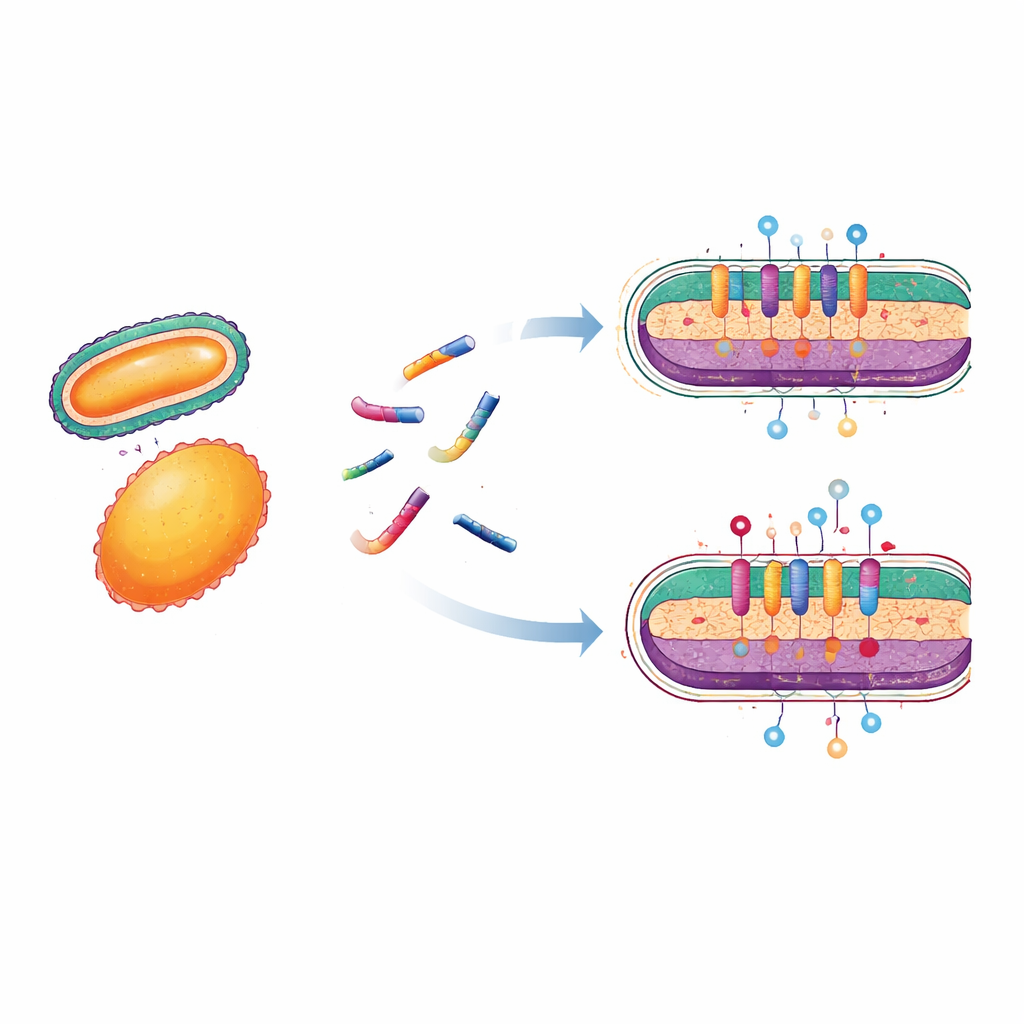
How the insect peptide attacks bacteria
To understand how the standout peptide works, the scientists zoomed in on bacterial envelopes, the layered structures that act as a cell’s outer armor. They used a fluorescent dye that can only enter bacteria if their inner barrier becomes leaky. When bacteria were exposed to Hill_BB_C7176, the dye quickly flooded in, especially in Gram-positive cells, showing that the peptide rapidly punched holes in the membrane. A second test tracked how strongly the peptide latched onto a component of the outer coat of Gram-negative bacteria known as lipopolysaccharide, or LPS. Hill_BB_C7176 displaced a fluorescent probe from LPS in a concentration-dependent way, indicating strong binding to this key surface structure and suggesting it may use LPS as an entry point to disrupt the membrane.
Testing in a living infection model
The team then moved beyond petri dishes and tested the peptide in a simple animal model using larvae of the wax moth Galleria mellonella, which are commonly used to mimic infection in a living host. Larvae were infected with either S. aureus or E. coli and then treated with Hill_BB_C7176, standard antibiotics, or a salt solution as a control. Across several days of monitoring, larvae that received the insect peptide survived far better than untreated infected animals. For E. coli, the survival pattern with the peptide closely matched that of a last-resort antibiotic used as a benchmark, while for S. aureus the peptide was somewhat less protective but still markedly better than no treatment.
What this means for future medicines
This work shows that a single peptide from the black soldier fly can kill a broad range of dangerous bacteria quickly, appears gentle toward human cells in laboratory tests, and improves survival in a living infection model. The evidence points to a mode of action where the peptide clings to bacterial surface molecules and then breaks open their protective membranes. While much more work is needed—including tests in mammals, checks on how long the peptide survives in the body, and chemical refinements—Hill_BB_C7176 emerges as a promising starting point for new treatments to tackle antibiotic-resistant infections.
Citation: Derin, E., Van Moll, L., Wouters, M. et al. Antimicrobial effects of novel Hermetia illucens peptides. Sci Rep 16, 10398 (2026). https://doi.org/10.1038/s41598-026-40997-3
Keywords: antimicrobial peptides, black soldier fly, antibiotic resistance, bacterial infections, insect-derived drugs