Clear Sky Science · en
Laser fluence dependent antibacterial activity of cadmium sulfide quantum dots prepared by one step laser ablation in liquid
Why tiny light-made particles matter
Antibiotic-resistant infections are becoming harder and harder to treat, turning once-routine illnesses into serious threats. This study explores a different kind of weapon: ultra-small particles called quantum dots, made from cadmium sulfide, that can be created with a flash of laser light in water. The work shows how adjusting the strength of the laser changes these particles and how effectively they can kill dangerous bacteria, hinting at a new tool in the fight against drug-resistant germs.
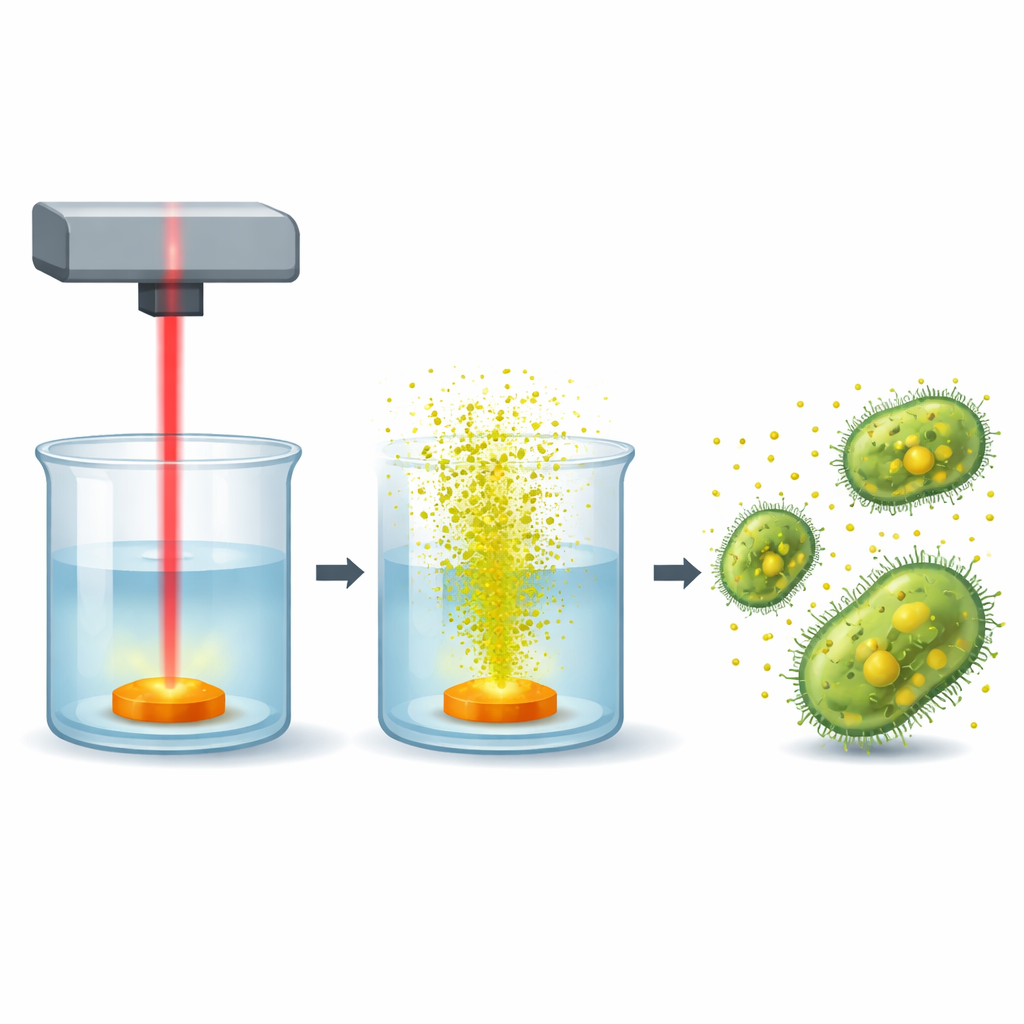
Making tiny fighters with a laser
The researchers produced cadmium sulfide quantum dots by pointing a pulsed laser at a small amount of cadmium sulfide powder submerged in pure water. Each laser pulse blasts a bit of the solid into the liquid, where it cools and forms nanoparticles only a few billionths of a meter across. By changing the laser fluence—the energy delivered per unit area—they could tune the size, structure, and surface charge of the resulting dots. Three laser settings were tested, ranging from relatively gentle to quite intense pulses, all in a surfactant-free, “clean” water environment.
Looking closely at the new particles
To understand what they had made, the team used a suite of standard materials tests. X-ray diffraction confirmed that the particles formed a well-defined crystalline structure known as hexagonal wurtzite, with crystal domains only about 6 to 11 nanometers across. Electron microscopes revealed nearly spherical particles in the 2 to 3 nanometer range, small enough for quantum effects to dominate their behavior. Light-absorption and glow (photoluminescence) measurements showed that the dots absorbed ultraviolet light and emitted blue-shifted light compared with ordinary cadmium sulfide, a fingerprint of quantum confinement that comes with shrinking a material to the nanoscale.
How laser strength shapes behavior
Changing the laser fluence had a clear impact on the particles. Higher-energy pulses tended to produce more concentrated and somewhat larger crystallites, but also more intense light emission, suggesting better crystal quality and more active sites on the surface. Measurements of zeta potential, which reflects surface charge and stability in liquid, showed that particles made with stronger laser pulses carried a larger negative charge and formed more stable suspensions, resisting clumping. Infrared spectroscopy confirmed the expected cadmium–sulfur bonds and showed water-related groups on the surface that help keep the tiny dots dispersed. Together, these tests demonstrated that laser settings can be used as a “knob” to tune both structure and stability without relying on extra chemicals.

Putting the particles to the test against germs
The crucial question was whether these laser-made quantum dots could actually harm bacteria. The team tested them against four clinically relevant strains: two Gram-negative (Escherichia coli and Pseudomonas aeruginosa) and two Gram-positive (Staphylococcus aureus and Streptococcus agalactiae). They placed different concentrations of the dots into wells on agar plates seeded with bacteria and measured the clear “kill zones” that formed. They also used a color-change test in microplates to determine the minimum inhibitory concentration, the lowest dose that stops visible growth. In both types of tests, particles made with the highest laser fluence showed the strongest antibacterial action, needing far lower doses to suppress growth.
How these tiny dots likely kill bacteria
Although the study did not track every step inside the cell, earlier work and these results point to a combined attack. The nanometer-sized dots can approach and attach to bacterial cell walls, where their surface charge and size help them cross the protective barrier. Once close to or inside the cell, they can release cadmium ions and generate reactive oxygen species—highly reactive forms of oxygen that damage fats, proteins, and DNA. This multi-pronged stress can disrupt membranes, block enzymes, and ultimately cause the cell to die. The stronger antibacterial effects seen for the better-dispersed, higher-fluence dots fit with this picture: more stable, well-exposed surfaces mean more contact with bacteria and more chemical damage.
What this could mean for future treatments
For a layperson, the key outcome is that a simple, relatively green laser-in-water method can create ultra-small particles that strongly inhibit several hard-to-treat bacteria, including drug-resistant strains. By dialing up or down the laser energy, scientists can steer the size, stability, and killing power of these particles without adding extra surfactants or complex reagents. While cadmium-based materials raise important safety and environmental questions that must be answered before clinical use, this work shows a promising way to design next-generation antibacterial agents. The same approach could also feed into water disinfection, smart coatings, or targeted drug delivery systems that use light-responsive nanomaterials to help keep infections under control.
Citation: Hassan, K.M., Taha, A.A., Ismail, R.A. et al. Laser fluence dependent antibacterial activity of cadmium sulfide quantum dots prepared by one step laser ablation in liquid. Sci Rep 16, 10684 (2026). https://doi.org/10.1038/s41598-026-40885-w
Keywords: antibiotic resistance, quantum dots, nanoparticles, laser ablation in liquids, antibacterial nanomaterials