Clear Sky Science · en
First comparison of commercial systems to prepare nanofat: technical performances and biological quality differ among obtained products
Why this matters for everyday medicine
Doctors increasingly use a patient’s own fat, not just to restore volume in the face or body, but to help damaged skin and soft tissue heal. A special form called “nanofat” is injected in very small amounts to smooth scars, improve skin texture, and support tissue repair. Yet clinics can buy many different kits to prepare nanofat, and no one really knew whether they all produce the same kind of regenerative material. This study is the first head‑to‑head comparison of popular commercial systems, asking a simple but important question: do different devices make different nanofats?
From regular fat to healing micro‑grafts
Nanofat starts as ordinary fat removed during liposuction, usually from the abdomen, hips, or thighs. Instead of being injected as chunky “fillers,” the fat is vigorously pushed back and forth through narrow connectors or sharp grids, sometimes followed by filtration. This mechanical processing breaks it into a fine, injectable suspension that can pass through very thin needles into the upper layers of the skin. The idea is less about adding bulk and more about delivering living support cells and tissue fragments that release growth factors to spur blood vessel growth, calm inflammation, and remodel scars.
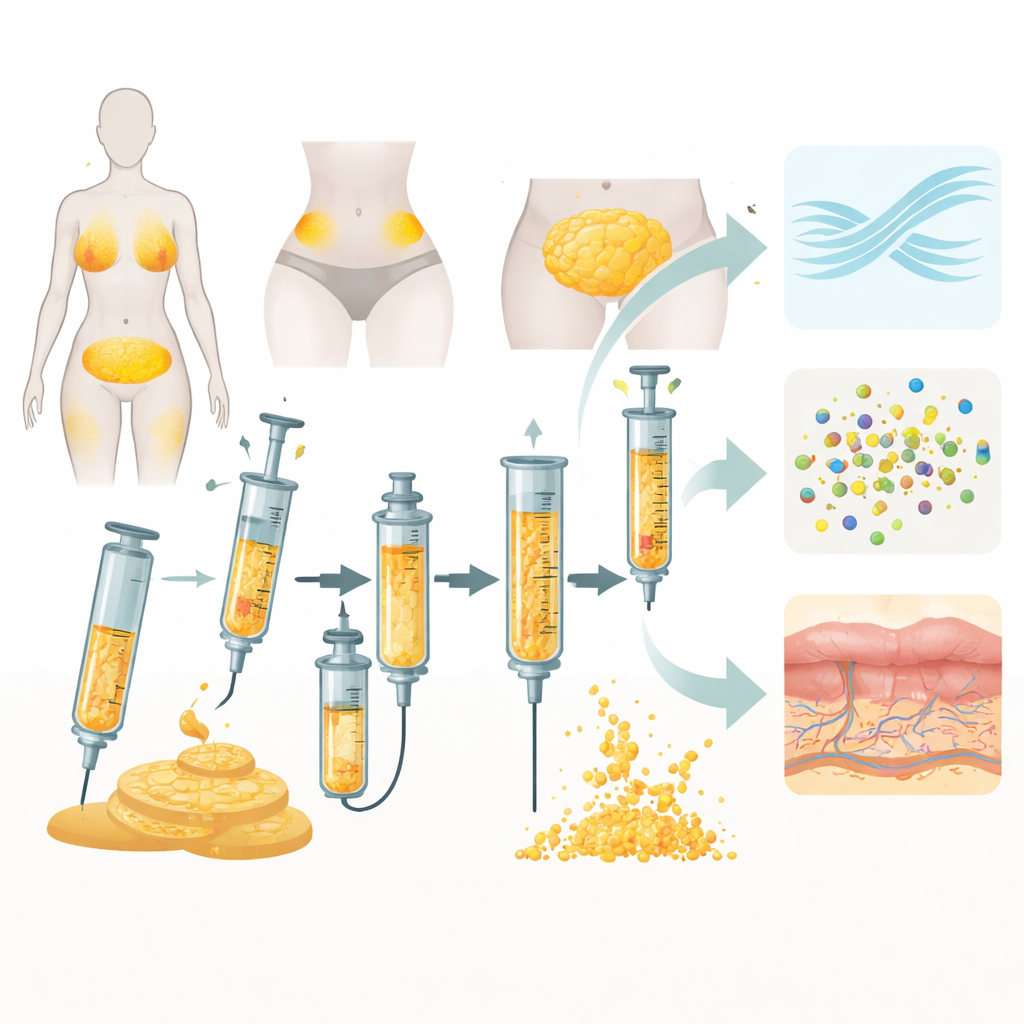
Putting eight devices to the test
In this study, fat from five healthy women undergoing cosmetic liposuction was processed with eight widely available devices that use either “emulsification” (mainly tearing tissue by flow) or “micronization” (forcing it through sharp grids). The team looked at practical aspects that matter in the operating room, such as preparation time, ease of use, how smoothly the product could be injected, how much usable nanofat was recovered, and how watery it was. They also performed detailed laboratory tests: how many cells survived, how many viable cells were obtained per milliliter, what types of cells were present, whether stem‑like cells could still form colonies, and what kinds of tiny signaling particles called extracellular vesicles were released.
Technical convenience versus biological richness
All devices worked reasonably well on the technical side, but some clearly stood out. The Emulsfat system and the newer Puregraft Boost V2 were the quickest to use, produced concentrated fat with little leftover fluid, and were rated easiest to handle and inject. However, the device that scored best biologically was Adinizer, which yielded the highest proportion of regenerative stromal or stem‑like cells while keeping cell survival above 85%—a level reached by all systems. In contrast, Hy‑Tissue Nanofat produced fewer cells overall and a higher share of white blood cells, which may not always be desirable. These findings show that a kit that feels great in the surgeon’s hand does not automatically produce the richest regenerative product; choosing a system may require balancing technical comfort against the desired biological profile.
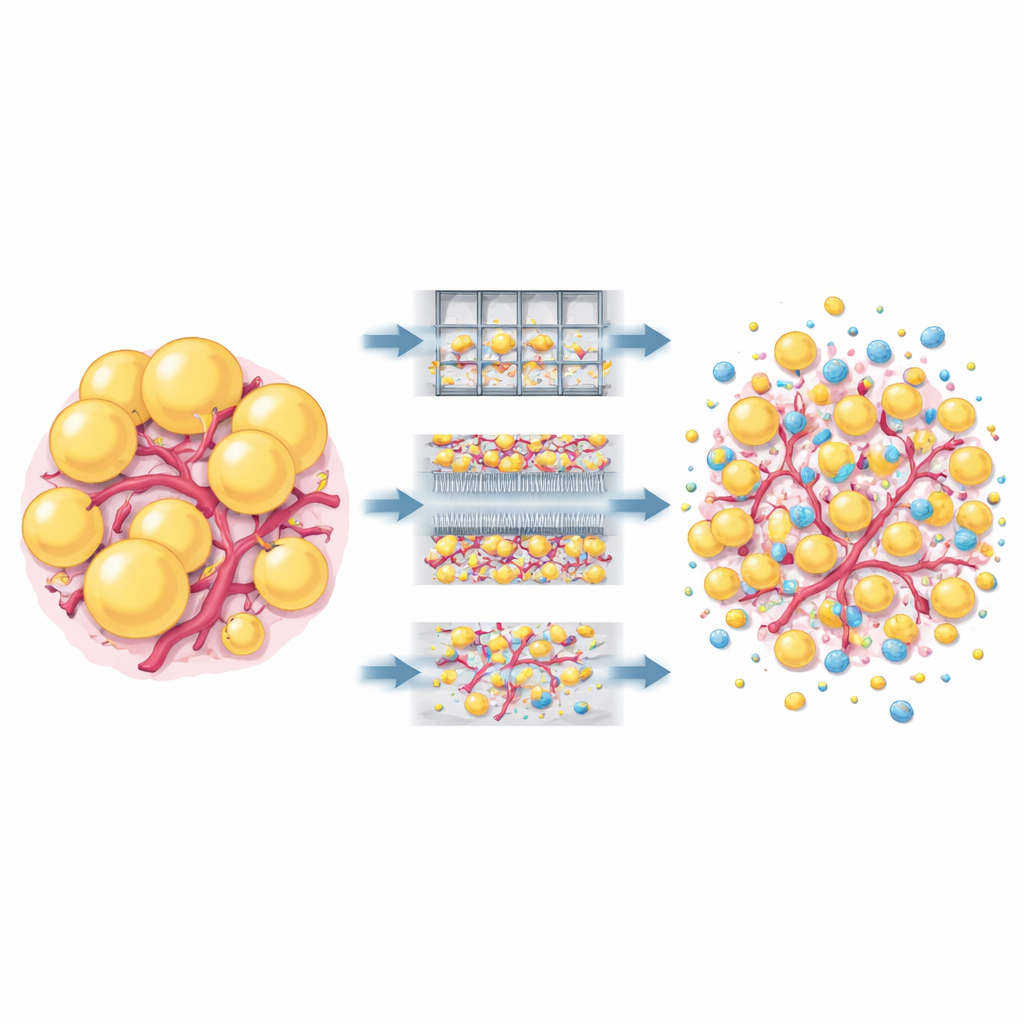
Hidden structure and tiny messengers
Microscope images offered a surprising insight: despite the vigorous mechanical treatment, nanofat from most devices still contained intact fat cells, small blood vessels, and a preserved web of supporting matrix proteins. This challenges the common belief that nanofat is simply a cell‑rich liquid with all structure destroyed. The researchers also collected the fluid in which nanofat was briefly cultured and counted extracellular vesicles—nanoscale bubbles that carry proteins and genetic signals. Overall vesicle numbers were similar across devices and varied more from donor to donor than from method to method, although Emulsfat samples were somewhat richer in vesicles originating from fat cells.
What this means for future treatments
For patients, the take‑home message is that “nanofat” is not a single uniform product; what ends up in the syringe depends strongly on how it was made. Some systems favor speed, smooth injection, and high volume, while others better preserve regenerative cells. The study’s combined scoring of technical and biological features gives clinicians a roadmap to match devices to specific goals, such as maximizing stem‑like cells for demanding regenerative procedures or prioritizing easy delivery for superficial skin treatments. More broadly, the work underlines the need for clear standards so that when doctors and researchers talk about nanofat, they are truly referring to comparable therapies, helping to make fat‑based regenerative medicine safer, more effective, and more predictable.
Citation: Arcani, R., Abellan, M., Simoncini, S. et al. First comparison of commercial systems to prepare nanofat: technical performances and biological quality differ among obtained products. Sci Rep 16, 9998 (2026). https://doi.org/10.1038/s41598-026-40847-2
Keywords: nanofat, fat grafting, adipose stem cells, regenerative medicine, extracellular vesicles