Clear Sky Science · en
Synergistic dye removal through curcumin functionalized MCM-22 zeolite as a photocatalyst nanocomposite via the simultaneous photocatalysis and adsorption method
Turning Bright Dyes into Clean Water
Colorful synthetic dyes make our clothes vivid and our products eye-catching, but once they wash down the drain they can linger in rivers and lakes for years. This study explores a new material that tackles these stubborn pollutants using both light and smart chemistry. By combining a common white pigment (titanium dioxide), a porous mineral (zeolite), a plant compound from turmeric (curcumin), and the rare-earth element cerium, the researchers created a tiny "water-cleaning sponge" that can strip blue dye from water with remarkable efficiency under visible light.
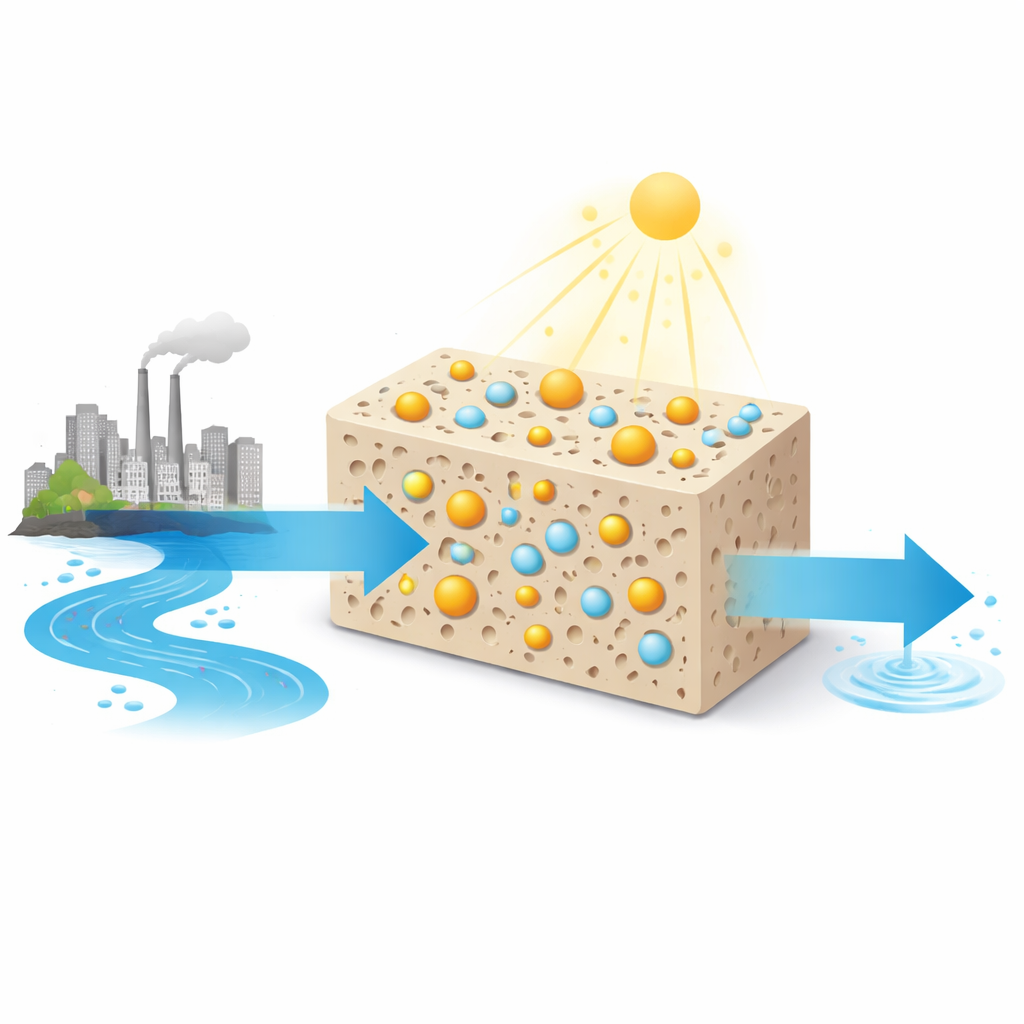
A New Way to Clean Colored Wastewater
Many factories that make textiles, inks, and plastics generate wastewater laced with stable dyes such as methylene blue. Traditional treatment methods—like filtering, adding chemicals, or relying on microbes—often struggle with such molecules or become expensive and hard to maintain. The team set out to build a material that does double duty: it first captures dye molecules on its surface (adsorption) and then breaks them apart using light-driven chemistry (photocatalysis). Their goal was a system that works with ordinary visible light, not just the ultraviolet portion of sunlight that typical titanium dioxide relies on.
Building a Tiny Layered Cleaner
At the heart of the design is MCM-22, a type of zeolite—a crystalline, sponge-like mineral full of nanoscale pores. The researchers first synthesized this porous framework, then extracted curcumin from turmeric using ethanol. They chemically attached the curcumin molecules to the zeolite surface, forming a thin organic layer that can bind metal ions. Next, they introduced titanium butoxide and a cerium salt so that, inside and on the plates of the zeolite, extremely fine titanium dioxide and cerium nanoparticles formed and spread out evenly. Microscopy and a suite of spectroscopic tests confirmed that the particles were well-dispersed, strongly attached, and that the organic curcumin layer was present and interacting with the metals.
How Light and Structure Work Together
The engineered material is designed so each component plays a distinct role. The zeolite provides a stable scaffold and large pores where dye molecules can reach active sites. Curcumin helps draw dye molecules to the surface and also acts like a light antenna, absorbing visible light and passing energy or electrons to titanium dioxide. Cerium particles help extend light absorption into the visible range and act as temporary "parking spots" for electrons, which slows down their unwanted recombination with positive charges. Measurements showed that, compared with plain titanium dioxide, the composite absorbs more visible light and has a smaller effective band gap, meaning it can be activated by a larger slice of the solar spectrum. At the same time, its light emission signal drops, a sign that charge carriers remain separated longer—exactly what is needed for efficient pollutant breakdown.
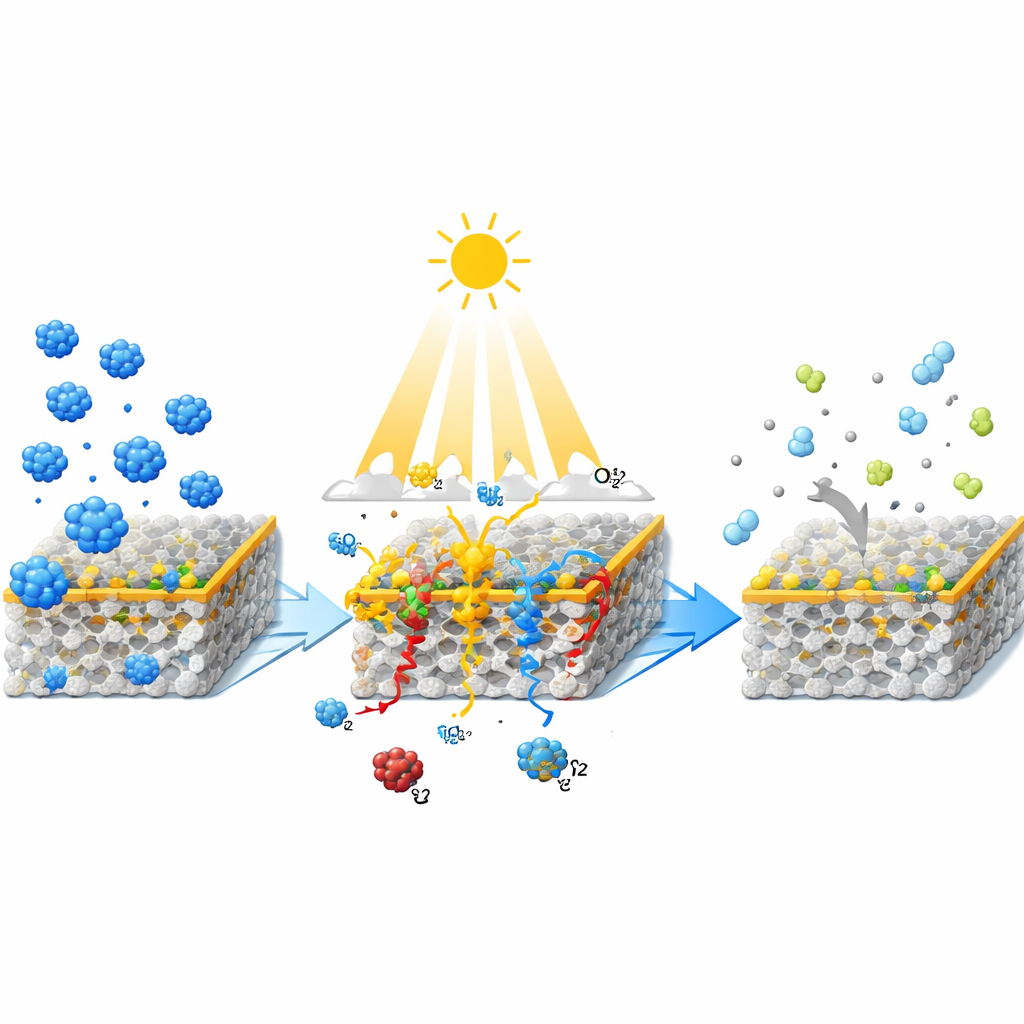
From Polluted Dye to Near-Clear Water
To test performance, the researchers placed the composite in a stirred reactor containing a dilute methylene blue solution and illuminated it with visible light from xenon lamps. They varied key conditions such as the amount of cerium, pH of the water, dye concentration, and how much photocatalyst was present. Under optimized conditions—moderate cerium loading around 9 percent by weight, alkaline pH, and sufficient catalyst amount—the combined adsorption and photocatalysis process removed about 96 percent of the dye in two hours. By contrast, using the same material only as a photocatalyst or only as an adsorbent achieved just 11 percent and 32 percent removal, respectively. Analysis of the reaction rates suggested that the process behaves like a first-order reaction, where the speed depends on how much dye remains in solution.
Lasting Power and Real-World Promise
For any treatment material to be practical, it must endure multiple cycles without losing its punch. The team ran the same batch of composite through five rounds of dye degradation, carefully washing and reusing it each time. After these cycles, its activity dropped only slightly—by about 8 percentage points—indicating solid stability. Altogether, the study shows that cleverly combining a natural molecule from turmeric with engineered minerals and metals can create a potent, reusable cleaner for colored wastewater. To a non-specialist, the take-home message is simple: by designing smart, light-activated materials at the nanoscale, it may be possible to turn some of our most persistent water pollutants into harmless fragments using nothing more than common ingredients and visible light.
Citation: Shadi, E., Amirinejad, M., Derakhshan, A.A. et al. Synergistic dye removal through curcumin functionalized MCM-22 zeolite as a photocatalyst nanocomposite via the simultaneous photocatalysis and adsorption method. Sci Rep 16, 10226 (2026). https://doi.org/10.1038/s41598-026-40712-2
Keywords: wastewater treatment, photocatalytic dye removal, titanium dioxide nanocomposite, zeolite adsorbent, curcumin functionalization