Clear Sky Science · en
Characterization of a novel assay for full-length endotrophin in heart failure with preserved ejection fraction
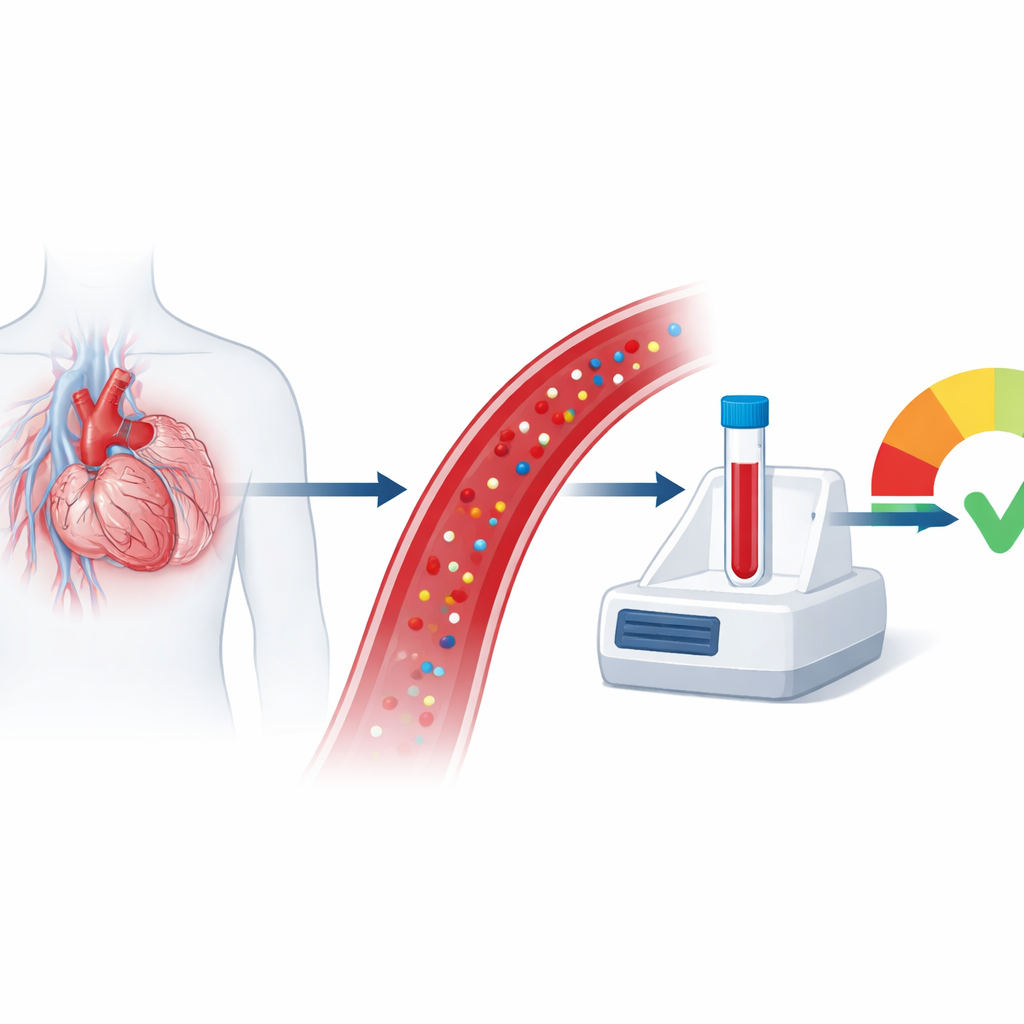
Why a hidden heart signal matters
Heart failure is often thought of as a weak heart that cannot pump enough blood, but for many people the heart’s squeeze is still normal. Instead, their heart becomes stiff and crowded with extra supporting tissue, making it hard to fill properly. This form, called heart failure with preserved ejection fraction, or HFpEF, is common in older adults and people with high blood pressure or obesity, yet it remains hard to diagnose and even harder to predict who will worsen. The study described here introduces a new blood test that reads a subtle signal from the heart’s scaffolding, offering a fresh way to judge risk and possibly guide future treatments.
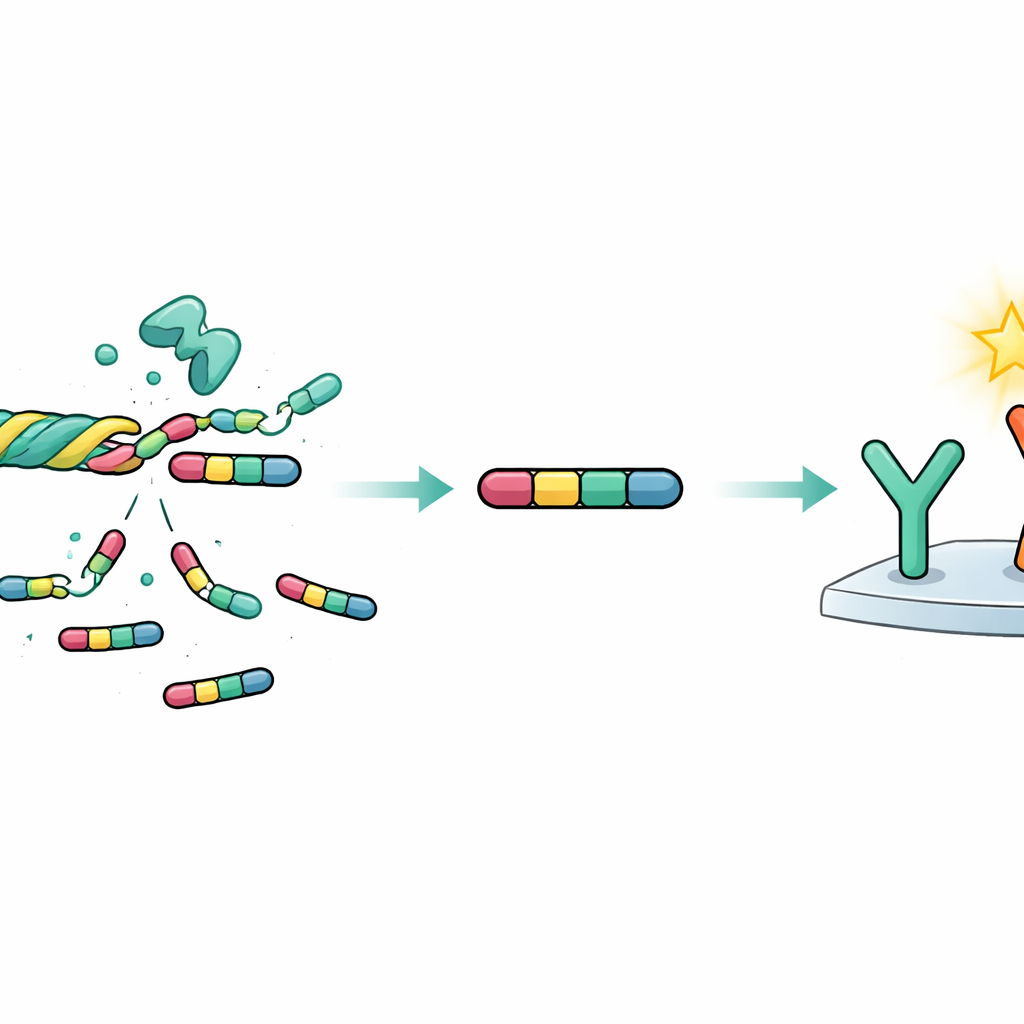
The heart’s supporting mesh
Our hearts are not just made of beating muscle cells. They are also wrapped in a mesh of proteins known as the extracellular matrix, which acts like scaffolding that holds everything in place and helps repair damage. In HFpEF, this mesh thickens and stiffens in a process called fibrosis, which can gradually choke the heart’s ability to relax between beats. A key ingredient in this mesh is collagen, a family of rope-like proteins. One member, type VI collagen, has a special tail on one of its chains; when this tail is cut loose, it forms a small fragment called endotrophin. This fragment is not just debris—it behaves like a messenger that can promote scarring, inflammation, and metabolic problems, making it a promising window into disease activity.
Turning a tiny fragment into a blood test
Earlier research used a test called PRO-C6 to measure pieces of the same collagen tail in the bloodstream. However, that assay detected a mixture of fragment sizes and did not distinguish the complete endotrophin piece from shorter breakdown products. In the new work, the researchers designed a more precise sandwich-style blood test that locks onto both ends of the full-length endotrophin molecule, ensuring that only the intact 77–amino acid fragment is measured. They developed and refined antibodies that recognize each end of endotrophin, adapted the test to run on an automated laboratory platform, and then rigorously checked its technical performance. The assay proved stable, accurate, repeatable, and resistant to common sources of interference such as fats in the blood, colored pigments from red cells, or extra biotin from supplements.
What the new test reveals in patients
To find out whether this more specific endotrophin signal matters for real patients, the team measured it in two groups of people with HFpEF. One group had recently been hospitalized and still showed signs of fluid overload and poorer kidney function, while the other group had long-standing high blood pressure and HFpEF but was relatively stable and seen in clinic. In both groups, higher endotrophin levels in the blood tracked closely with an older assay (PRO-C6) and were moderately linked to a standard heart failure marker called NT-proBNP, suggesting that they reflect related but not identical processes. People in the sicker, recently hospitalized group had higher endotrophin levels overall, tying the marker to a heavier burden of disease.
Linking the signal to survival
The crucial question was whether endotrophin could help identify who was most likely to die from any cause or from heart-related reasons over several years of follow-up. When the researchers divided patients into low, medium, and high endotrophin groups, those with the highest levels consistently had worse survival. In the unstable, post-hospital group, endotrophin remained strongly associated with both all-cause and cardiovascular death even after accounting for age, sex, body weight, kidney function, and NT-proBNP. In the more stable clinic group, endotrophin predicted risk on its own, but its added value shrank once NT-proBNP was included in the models. Overall, statistical tests showed that models including endotrophin often fit the data better than models based on other single markers, especially in the more fragile patients.
What this means for patients and care
For people living with HFpEF and their clinicians, this study suggests that the heart’s scaffolding can tell an important story that traditional tests may miss. The new assay offers a way to measure the full-length endotrophin messenger circulating in the blood, capturing a signal of ongoing scarring and tissue remodeling. In high-risk, recently hospitalized patients, this signal seems to carry independent information about the chance of dying, beyond what is provided by long‑used markers like NT‑proBNP. While the test is not yet ready for everyday clinic use and needs validation in larger and more diverse groups, it opens the door to more personalized risk assessment and to future therapies that might aim directly at the fibrotic processes driving HFpEF.
Citation: Angeli, E., Revuelta-López, E., López, B. et al. Characterization of a novel assay for full-length endotrophin in heart failure with preserved ejection fraction. Sci Rep 16, 13959 (2026). https://doi.org/10.1038/s41598-026-40557-9
Keywords: heart failure with preserved ejection fraction, endotrophin, collagen, cardiac fibrosis, biomarkers