Clear Sky Science · en
Synthesis, characterization, antioxidant and anticancer potential of Kalanchoe pinnata green synthesized silver nanoparticles targeting p53/MDM2 nexus in hepatic cancer: integrated in vitro and in silico study
From Garden Plant to Cancer-Fighting Tool
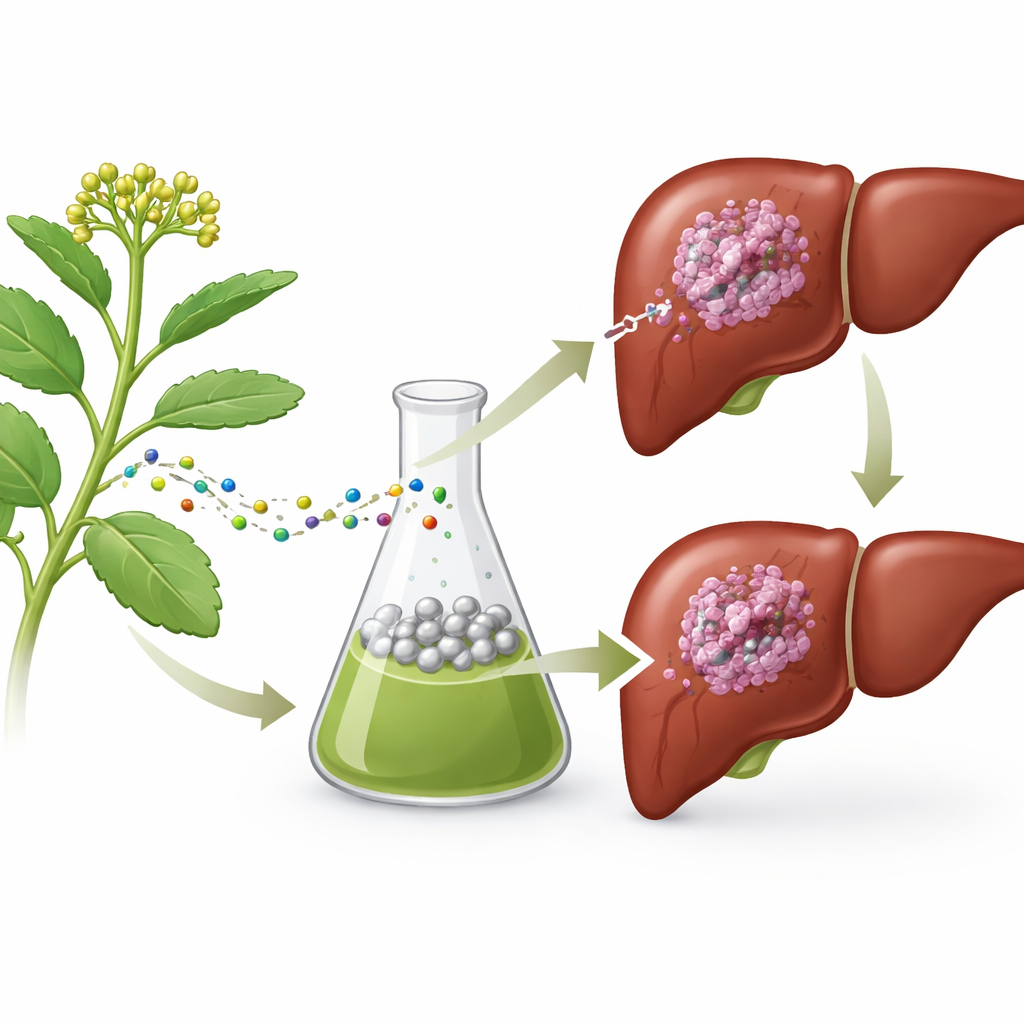
Many of today’s medicines have their roots in ordinary plants. This study explores whether a common succulent, Kalanchoe pinnata, can help create tiny particles of silver that act as both strong antioxidants and potential cancer-fighting agents against liver cancer, one of the deadliest cancers worldwide. By using a "green" manufacturing method that avoids harsh chemicals, the researchers aim to harness nature’s chemistry to build safer, more targeted treatments for tumors while also protecting healthy cells.
Why Liver Cancer Needs New Options
Liver cancer, particularly hepatocellular carcinoma, is hard to treat and often diagnosed late. Standard treatments like chemotherapy, surgery, and liver transplants can extend life, but they come with serious side effects and do not work for everyone. Many tumors also carry defects in key safety switches inside cells, such as the p53 protein and its regulator MDM2, which help decide whether damaged cells should repair themselves or die. When this control system goes wrong, cancer cells can survive when they should not. Finding therapies that can gently push these cells back toward self-destruction, without harming normal tissue, is a major goal in modern oncology.
Turning a Healing Plant into Tiny Silver Helpers
The researchers began by preparing an alcohol-based extract from the leaves of Kalanchoe pinnata, a plant long used in traditional medicine. Using chemical fingerprinting (GC–MS), they found 33 different natural substances in the extract, including well-known molecules like squalene and β-amyrin that have antioxidant and anti-inflammatory properties. They then mixed this plant extract with a silver salt solution. The plant compounds acted as natural reducing agents, turning silver ions into metallic silver nanoparticles and coating their surfaces, all in a single, gentle step. Measurements showed that the resulting particles were very small, roughly tens of nanometers across, mostly spherical, and carried an electrical charge that helps keep them stable and well-dispersed in liquid.
How the Nanoparticles Protect and Attack
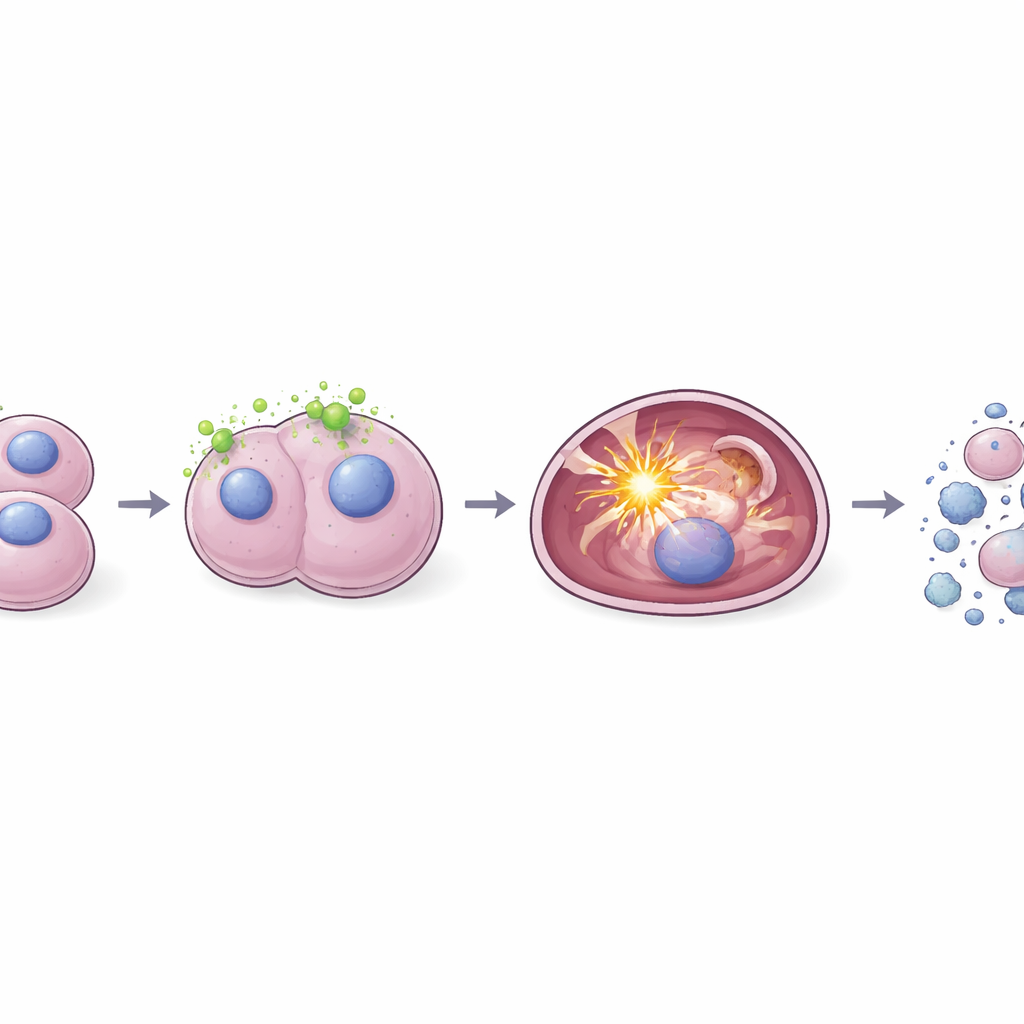
Next, the team tested whether these Kalanchoe-based silver nanoparticles could neutralize harmful free radicals—highly reactive molecules that damage DNA, fats, and proteins. Using two standard lab tests (DPPH and ABTS), they found that the particles mopped up these radicals efficiently, and their activity increased with higher doses. The same particles were then applied to human liver cancer cells grown in dishes. Here, they had a very different role: instead of protecting, they pushed the cancer cells toward death. The nanoparticles reduced cancer cell survival more strongly than the plant extract alone, caused obvious changes in cell shape, and led to shrunken, fragmented cell nuclei, all hallmarks of programmed cell death rather than simple poisoning.
Triggering Internal Stress and Clean Cell Self-Destruction
To understand what was happening inside the cancer cells, the researchers looked at reactive oxygen species, a particular form of internal chemical stress. They observed that as nanoparticle doses increased, so did the levels of these reactive molecules within liver cancer cells. Fluorescent dyes and imaging showed a bright, dose-dependent signal, indicating that the cells were under intense oxidative pressure. Additional staining revealed that more and more cells were entering early and late stages of apoptosis, the tidy self-destruct program that breaks cells into small, cleanly removable pieces. Importantly, the nanoparticles showed little harm to normal immune cells in culture at similar doses, hinting at some selectivity for tumor cells.
Peeking into Molecular Targets with Computer Models
The study also used computer-based docking simulations to ask whether β-amyrin, one of the plant’s major compounds, could latch onto crucial proteins that control cell fate. The models suggested that β-amyrin can bind strongly to p53, its regulator MDM2, and the executioner enzyme caspase-3, with binding energies comparable to or approaching those of standard anticancer drugs. These interactions are consistent with a scenario in which β-amyrin helps free p53 from suppression, supports activation of caspase-3, and thereby enhances the cell’s own machinery for recognizing damage and committing to apoptosis.
What This Could Mean for Future Treatments
Overall, this work shows that a familiar ornamental plant can be used to fabricate tiny, stable silver particles that both neutralize damaging free radicals and selectively push liver cancer cells toward controlled self-destruction, possibly by nudging the p53/MDM2 system and related death pathways. While these findings are still limited to lab dishes and computer models—and much more testing in animals and humans would be needed—they point toward a future where gentle, plant-based ingredients and precision nanotechnology could be combined to create more natural, targeted treatments for liver cancer and other diseases driven by oxidative stress.
Citation: Moin, A., Hussain, T., Alafnan, A. et al. Synthesis, characterization, antioxidant and anticancer potential of Kalanchoe pinnata green synthesized silver nanoparticles targeting p53/MDM2 nexus in hepatic cancer: integrated in vitro and in silico study. Sci Rep 16, 11943 (2026). https://doi.org/10.1038/s41598-026-40542-2
Keywords: green nanomedicine, Kalanchoe pinnata, silver nanoparticles, liver cancer, p53 MDM2