Clear Sky Science · en
Neonatal brain age estimation from T2-weighted magnetic resonance images using shallow regression analysis
Why timing matters for tiny brains
In the first weeks of life, a baby’s brain grows at an astonishing pace, reshaping itself day by day. Doctors caring for preterm and full-term infants would like a precise way to know whether this growth is on track, long before delays show up in behavior. This study shows how detailed MRI scans of newborns’ brains can be turned into an accurate “brain age” estimate, giving clinicians a sensitive early window into brain health.
Measuring age beyond the calendar
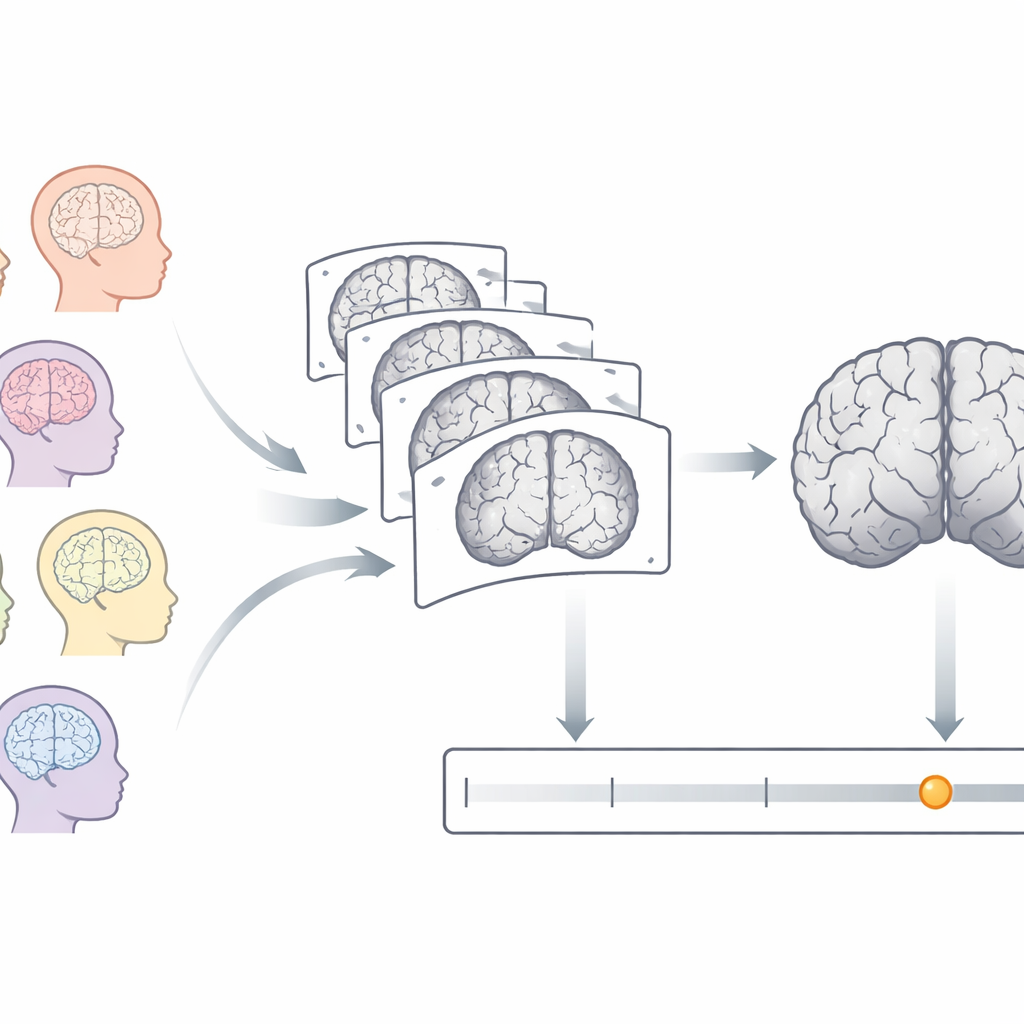
Newborns are usually described by how many weeks have passed since conception, known as postmenstrual age. Yet two babies of the same calendar age can have quite different brain maturity, especially if one was born early. Magnetic resonance imaging (MRI) can capture the brain’s structure without radiation, revealing how folds deepen and tissues change as babies grow. The researchers set out to turn these rich images into a single number: how old the brain looks, in weeks, compared with typical development. Differences between this brain age and the baby’s actual age could then flag unusually slow or fast maturation.
Scanning hundreds of newborns
The team used MRI data from 885 newborns in the Developing Human Connectome Project, including both preterm and full-term infants between 26 and 46 weeks postmenstrual age at the time of scanning. All babies were scanned during natural sleep in a specialized neonatal MRI system, producing high-quality T2-weighted images that highlight differences between brain tissues. Before any analysis, non-brain tissues such as the skull were removed, image brightness was corrected, and each brain was resampled into a standard size and resolution so that brains from different infants could be meaningfully compared.
Aligning brains and picking the right reference
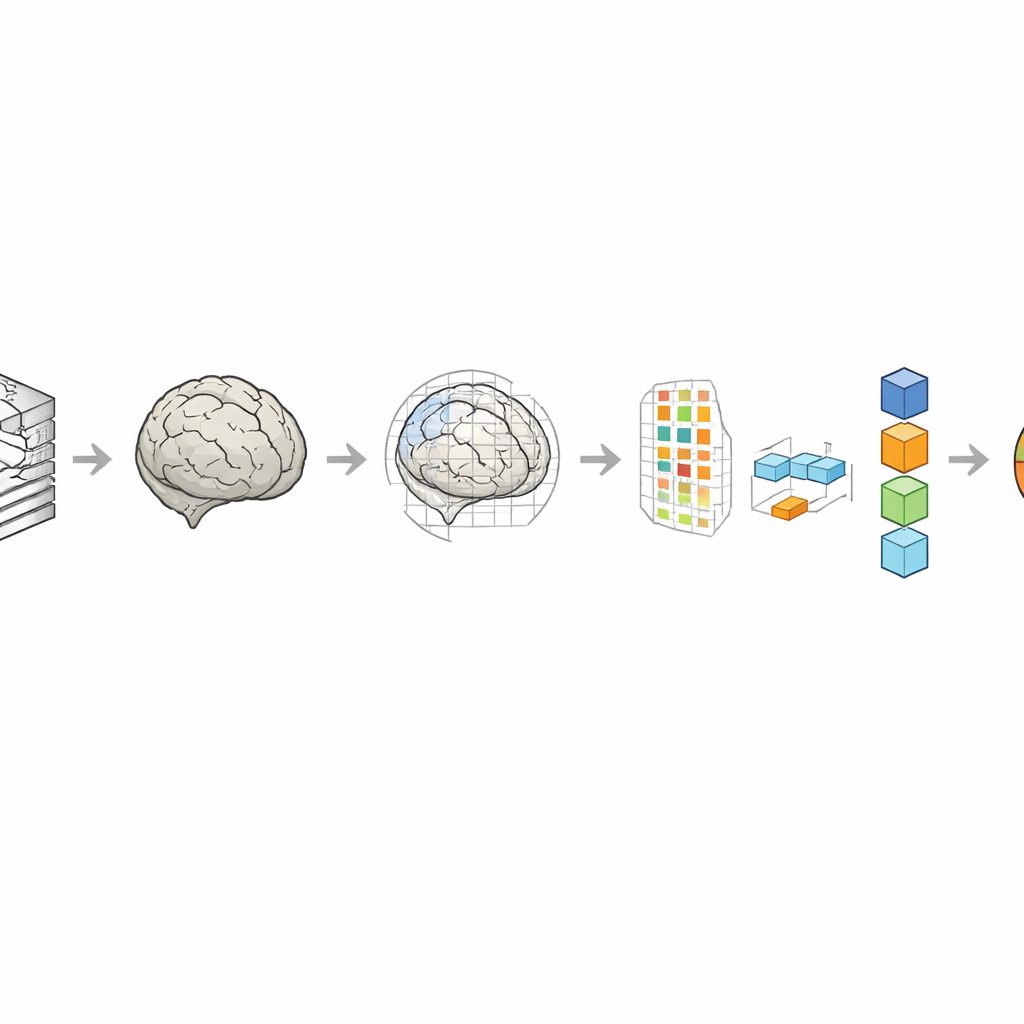
A central step in the work was deciding how to align each baby’s brain with a reference brain, called a template. The authors compared three choices: a template closely matched to each baby’s age, a single template built from full-term 40-week-old brains, and a randomly selected neonatal template. They also tested two ways of fitting each brain to these templates: a simpler linear adjustment that scales and rotates the brain, and a more flexible combination of linear and nonlinear warping that can fine-tune local shapes. These decisions matter because better alignment lets the computer focus on real developmental differences rather than on irrelevant shifts and distortions.
Turning images into numbers the computer can learn from
Each aligned brain scan contains millions of tiny volume elements, or voxels, far too many to feed directly into a prediction model. To handle this, the team used a mathematical technique that compresses the data into a smaller set of components capturing the main patterns of variation across babies. They then ranked these components by how strongly they tracked age and kept only the most informative ones. With these features in hand, they trained three established regression methods—Gaussian process regression, support vector regression, and relevance vector regression—to predict brain age, checking performance with repeated cross-validation so that each baby served as an unseen test at some point.
How close can brain age come to the truth?
When the images were aligned using both linear and nonlinear warping to an age-specific template, brain age estimates were remarkably precise. The best models missed the true age by about 0.44 weeks on average—less than half a week—with around 95% of the variation in age explained by the imaging data. Using a full-term template gave slightly larger errors, and using a random template performed worst, especially for preterm babies. The more flexible registration consistently beat the simpler linear approach, though the gains were modest enough that the simpler method may still be attractive when computing power is limited. Among the three prediction methods, Gaussian process regression offered the best balance of accuracy and stability.
What this means for fragile beginnings
By carefully choosing how newborn brain scans are aligned and summarized, this work shows that a single MRI can provide a very accurate estimate of how mature a baby’s brain is, down to about the span of a few days. Such a “brain age” measure could help clinicians spot subtle lags in development for preterm or high-risk infants well before problems are visible in behavior or movement. While the study relies on a single type of MRI and data from a specific research project, it lays the groundwork for practical tools that could track early brain growth more objectively, supporting earlier intervention during a period when the brain is uniquely sensitive to help.
Citation: Taji, S.M., Kazemi, K., Qodrati, Z. et al. Neonatal brain age estimation from T2-weighted magnetic resonance images using shallow regression analysis. Sci Rep 16, 9980 (2026). https://doi.org/10.1038/s41598-026-40336-6
Keywords: neonatal brain development, brain age estimation, neonatal MRI, preterm infants, machine learning