Clear Sky Science · en
Waste citrus pseudolimon peels derived biochar assisted magnetic Zn + Al (LDH) nanocomposites for As (III) adsorption
Turning Fruit Waste into a Water Clean‑Up Tool
Many regions around the world struggle with drinking water contaminated by arsenic, a poisonous element that can cause cancer and other serious illnesses. At the same time, tons of fruit peels from the food industry are thrown away every day. This study brings these two problems together and shows how discarded citrus peels can be transformed into a powerful material that helps pull arsenic out of water, and then be quickly collected with a magnet for reuse.
Why Arsenic in Water Matters
Arsenic often seeps into groundwater from natural minerals and from human activities such as mining and industry. In some wells, its level is hundreds of times higher than what health agencies consider safe. The most harmful form, called As (III), is especially difficult to remove. Many existing treatment methods are either too costly, too complex, or produce sludge that is hard to handle. That has driven scientists to search for low‑cost, easy‑to‑use materials that can capture arsenic effectively without creating new environmental problems.
Making Value from Citrus Peels
In this work, the researchers used waste peels of Citrus pseudolimon, a type of lemon widely grown in India, as their starting point. They first heated the dried, ground peels in the absence of air to turn them into a charcoal‑like solid known as biochar. This biochar is full of tiny pores and reactive surface groups that can latch onto pollutants. The team then combined the biochar with very small particles of iron oxide, which respond strongly to magnets, and with a zinc–aluminum layered material known for its ability to exchange negatively charged particles. The final product is a dark, porous, magnetic powder called M‑CPB/LDH that can be stirred into contaminated water and later removed simply by applying a magnet. 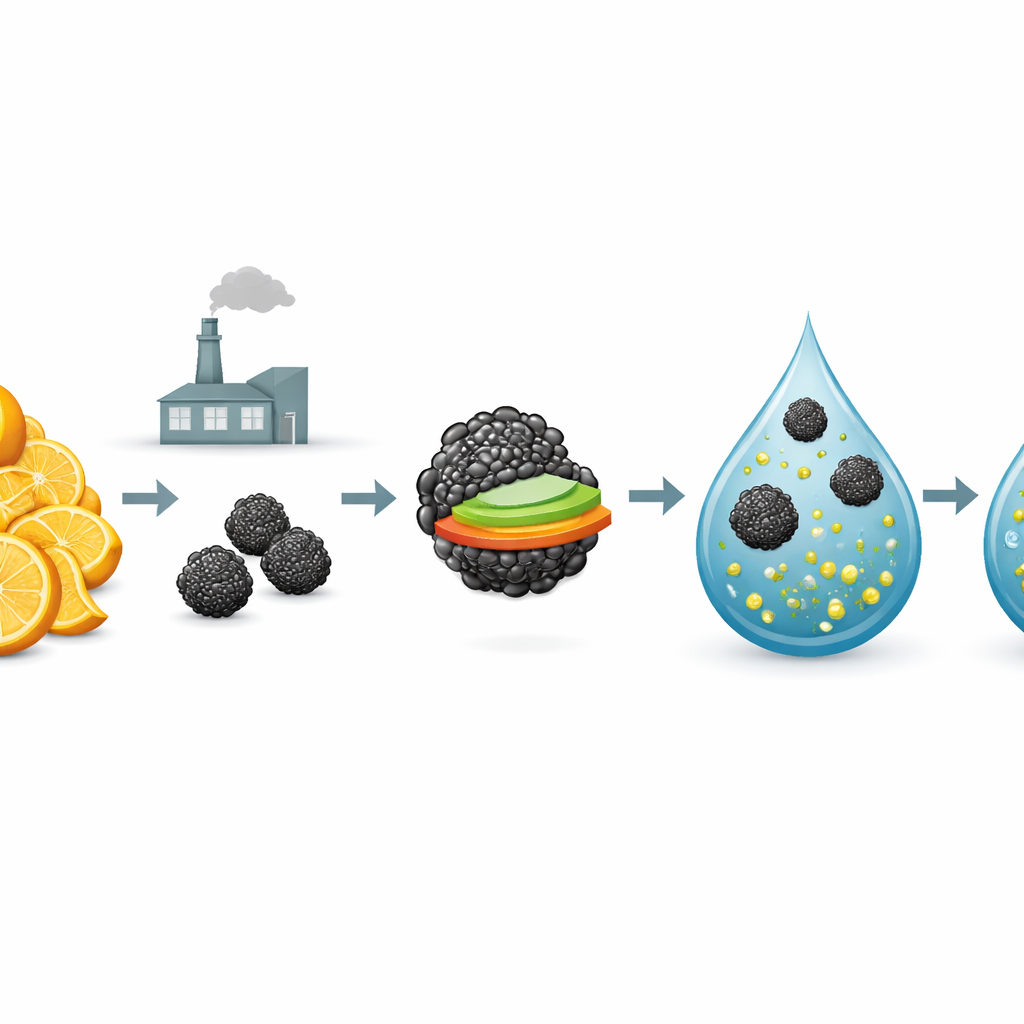
How the New Material Grabs Arsenic
The scientists carefully examined the structure of their material using a suite of modern tools that reveal its chemistry, internal layers, pore sizes, and magnetic behavior. They found that adding the layered zinc–aluminum component and magnetic iron oxide nearly doubled the surface area compared with biochar alone, creating more room for arsenic to attach. Tests across a range of conditions showed that the material works best in slightly acidic water, around pH 4, and that higher temperatures help rather than hurt its performance. Detailed analysis of how fast and how much arsenic is taken up suggests that the arsenic forms a single coating layer on the surface and binds strongly through chemical interactions rather than just weak physical sticking.
Peeking into the Clean‑Up Process
By combining microscopic imaging and surface‑sensitive techniques, the team could see arsenic present on the material after treatment and track changes in the bonding of its metal atoms. These observations support a picture in which arsenic species in water are first drawn toward positively charged sites on the composite and then swap places with surface groups such as hydroxyls, forming more permanent metal–oxygen–arsenic linkages. The many pores in the biochar allow arsenic to penetrate into the particle rather than only coating the outside. Because iron oxide is built in, the loaded particles can be pulled out in seconds with a simple magnet, avoiding slow filtration or energy‑intensive centrifugation. 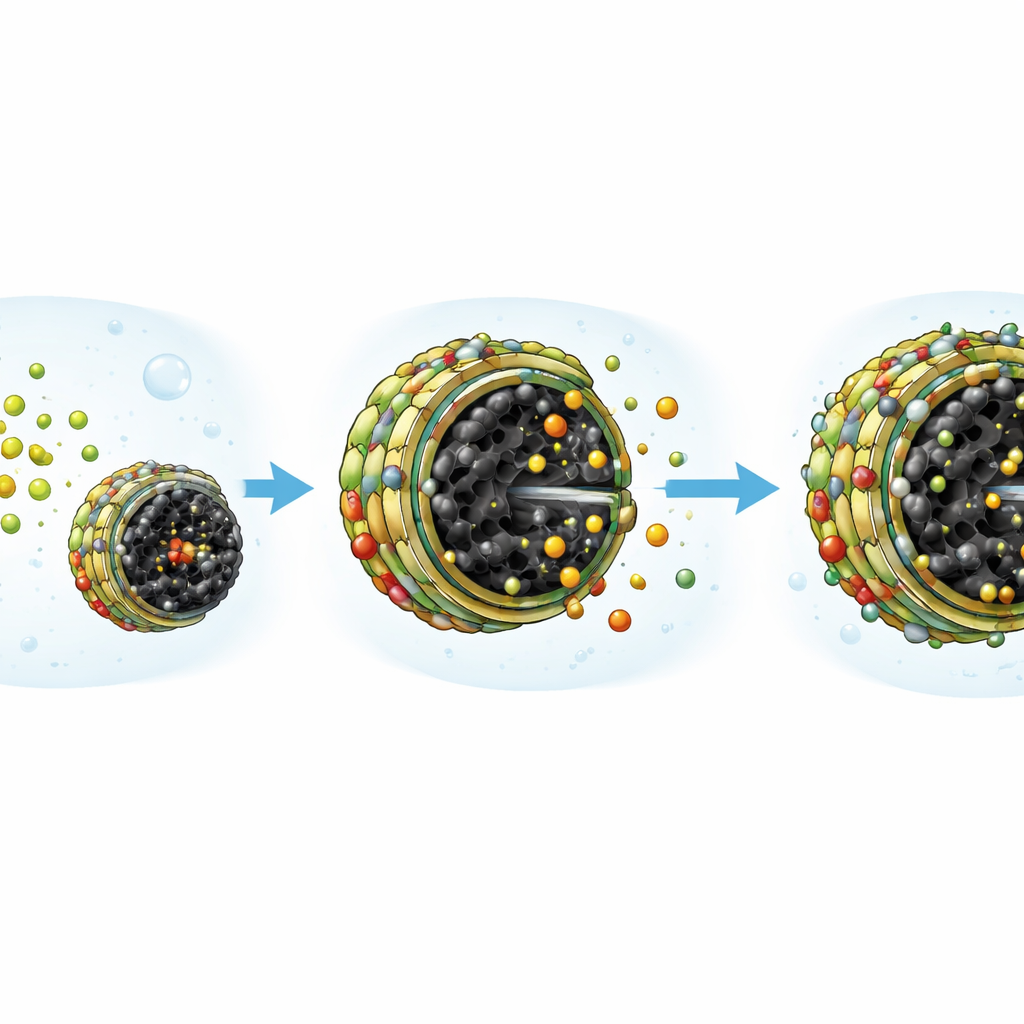
Performance, Reuse, and Real‑World Promise
When tested in laboratory solutions, the citrus‑based material captured more arsenic per gram than many similar adsorbents reported in the literature, with the best version (M‑CPB/LDH) outperforming both plain biochar and the magnetic layered material without biochar. Under optimal conditions it removed more than 96 percent of As (III) from water and showed that its structure remains stable at high temperatures. Just as important for real‑world use, the particles could be washed with a mild acid solution to release the trapped arsenic and then reused at least seven times, still retaining over 90 percent of their original removal ability.
What This Means for Safer Water
To a non‑specialist, the main takeaway is that this study turns an everyday waste—lemon peels—into a smart, magnet‑friendly sponge for one of the world’s most dangerous water contaminants. The combination of natural biochar, a layered metal coating, and built‑in magnetism creates a material that is effective, relatively inexpensive, and easy to handle. While further tests with real wastewater and complex mixtures of pollutants are still needed, the work points toward practical filters or treatment units where farmers’ or juice‑industry waste could help protect communities from long‑term arsenic exposure.
Citation: Sharma, S., Sharma, N., Somvanshi, A. et al. Waste citrus pseudolimon peels derived biochar assisted magnetic Zn + Al (LDH) nanocomposites for As (III) adsorption. Sci Rep 16, 11645 (2026). https://doi.org/10.1038/s41598-026-40288-x
Keywords: arsenic removal, biochar, citrus peel waste, magnetic nanocomposite, water purification