Clear Sky Science · en
Nanoscale solid-fluid interaction and amphibole formation in the lithospheric mantle
Hidden Highways Deep Below Our Feet
Far beneath the continents, rocks in Earth’s mantle are not as solid and unchanging as they seem. Tiny amounts of hot, pressurized fluid move through this deep, dark realm and quietly reshape the minerals they touch. This study peers down to the nanometer scale—billions of a meter—to show how a common mantle mineral is transformed into a water-bearing one, and how that process creates microscopic “highways” that help carbon-rich fluids escape toward the surface. Understanding these hidden interactions can illuminate how Earth stores and releases water and carbon over geologic time.
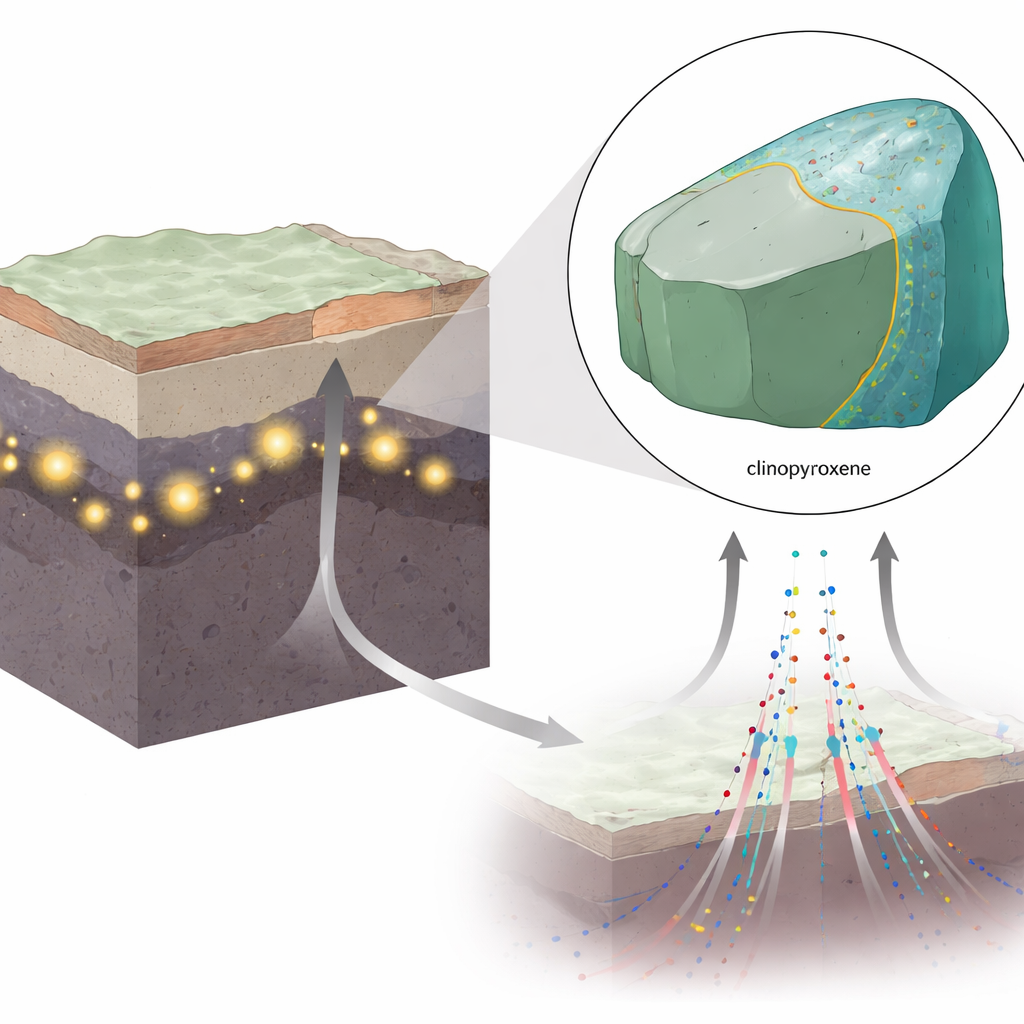
Fluids in the Deep Earth
In the upper mantle, rocks contain scattered pockets of supercritical fluid, mostly carbon dioxide with some water. Under the extreme pressures and temperatures at 70 kilometers depth and deeper, this fluid behaves neither like an ordinary liquid nor a gas. It seeps into cracks and grain boundaries within the rock and can become trapped as tiny inclusions inside minerals. The xenolith studied here—a piece of mantle rock brought to the surface by volcanic activity from the Perșani Mountains in Central Europe—contains such trapped pockets inside a mineral called clinopyroxene, alongside thin slivers of another mineral, amphibole, that is rich in water stored within its crystal structure.
A Thin Film That Starts a Big Change
The authors combined high-resolution electron microscopy with chemical modeling to reconstruct what happens at the boundary between the trapped fluid and the host clinopyroxene crystal. They argue that even when the fluid as a whole is rich in carbon dioxide, water molecules concentrate along the mineral surface, forming an ultra-thin, water-rich film only a few nanometers thick. In this film, water carries dissolved fragments of the surrounding rock, including sodium, aluminum and silica. Together, the outermost layer of the clinopyroxene plus this hydrous film gradually approach the composition needed to form amphibole, setting the stage for a new mineral to grow right at the solid–fluid interface.
From Solid Edge to Water-Rich Mineral
Over time, tiny distortions and defects in the crystal surface trigger local dissolution of clinopyroxene into the hydrous film, oversaturating it with the ingredients required to build amphibole. Amphibole then begins to re-precipitate exactly where clinopyroxene dissolves, turning a simple two-part system—solid plus fluid—into a more complex three-part one: clinopyroxene, amphibole, and residual fluid. The water-rich film thins as its components are locked into the growing amphibole, and the remaining trapped fluid becomes relatively richer in carbon dioxide. The study translates these nanoscale rearrangements into a chemical “reaction recipe,” showing how water-bearing complexes in the fluid feed the growth of amphibole while freeing extra silica and calcium back into the fluid.
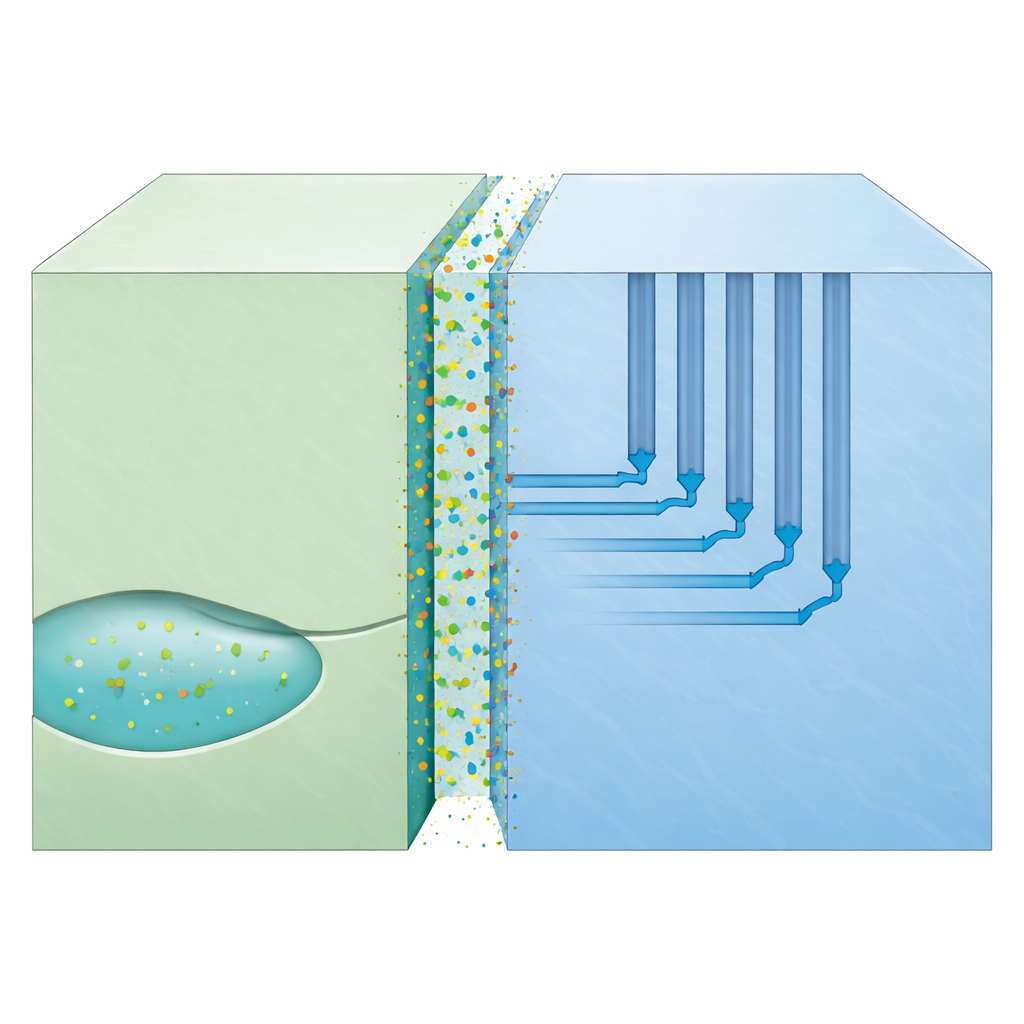
Nanochannels: Invisible Pipes Through Solid Rock
As amphibole replaces clinopyroxene, the mismatch between their crystal structures does something unexpected: it creates long, narrow, empty spaces—nanochannels—along the boundary where the two minerals meet. These channels are only a few billionths of a meter wide, but they run along preferred directions in the crystal, forming efficient pathways for fluid components to move even where ordinary pores are absent. Water and certain elements cling to the channel walls, forming bonds that actually help pull atoms like sodium and aluminum along the interface. In deformed mantle zones, where crystals of clinopyroxene and amphibole share similar orientations, many channels can align, forming organized networks that guide carbon-rich fluids through otherwise impermeable rock.
From Nanoscale Films to Global Gas Release
The authors conclude that the formation of amphibole from clinopyroxene in the presence of supercritical fluids is a key step in how the deep lithosphere evolves chemically and mechanically. At the tiniest scale, water-rich films at mineral surfaces and the nanochannels they help create lock water and many rock-loving elements into new minerals while leaving the remaining fluid enriched in carbon dioxide. Those carbon-rich fluids can then travel upward along deep-seated weakness zones in the lithosphere, contributing to the steady, non-volcanic release of CO2 from Earth’s interior. In short, this work shows how nanometer-thick films and channels in mantle rocks can influence planet-scale cycles of water and carbon.
Citation: Lange, T.P., Pósfai, M., Berkesi, M. et al. Nanoscale solid-fluid interaction and amphibole formation in the lithospheric mantle. Sci Rep 16, 11009 (2026). https://doi.org/10.1038/s41598-026-40179-1
Keywords: lithospheric mantle, amphibole, supercritical fluid, nanochannels, mantle degassing