Clear Sky Science · en
Oxidative stress imbalance and cellular damage mediated by the ND4 G11778A mutation
Why a tiny flaw in cell batteries can steal sight
Some young adults, mostly men, suddenly lose central vision in both eyes, often without warning. This condition, called Leber hereditary optic neuropathy, is caused by defects in the tiny power plants inside our cells, the mitochondria. This study investigates one of the most common and severe genetic changes linked to the disease and shows, in detail, how it throws the cell’s chemistry off balance and injures the delicate nerve fibers that carry visual information from the eye to the brain.
A hidden weakness in the eye’s wiring
The researchers focused on a single letter change in mitochondrial DNA, known as ND4 G11778A (also called R340H). This mutation affects complex I, a key component of the machinery mitochondria use to turn nutrients into usable energy. Because the optic nerve and light-sensing cells in the retina are among the most energy-hungry tissues in the body, even a modest drop in power supply can be disastrous. Earlier work had linked this mutation to poor vision and limited recovery, but the exact chain of events from genetic flaw to nerve damage was not fully understood.
Putting the mutation to the test in nerve-like cells
To probe this chain of events, the team engineered mouse retinal cells (called 661W cells) to produce either normal ND4 or the mutant version. They then grew these cells under two conditions: one rich in glucose, where cells can rely heavily on standard sugar breakdown, and another with galactose, which forces cells to lean on mitochondria for energy. Using a specialized instrument to measure oxygen consumption, they found that cells carrying the mutant ND4 had the weakest mitochondrial “breathing,” especially when they were pushed to rely on their mitochondria. Their basal and maximal oxygen use dropped, and their capacity to boost energy output when needed was sharply reduced, revealing a serious power shortage.
When power failure sparks chemical burnout
Power failure in mitochondria rarely comes alone. As the energy machinery falters, it tends to leak highly reactive molecules known as reactive oxygen species, which can attack proteins, fats, and DNA. The mutant ND4 cells produced more of these harmful byproducts, particularly under the energy-stressful galactose condition. At the same time, the cell’s natural defenses—enzymes and small molecules that normally mop up these reactive species—were weakened. Levels of key protectors such as superoxide dismutase, catalase, and glutathione all fell in the mutant cells. This double hit of more damage and less protection created a marked oxidative imbalance that pushed the cells toward injury.
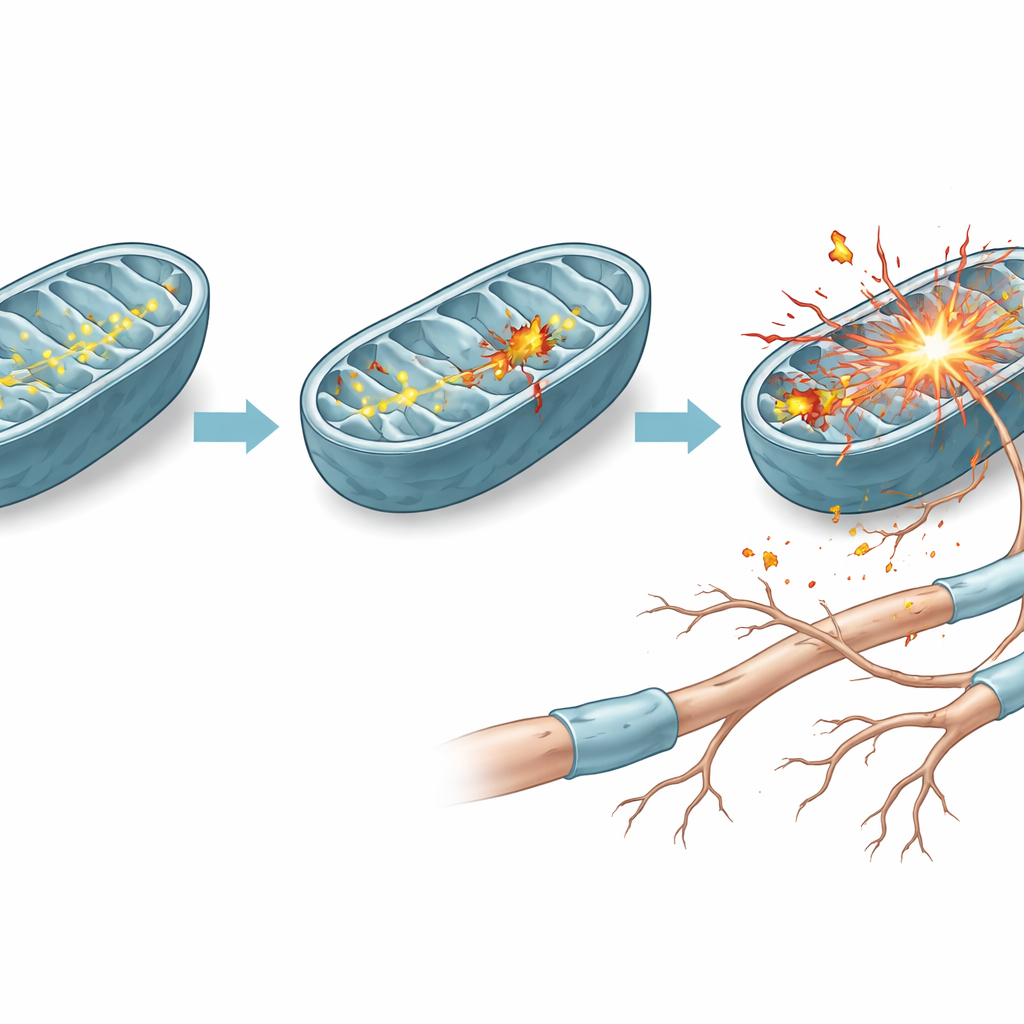
From stressed cells to dying nerve fibers
The consequences of this imbalance showed up clearly in cell survival tests. Under everyday, high-sugar conditions, cells with and without the mutation looked similar. But when forced to depend on mitochondrial power, cells with the mutant ND4 declined in number, showed poorer viability, and displayed more signs of programmed cell death. The team confirmed this by staining for fragmented DNA, a hallmark of apoptosis, and by measuring drops in a survival-related protein. To see whether the same processes occur in living animals, they delivered the mutant ND4 to one eye of mice and the normal version to the other. Months later, electron microscopy revealed that optic nerves exposed to the mutant gene had fewer and more disrupted nerve fibers, mirroring the damage seen in human disease.
What this means for protecting vision
In simple terms, the study shows that this single mitochondrial mutation weakens the cell’s power supply, increases toxic byproducts, blunts the natural clean-up system, and ultimately drives nerve cells in the eye toward self-destruction. The work clarifies why optic nerves are so vulnerable in Leber hereditary optic neuropathy and highlights several points where treatment might help: shoring up mitochondrial function, reducing oxidative stress, or boosting antioxidant defenses. While the experimental setup does not capture every detail of the human condition, it offers a clear mechanistic map that can guide future therapies aimed at preserving sight.
Citation: Fang, L., Fu, K., Yang, M. et al. Oxidative stress imbalance and cellular damage mediated by the ND4 G11778A mutation. Sci Rep 16, 10122 (2026). https://doi.org/10.1038/s41598-026-40061-0
Keywords: mitochondria, oxidative stress, optic nerve, Leber hereditary optic neuropathy, ND4 mutation