Clear Sky Science · en
An exploratory in vitro co-culture of enteric neurons and smooth muscle cells demonstrates neuronal contribution to muscle layer formation
Why Growing New Gut Muscle Matters
For people born with, or who lose, large parts of their small intestine, everyday life can depend on intravenous feeding because their remaining gut cannot absorb enough nutrients. Transplants are risky and often fail over time. This study explores a very different idea: can scientists grow working pieces of intestinal muscle in the lab, complete with their own built-in "gut brain" to coordinate movement? If so, it could one day help build replacement bowel tissue for patients with short bowel syndrome.
The Hidden Brain Inside the Gut
The wall of the intestine is more than a simple tube. It contains layers of smooth muscle that rhythmically squeeze food along, guided by a dense network of nerves called the enteric nervous system, sometimes nicknamed the "brain in the gut." When these nerves are missing or damaged, as in Hirschsprung’s disease or after major surgery, the muscle layer alone cannot move food properly. The authors reasoned that any lab-grown replacement tissue must therefore include both muscle cells and enteric nerve cells, not just one or the other, and set out to build a stripped-down model containing only these two key players.
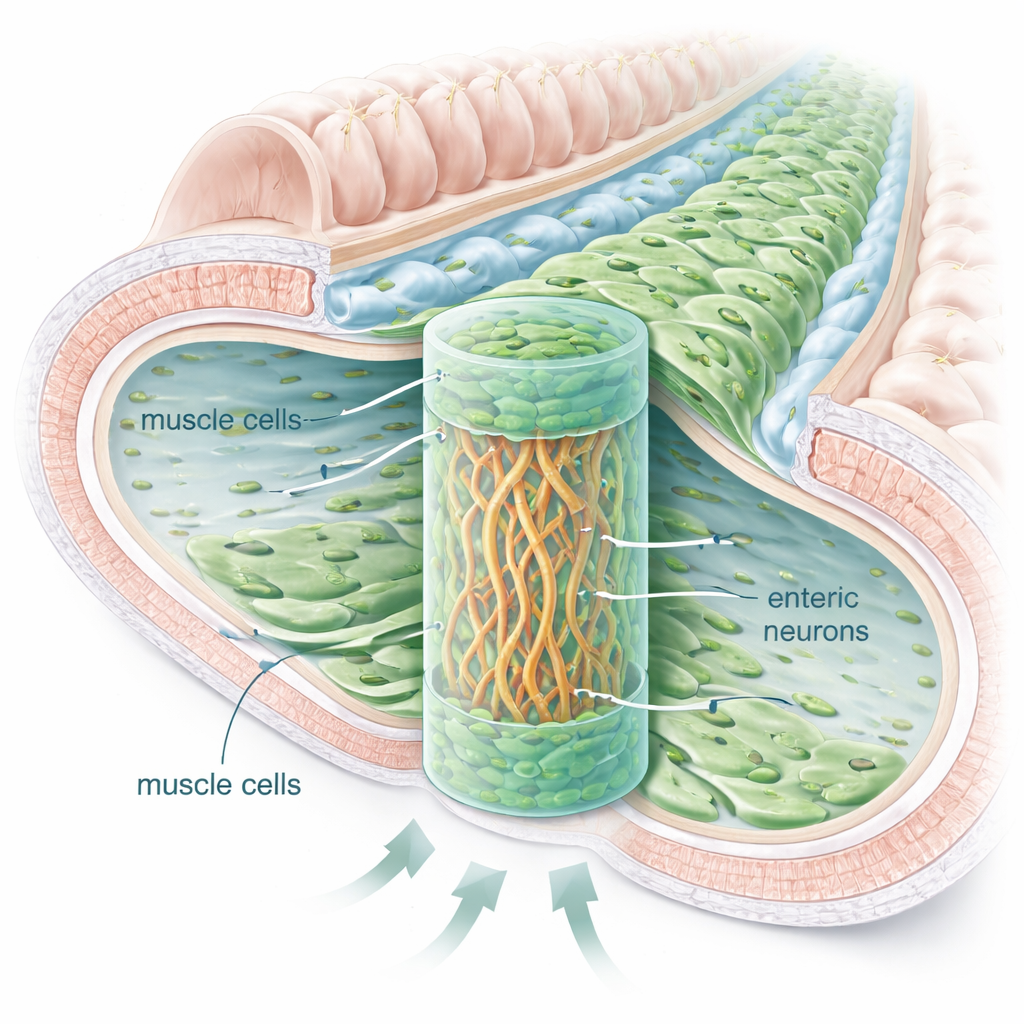
Building a Mini Gut Wall in a Gel
The team isolated enteric nerve cells from young rats and combined them with commercially sourced intestinal smooth muscle cells inside a soft, jelly-like scaffold made from hyaluronic acid. They tested different liquid nutrient mixes until they found one that supported both cell types, then experimented with how to arrange the cells in three dimensions. The most successful layout resembled a sandwich: a dense band of nerve cells in the middle layer of the gel, flanked above and below by smooth muscle cells. In this setup, both cell types survived for weeks to months and formed layered structures reminiscent of the natural intestinal wall.
From Random Cells to Organized, Moving Fibers
Microscopy revealed that the presence of enteric nerve cells dramatically changed how the muscle cells behaved. Alone in the gel, smooth muscle cells stayed round, weakly expressed their contractile proteins, and failed to line up into fibers. When nerve cells were present nearby or in direct contact, the muscle cells elongated, aligned with one another, and formed long bundles similar to native muscle layers. The researchers saw networks of nerve fibers weaving between the muscle bundles, along with glial cells that normally support neurons. Using fluorescent markers and electron microscopy, they identified structures resembling synapses—tiny contact points where nerves communicate with muscle cells.
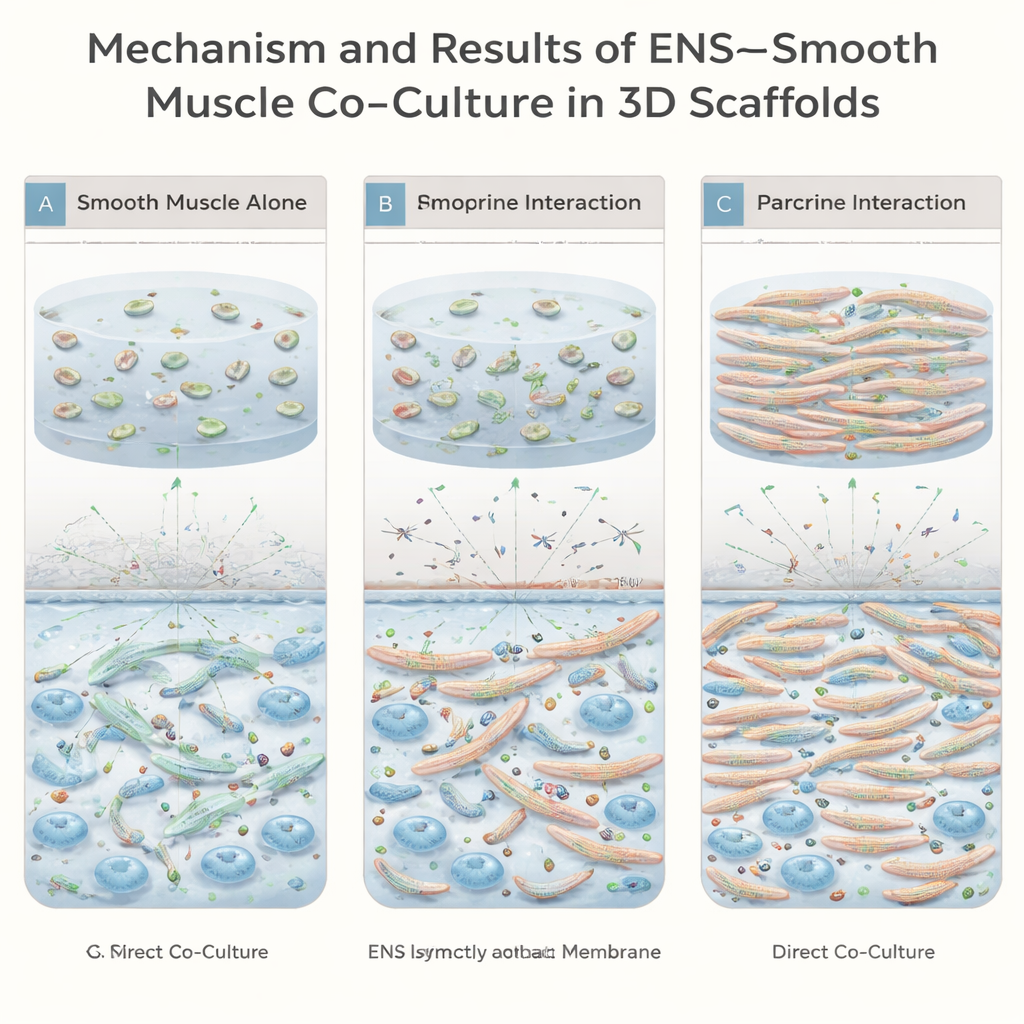
Do These Lab-Grown Fibers Actually Contract?
Starting around three weeks in culture, the constructs that contained both muscle and enteric nerve cells began to show spontaneous contractions visible under a light microscope. Thin and thick muscle bundles repeatedly shortened and relaxed, suggesting that the engineered tissue could actively move rather than just sit passively in the gel. Direct co-culture, where nerve and muscle cells touched each other inside the same scaffold, produced the most robust, well-aligned fibers and the richest nerve networks. In comparison, when the two cell types were separated by a membrane that allowed only soluble signals to pass, muscle fibers did form but were fewer, weaker, and less organized.
What This Means for Future Gut Repair
This work does not yet prove that the contractions are fully controlled by the nerve cells, nor does it recreate the entire intestinal wall with its lining and immune cells. Still, it provides a clear proof-of-concept: enteric nerve cells can help guide smooth muscle cells to organize into aligned, innervated bundles that behave more like living gut muscle. For patients with short bowel syndrome, such simplified but functional muscle layers are a crucial step toward building replacement intestinal segments. Future studies combining these neuromuscular constructs with the gut’s inner lining and more detailed tests of nerve-controlled motion could bring tissue-engineered intestine closer to clinical reality.
Citation: Khasanov, R., Tapia-Laliena, M.Á., Schulte, S. et al. An exploratory in vitro co-culture of enteric neurons and smooth muscle cells demonstrates neuronal contribution to muscle layer formation. Sci Rep 16, 7732 (2026). https://doi.org/10.1038/s41598-026-39409-3
Keywords: short bowel syndrome, intestinal tissue engineering, enteric nervous system, smooth muscle, 3D co-culture