Clear Sky Science · en
Production of low sulfur diesel fuel through ultrasonic catalytic oxidative route using novel mixed oxides nanocomposites assisted by solvent extraction
Why Cleaning Up Diesel Matters
Every time a diesel engine runs, tiny sulfur compounds in the fuel can turn into harmful gases and fine particles that pollute the air and endanger human health. Governments worldwide now demand very low-sulfur fuels, but the standard industrial cleanup method is energy-hungry and expensive. This study explores a smarter way to strip sulfur from real diesel fuel using sound waves, smart nanomaterials, and a final washing step with carefully chosen solvents, aiming to deliver cleaner fuel with less energy and lower cost.
A New Path Beyond Traditional Refinery Methods
Refineries usually rely on hydrodesulfurization, a process that forces sulfur out of fuels at high temperature and pressure with large amounts of hydrogen gas. While effective for simple sulfur compounds, it struggles with stubborn ring-shaped molecules that cling tightly to diesel, and it consumes significant energy and costly hydrogen. The researchers set out to develop an alternative that could work at much milder conditions. Their approach combines oxidative chemistry, which converts sulfur into easier-to-remove forms, with ultrasound, which uses high-frequency sound waves to shake and mix the reacting liquids more efficiently.
Tiny Mixed-Metal Helpers
At the heart of the new method are mixed-metal oxide nanoparticles made from iron, cobalt, and nickel. These particles were prepared by a simple precipitation route, then heated to form tiny, solid oxide grains. By adjusting how long the particles were allowed to form—from half an hour up to eight hours—the team tuned their size, internal structure, magnetism, and surface area. The sample made in just 0.5 hours, called T1, had the highest surface area, small and fairly uniform particles, and many accessible pores. These features made it especially good at contacting sulfur molecules in diesel and at activating hydrogen peroxide, the chosen oxidizing agent, to attack sulfur.
Sound Waves Supercharge the Reaction
The desulfurization step takes place in a liquid mixture of diesel, hydrogen peroxide, and the Fe–Co–Ni catalyst while ultrasound is applied. The sound waves create microscopic bubbles that rapidly grow and collapse, generating tiny hot spots and intense local mixing. Under carefully chosen conditions—around 60 °C, 90 minutes of treatment, a 1:1 volume ratio of hydrogen peroxide to diesel, and 10 grams of catalyst per liter—the process oxidizes a large fraction of the sulfur compounds into more polar products called sulfones. With the best unmodified catalyst (T1), the sulfur content of the diesel was cut by more than half in this single step, and the study mapped how reaction time, temperature, oxidant amount, and catalyst dose each influenced performance and stability.
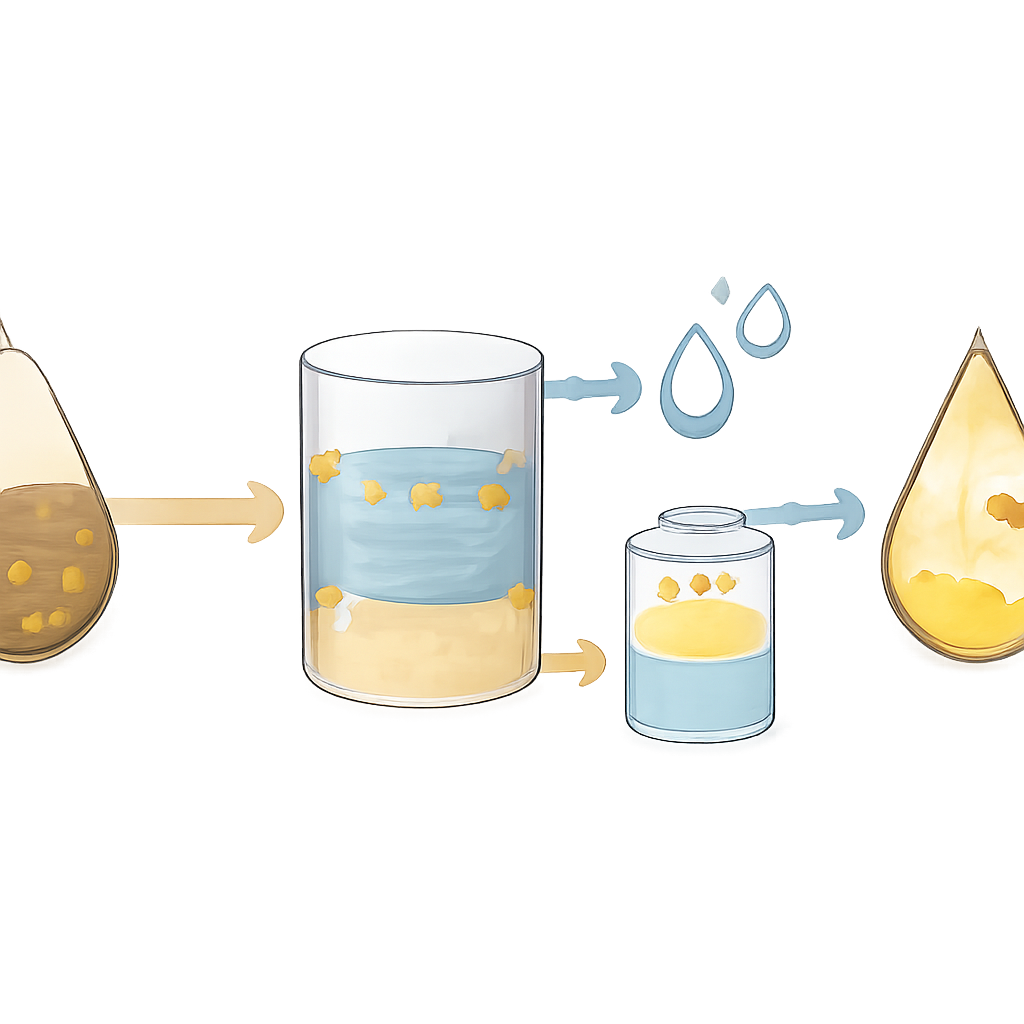
From Oxidation to Washing Out the Sulfur
Oxidation alone does not remove the sulfur from the liquid; it changes the sulfur compounds into forms that prefer to sit in polar liquids rather than in the oily diesel. The researchers therefore followed the ultrasonic step with a solvent extraction stage, where the treated fuel was contacted with a polar solvent that selectively pulled the oxidized sulfur out. They compared several options and found that a mixture of dimethylformamide (DMF) and acetonitrile worked especially well. With a 4:1 solvent-to-fuel ratio, this post-treatment raised total sulfur removal from about 55–60% after oxidation alone up to roughly 89%, showing that oxidation and extraction work far better together than either could on its own.
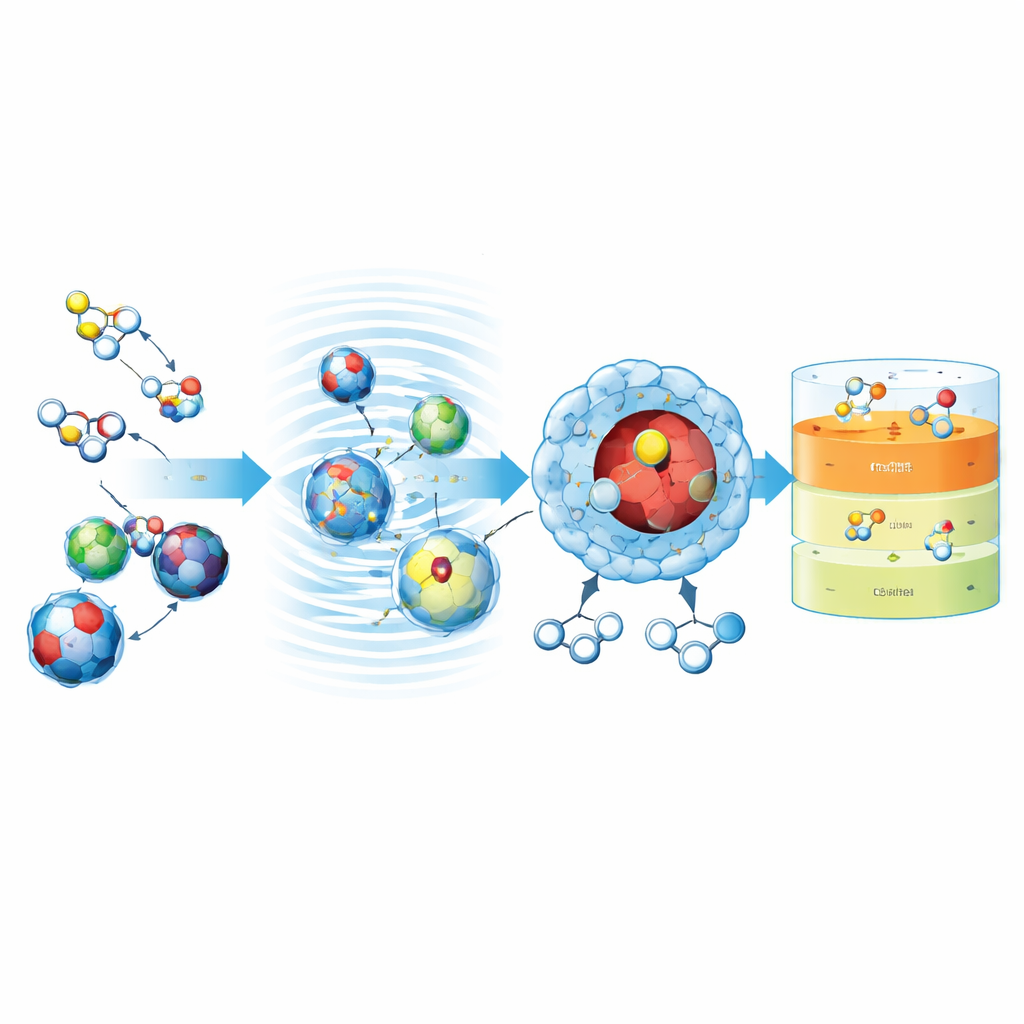
Boosting Performance with a Protective Shell
To push the process even further, the team wrapped the mixed-metal oxide particles in a thin layer of polystyrene, creating a core–shell structure. The polymer shell provides extra porosity and has aromatic rings that interact strongly with the ring-shaped sulfur molecules in diesel, helping to draw them toward the active metal-oxide core. Under optimized ultrasonic and solvent-extraction conditions, this modified catalyst lowered the sulfur content of a real diesel sample from about 21,700 parts per million to just 920 parts per million. That corresponds to roughly 96% sulfur removal, all achieved under relatively mild temperatures and without the extreme pressures and hydrogen demands of traditional refinery units.
What This Means for Cleaner Fuels
In plain terms, the study shows that combining smart nanomaterials, sound waves, and a tailored solvent wash can strip most of the sulfur out of real diesel fuel without the heavy energy bill of conventional methods. The mixed-metal particles speed up the chemistry, ultrasound helps the different liquids meet and react, and the final solvent step physically carries the oxidized sulfur away. Although further engineering will be needed to scale this approach and meet the most stringent ultra-low-sulfur targets, the work points toward more flexible, energy-efficient fuel-cleaning technologies that could help reduce air pollution and ease the environmental footprint of diesel use.
Citation: Zahran, A.I., El-Fawal, E.M., Naggar, A.M.A.E. et al. Production of low sulfur diesel fuel through ultrasonic catalytic oxidative route using novel mixed oxides nanocomposites assisted by solvent extraction. Sci Rep 16, 12058 (2026). https://doi.org/10.1038/s41598-026-39220-0
Keywords: diesel desulfurization, ultrasonic catalysis, nanoparticle catalysts, oxidative cleaning, low-sulfur fuel