Clear Sky Science · en
Innovative sodium benzoate-modified hydroxyapatite for enhanced dye removal using a combined experimental and DFT approach
Cleaning Color from Dirty Water
Across the globe, brightly colored industrial dyes that make our clothes, pills, and cosmetics so appealing are quietly staining rivers and lakes. Many of these dyes do not break down easily and can harm fish, ecosystems, and even human health. This paper explores a new, low-cost material inspired by bone mineral that can pull a common blue dye out of water efficiently, pointing toward simpler treatment options for dye-contaminated wastewater.
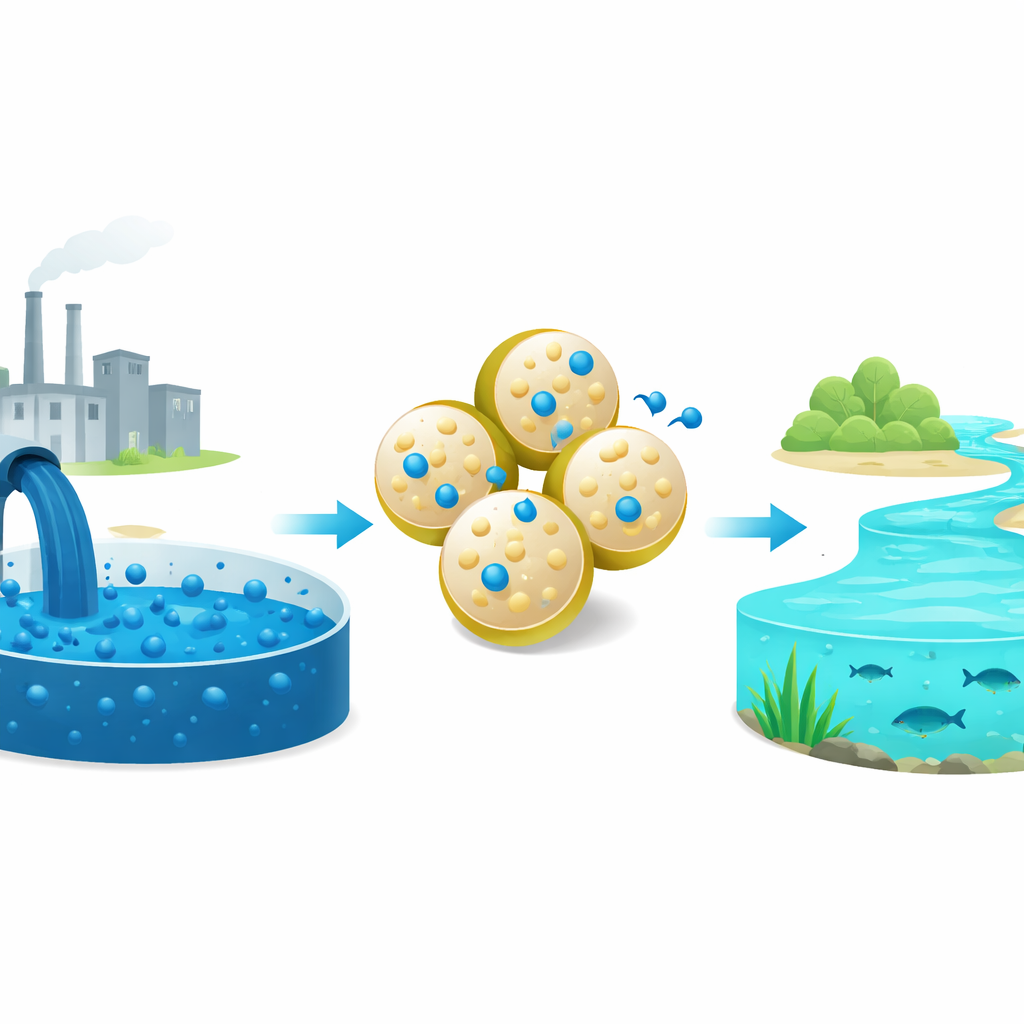
Borrowing Ideas from Bones
The central ingredient in this study is hydroxyapatite, a mineral that makes up much of our bones and teeth. Because it is rich in calcium and phosphate and already widely used in medicine, it is considered safe, cheap, and easy to produce. Scientists have recently realized that this mineral can also act like a tiny sponge for many pollutants in water, especially metals and some organic chemicals. However, its natural surface is not perfect for grabbing stubborn dye molecules, so the team looked for a way to “upgrade” it without losing its low cost and environmental friendliness.
Giving the Mineral a Smart Coating
The researchers modified hydroxyapatite by attaching sodium benzoate, a simple and inexpensive compound often used as a food preservative. They mixed the mineral’s building blocks with different amounts of sodium benzoate (5%, 10%, and 15% by weight) during synthesis so that the organic groups became part of the surface. A suite of techniques—X-ray diffraction, infrared spectroscopy, electron microscopy, and thermal analysis—showed that the bone-like crystal structure remained intact, while the surface became more porous, more orderly, and more heat-resistant. In other words, the mineral kept its strong skeleton but gained an active skin rich in negatively charged groups that can attract positively charged dye molecules.
Putting the New Material to Work
To test performance, the team focused on methylene blue, a widely used blue dye known for its persistence and potential health risks. They stirred the modified and unmodified powders in dye solutions under controlled conditions and tracked how much color disappeared from the water. The best material, containing 15% sodium benzoate, removed about 90% of the dye and stored roughly two-thirds more dye per gram than plain hydroxyapatite. Tests across different pH values showed that the sodium benzoate coating makes the particle surfaces more negatively charged under typical treatment conditions, which strongly attracts the positively charged dye. Time-based experiments revealed that most removal happens in the first 20 minutes and follows a pattern consistent with a controlled, chemical-like binding rather than weak, reversible sticking.
Peeking into the Invisible Interactions
To understand why the coating works so well, the researchers used advanced computer calculations based on quantum chemistry. These simulations examined how electrons are distributed within the sodium benzoate molecule and how it sits on the hydroxyapatite surface. The results indicate that the carboxylate part of sodium benzoate forms strong, but still non-destructive, interactions with calcium sites on the mineral. This anchoring creates stable negative patches and aromatic rings that, together, favor strong electrostatic attraction and stacking interactions with the methylene blue molecules. Theoretical models of dye uptake matched the experiments, suggesting that dye molecules form a tidy one-molecule-thick layer on the modified surface rather than piling up in a disordered way.
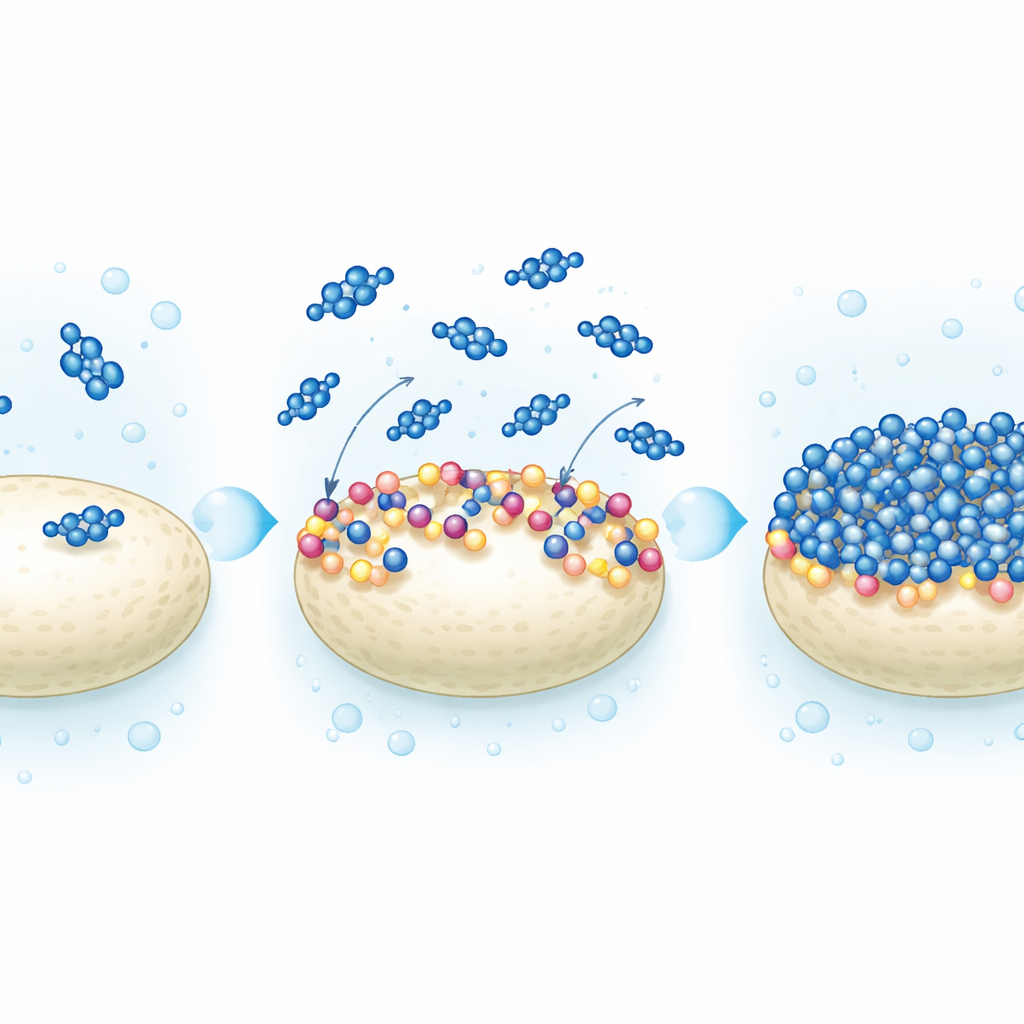
Toward Cleaner Water with Simple Powders
In everyday terms, the study shows that giving a bone-like mineral a thin “preservative” coating transforms it into a powerful color sponge: it pulls dye molecules from water quickly, holds them tightly in a single layer on its surface, and remains stable under heat and in alkaline conditions. Because both ingredients—hydroxyapatite and sodium benzoate—are inexpensive and already considered safe, this approach could be scaled up for treating dye-laden wastewater from textiles, pharmaceuticals, and other industries, helping to return clearer, safer water to rivers and lakes.
Citation: Boukra, A., Boukra, O., Latifi, S. et al. Innovative sodium benzoate-modified hydroxyapatite for enhanced dye removal using a combined experimental and DFT approach. Sci Rep 16, 9870 (2026). https://doi.org/10.1038/s41598-026-39075-5
Keywords: wastewater treatment, dye adsorption, hydroxyapatite, methylene blue, sodium benzoate