Clear Sky Science · en
Disruption of neutrophil homeostasis is associated with functional alterations in mitochondria of critically ill COVID−19 patients
Why this matters for COVID-19 patients
Most people now know that COVID-19 is more than just a lung infection, but how exactly the virus turns the body’s own defenses against it is still being uncovered. This study looks closely at neutrophils, a common type of white blood cell, in patients with life-threatening COVID-19 in intensive care. By examining how these cells live, die, and use their tiny internal “power plants” (mitochondria), the researchers reveal a pattern of stubborn, overactive neutrophils that may help explain persistent inflammation, blood damage, and organ injury in severe disease.
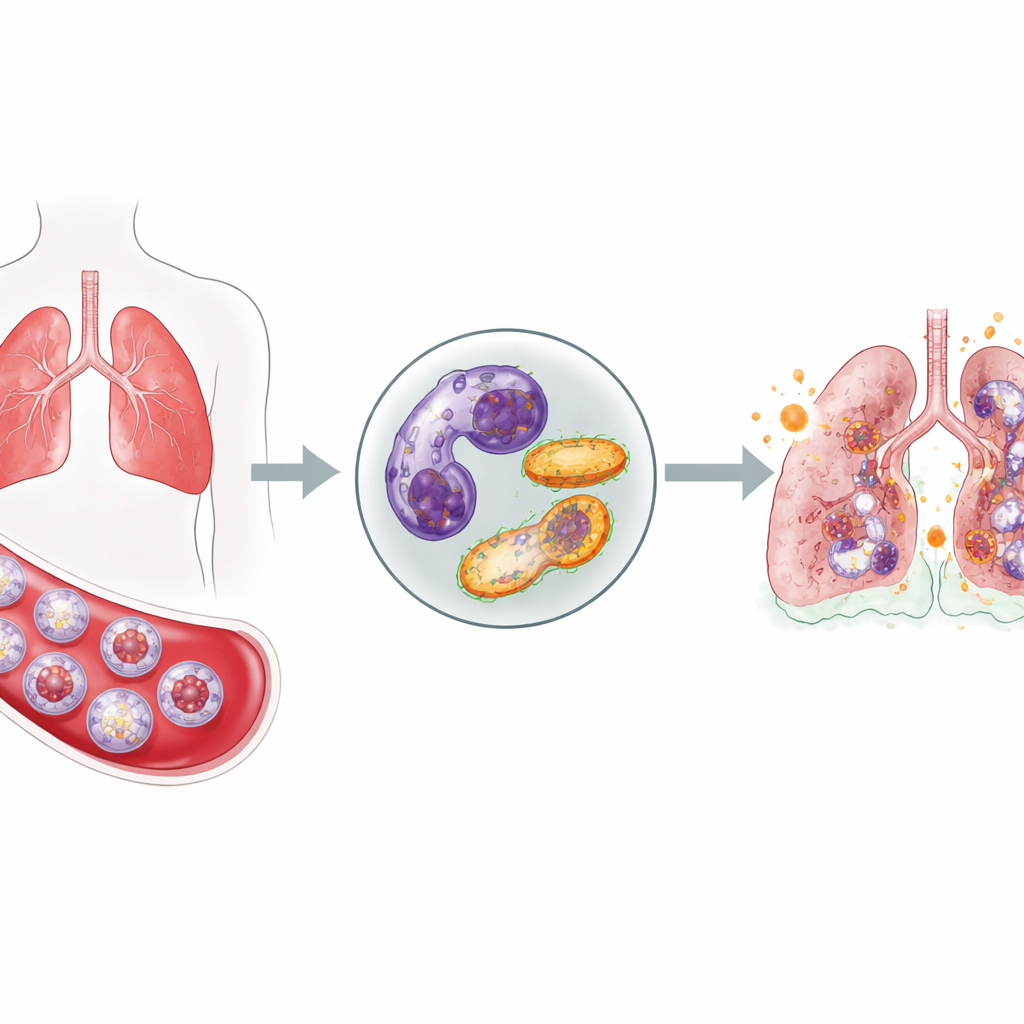
Front-line cells that do not stand down
Neutrophils are among the body’s first responders to infection: they rush into the bloodstream, move into tissues, and release toxic molecules to kill invading microbes. Normally, after doing their job, they quietly self-destruct and are cleared away, which prevents them from harming healthy tissue. In critically ill COVID-19 patients, the team found a very different picture. Compared with healthy volunteers, these patients had a surge of neutrophils in their blood and, importantly, many of them were immature cells that had been hurried out of the bone marrow. At the same time, multiple tests showed that fewer neutrophils were undergoing their normal programmed death, suggesting that these aggressive cells linger longer than they should.
Subtle genetic signals but a clear survival pattern
The researchers also looked at tiny regulatory molecules called microRNAs in neutrophils from intensive care survivors and non-survivors. Some of these molecules were altered in ways that, on paper, point toward disrupted pathways controlling cell death and calcium balance. However, when they measured direct markers of cell death, they did not see major differences between survivors and non-survivors—both groups showed similarly reduced neutrophil self-destruction compared with healthy people. This means the microRNA patterns are more like leads for future work than firm indicators of who will live or die, and they underline that the overall failure of neutrophils to switch off is a shared feature of severe COVID-19.
Calcium shifts and overcharged power plants
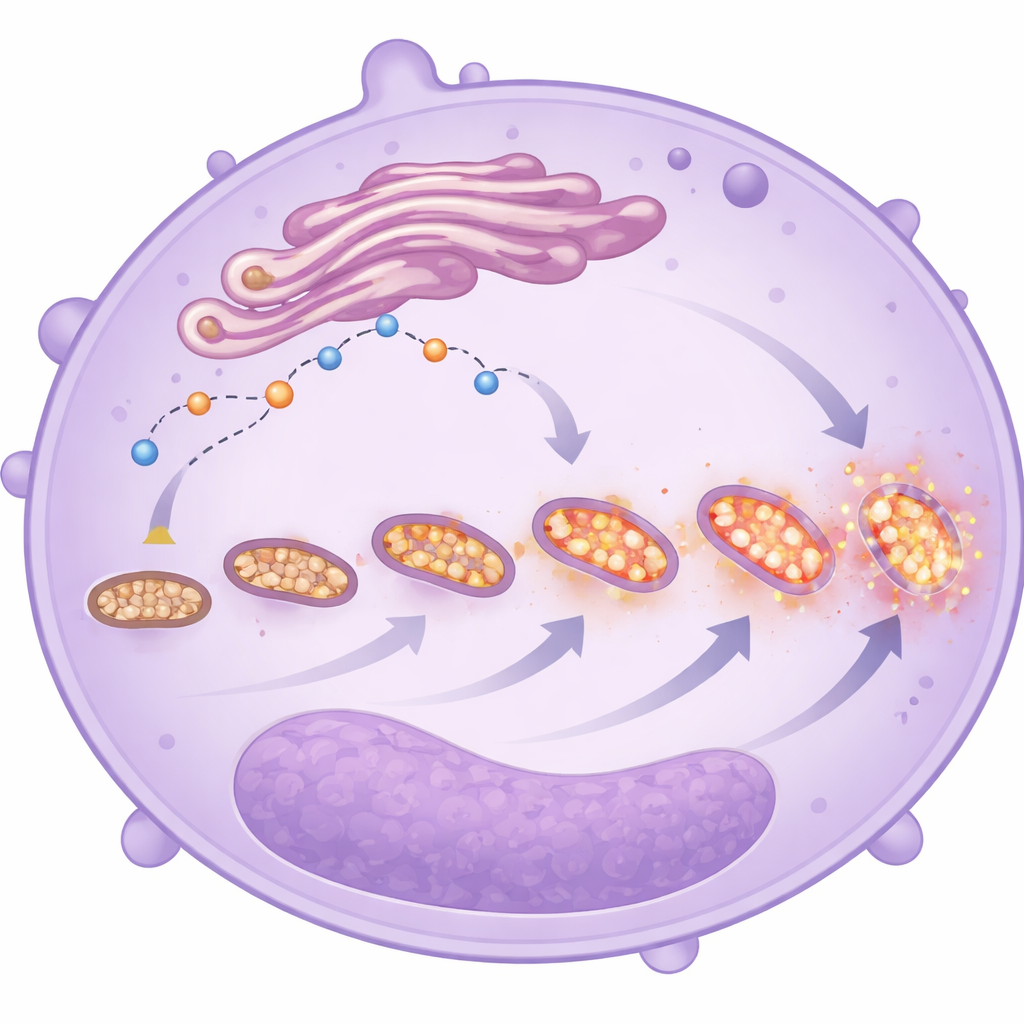
To understand why neutrophils were not dying on schedule, the team focused on two closely linked systems inside the cells: calcium signals and mitochondria. Calcium ions act as tiny on–off switches for many cell decisions, including when to self-destruct. The researchers found that neutrophils from severe COVID-19 patients had less free calcium inside, along with reduced levels of key proteins that normally help move calcium into mitochondria and open a safety valve that can trigger cell death. At the same time, the mitochondria themselves were unusually “charged up,” with a higher electrical gradient across their membranes and increased oxygen consumption, especially through one major respiratory pathway. Instead of shutting down, these overactive mitochondria were producing more reactive oxygen molecules, which can damage surrounding tissues and fuel inflammation.
Stressed and fragmenting mitochondria
Electron microscope images and fluorescent dyes provided a closer look at the shape and behavior of these mitochondria. In neutrophils from the sickest patients, mitochondria were more numerous but smaller and more fragmented—features that often signal stress. These cells also showed higher levels of mitochondrial reactive oxygen species, regardless of whether the patient ultimately survived. Together with the altered calcium handling and delayed cell death, these changes paint a picture of neutrophils that have shifted their energy strategy: rather than relying mainly on simple sugar breakdown, they lean more on mitochondrial metabolism, especially in immature cells. This switch may enhance their ability to migrate, persist, and release damaging molecules in the lungs and other organs, even though the study cannot prove they are the direct cause of worse outcomes.
What this could mean for future care
In plain terms, the study suggests that in critical COVID-19, neutrophils become too many, too young, and too hard to turn off. Their internal calcium signals and mitochondria are rewired in a way that keeps them alive and highly active, producing bursts of harmful oxygen-based chemicals. While the work is observational and does not show cause and effect, it highlights several potential “control knobs” in these cells—such as calcium regulators and mitochondrial checkpoints—that might one day be targeted to gently nudge neutrophils back toward normal behavior. If future research confirms these ideas, treatments that restore proper neutrophil shutdown or calm their overcharged mitochondria could help reduce the collateral damage these crucial cells cause during severe COVID-19 and possibly in other critical illnesses.
Citation: Elkhodiry, A.A., Yasseen, B.A., El-sayed, H. et al. Disruption of neutrophil homeostasis is associated with functional alterations in mitochondria of critically ill COVID−19 patients. Sci Rep 16, 7838 (2026). https://doi.org/10.1038/s41598-026-38741-y
Keywords: COVID-19, neutrophils, mitochondria, inflammation, critical illness