Clear Sky Science · en
Design, ecofriendly synthesis and spectroscopy of new pyrazolopyrimidine derivatives as potential candidates for dyeing and ultraviolet protection of wool fabrics
Brighter, Safer Wool for Everyday Wear
Wool clothing is prized for warmth and comfort, but traditional dyeing can be energy-hungry and many dyes do little to shield our skin from the sun’s ultraviolet (UV) rays. This study introduces a family of newly designed dyes that color wool vividly, are made by a more environmentally friendly method, and greatly boost the fabric’s ability to block UV radiation, all while preserving its natural strength and feel.
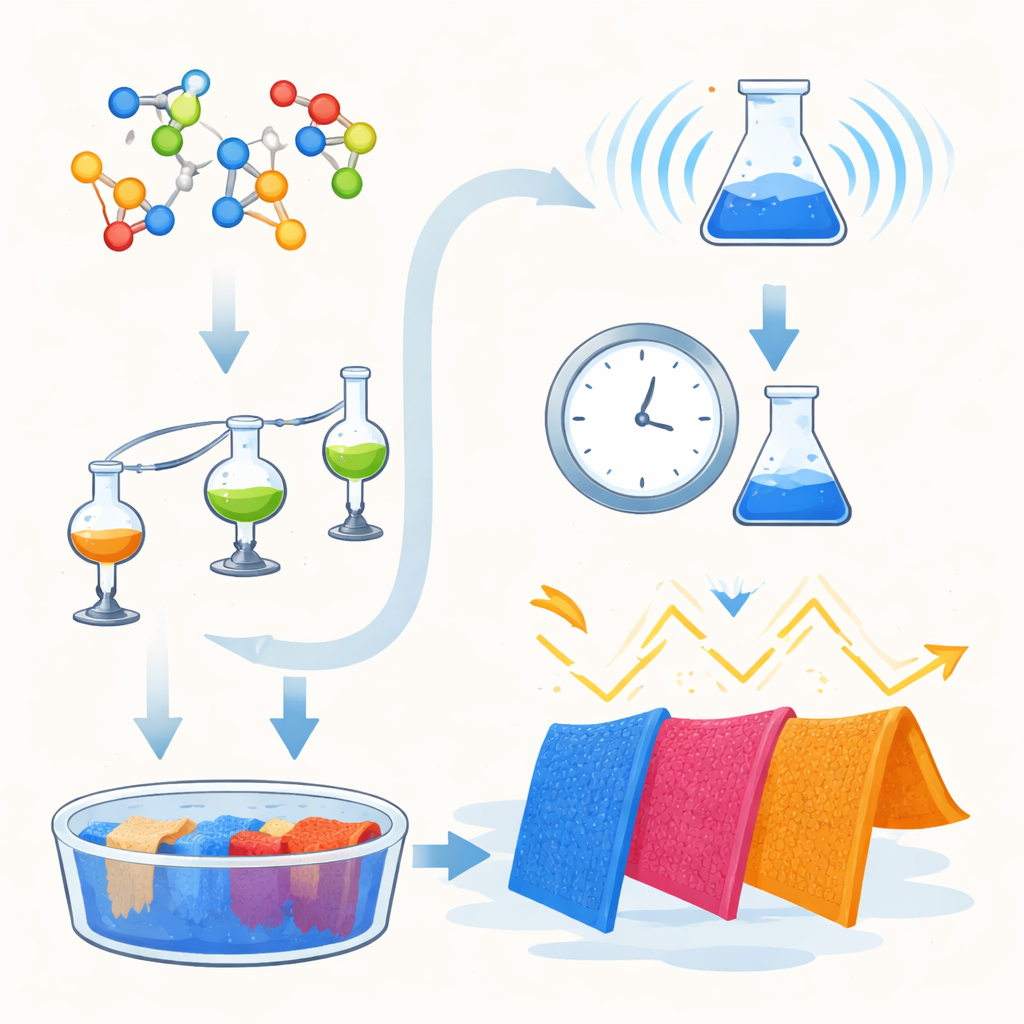
New Color Molecules with a Dual Purpose
The researchers focused on a class of color molecules called pyrazolopyrimidine dyes. These dyes are usually used on synthetic fibers such as polyester because they do not dissolve well in water. By redesigning the molecules to carry sulfonic acid groups, the team made them water‑soluble and able to behave like classic acid dyes, which are well suited to protein-based fibers such as wool. In total, they created six new related dyes, each carrying a different chemical “tail” that subtly tunes the shade and intensity of the final color. Beyond simply coloring fabric, these molecules were also intended to act as tiny built‑in UV shields.
Greener Chemistry Through Microwaves
To make the dyes, the team compared two heating methods: conventional oil‑bath reflux, which can take many hours, and modern microwave heating, which delivers energy directly and efficiently to the reaction mixture. In both cases, the same starting building blocks were combined in a one‑pot, multi‑component process, meaning several ingredients react together in a single step to form the complex dye structure. Under traditional heating, reactions needed 4 to 10 hours. With microwave irradiation, the same dyes formed in just 1 to 4 minutes, often in higher yield. Because the reactions are faster, solvent‑free, and more energy‑efficient, the microwave route aligns well with the goals of green chemistry.
Coloring Wool While Saving Energy
The new dyes were then used to color merino wool fabric under a range of practical conditions, varying dye amount, bath acidity (pH), temperature, and time. Measurements of color strength showed that several of the dyes produced deep, uniform shades, especially in mildly acidic baths where wool’s amino groups are positively charged and can attract the negatively charged sulfonate groups on the dye. One dye, containing a nitro‑substituted ring, gave particularly strong coloration at 3% shade, pH 4, and 90 °C. For more energy‑conscious processing, slightly lower temperatures such as 80 °C still produced nearly comparable color depth, suggesting that mills could reduce energy use without sacrificing appearance. Statistical analysis confirmed that both dye structure and bath pH strongly affect how much color the wool takes up.
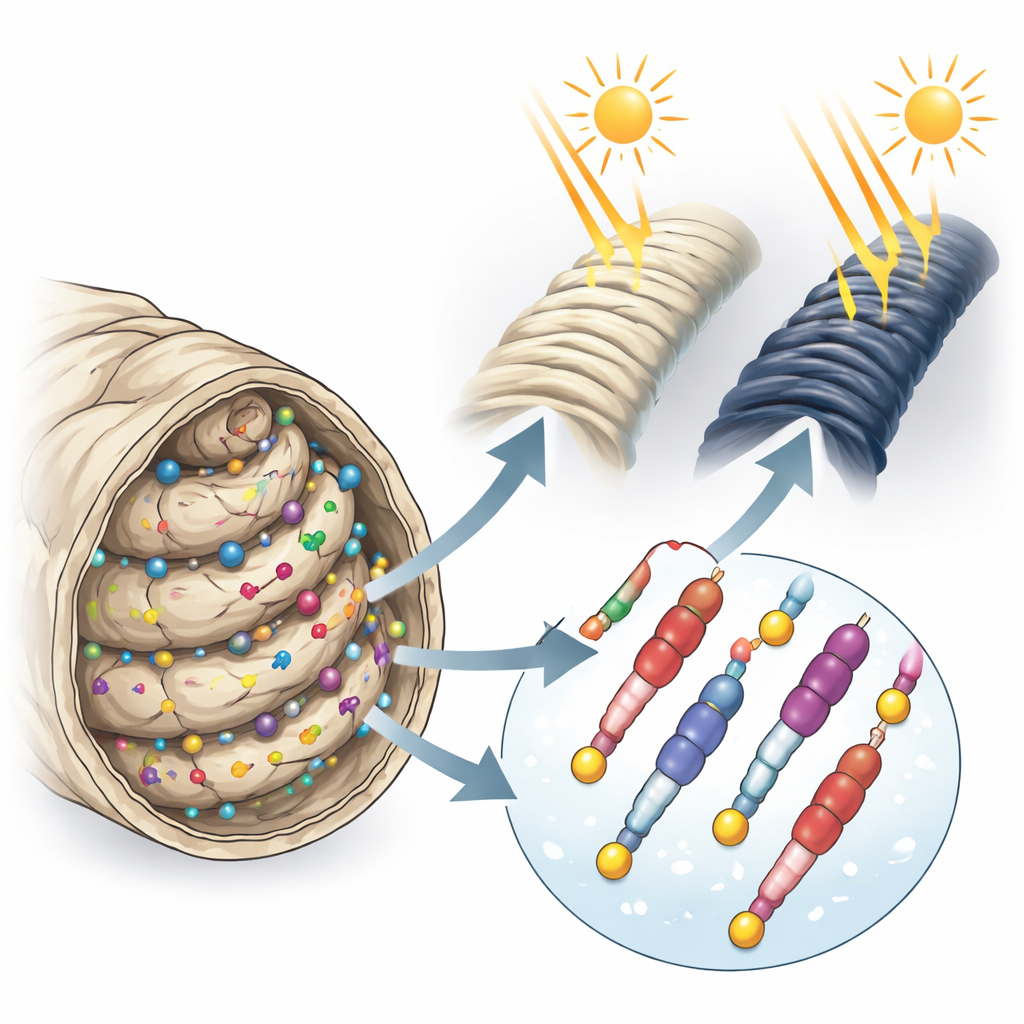
Built‑In Sun Shield Without Harming the Fiber
Beyond color, the team evaluated how the dyes interact with wool at the molecular level and how they change fabric performance. Tests of urea‑bisulfite solubility and base‑combining capacity showed that dyeing did not create new crosslinks within the wool’s protein chains, meaning the dyes attach mainly through salt‑like attractions rather than permanent covalent bonds. Mechanical tests confirmed only a very small loss in tensile strength and elongation, so the fabric’s handle and durability remain largely intact. In contrast to these minimal structural changes, the boost in UV protection was dramatic: the ultraviolet protection factor (UPF) of undyed wool (already decent) rose to very high levels when dyed, reaching values over 500 for some shades. Darker, more intensely colored samples tended to provide the greatest shielding. At the same time, the dyed fabrics showed good resistance to washing, rubbing, perspiration, and light under standard textile tests.
What This Means for Everyday Textiles
Taken together, the work shows that carefully engineered pyrazolopyrimidine dyes can be synthesized rapidly by a greener microwave process and then used to produce richly colored wool fabrics with excellent built‑in UV protection. Because the new dyes bind through reversible ionic interactions, they do not damage the wool’s protein structure and preserve its mechanical integrity. These features make the dyes promising candidates for eco‑conscious, functional wool garments and wool/polyester blends that keep wearers both warm and better protected from the sun.
Citation: Anwer, K.E., Abou-Taleb, M., Sayed, G.H. et al. Design, ecofriendly synthesis and spectroscopy of new pyrazolopyrimidine derivatives as potential candidates for dyeing and ultraviolet protection of wool fabrics. Sci Rep 16, 9803 (2026). https://doi.org/10.1038/s41598-026-38297-x
Keywords: eco-friendly dyeing, wool fabrics, ultraviolet protection, microwave synthesis, functional textiles