Clear Sky Science · en
Synthesis and biological evaluation of ibuprofen/o-vanillin Schiff base complexes with anti-inflammatory, anti-proliferative and anti-SARS-COV-19 activities
Why upgrading a familiar painkiller matters
Ibuprofen is one of the world’s most widely used pain and fever medicines, but long-term use can irritate the stomach and it was never designed to fight infections, cancer, or new viruses. In this study, chemists re-engineer ibuprofen by fastening it to a natural vanilla-like molecule and then attaching different metals. The result is a family of new compounds that act not just as pain relievers, but as potential antibiotics, anticancer agents, anti‑inflammatory drugs, and even blockers of a key SARS‑CoV‑2 enzyme, all while offering clues about how to design smarter medicines from everyday drugs.
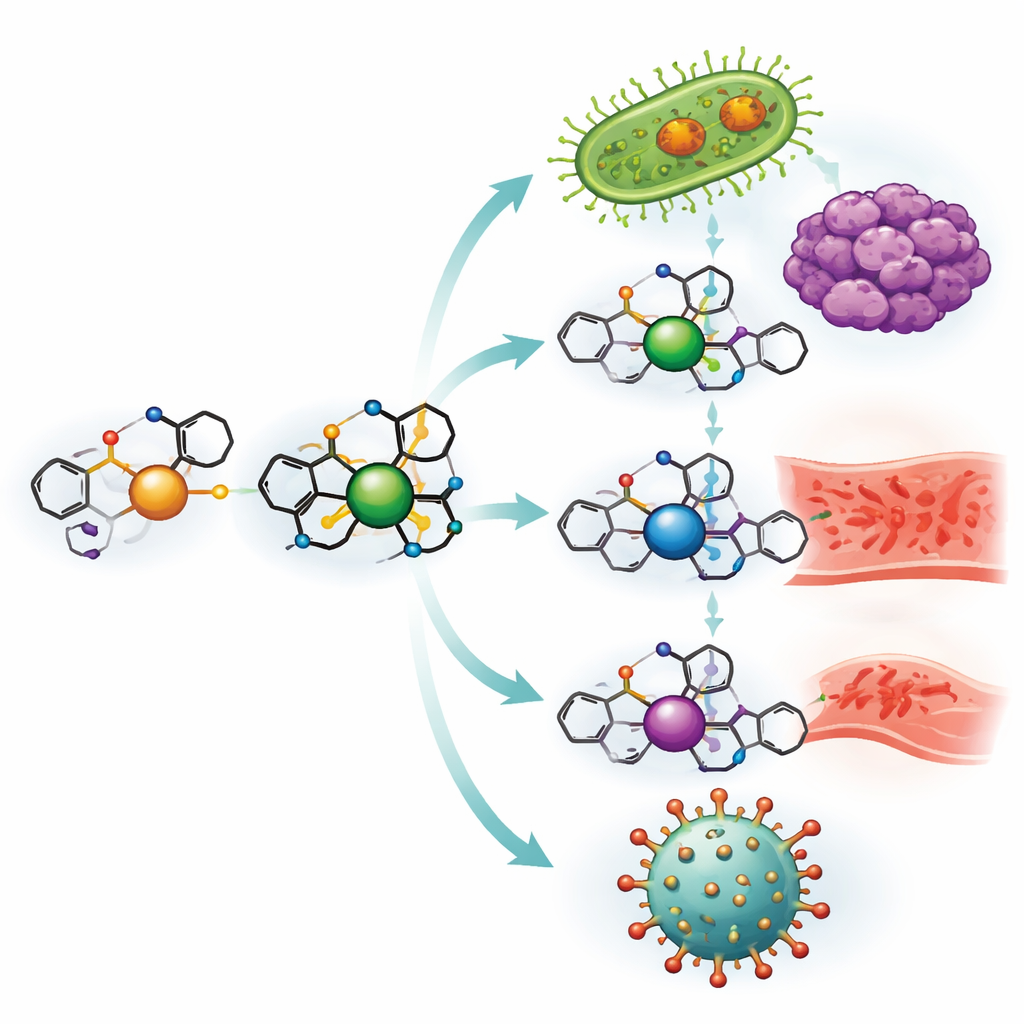
Building new drug-like molecules from ibuprofen
The team began by transforming ibuprofen into a more versatile building block and then linking it to o‑vanillin, a close chemical cousin of vanilla’s flavor compound. Together they form a “Schiff base” ligand—essentially a tailored claw that can grip metal ions. This ligand was then combined separately with copper, nickel, zinc, and vanadium salts to produce four distinct metal complexes. Careful laboratory tests, including measurements of melting behavior, light absorption, magnetic properties, and crystal structure, confirmed how the ligand wraps around each metal and how stable the resulting complexes are, even at high temperatures and across a wide range of acidity.
How the complexes interact with cells and DNA
Because a drug’s power often depends on how it meets biological targets, the researchers probed how these new complexes interact with DNA and living cells. In test-tube experiments, the copper complex in particular bound tightly to strands of DNA and was able to damage or “cleave” them, a behavior often seen in potent anticancer and antibacterial agents. When tested against human cancer cell lines from breast, colon, and liver tumors, all four complexes were more toxic to cancer cells than the ibuprofen-based ligand alone, with the copper complex approaching the strength of the chemotherapy drug vinblastine at similar doses.
Fighting germs, fungi, and inflammation
The same compounds were challenged with common disease-causing microbes, including Gram-positive and Gram-negative bacteria and two problematic fungi. Again, the metal complexes outperformed the free ligand, and copper led the pack, strongly inhibiting both bacteria and fungi. The authors interpret this through “chelation” theory: once the metal is held by the ligand, its charge is partly shielded and the whole complex becomes more fat-loving, allowing it to slip into microbial membranes and disrupt vital enzymes. In a simple test of inflammation based on heat‑damaged egg protein, the new complexes also reduced protein denaturation more effectively than ibuprofen itself, suggesting they might calm inflammatory processes at lower doses.
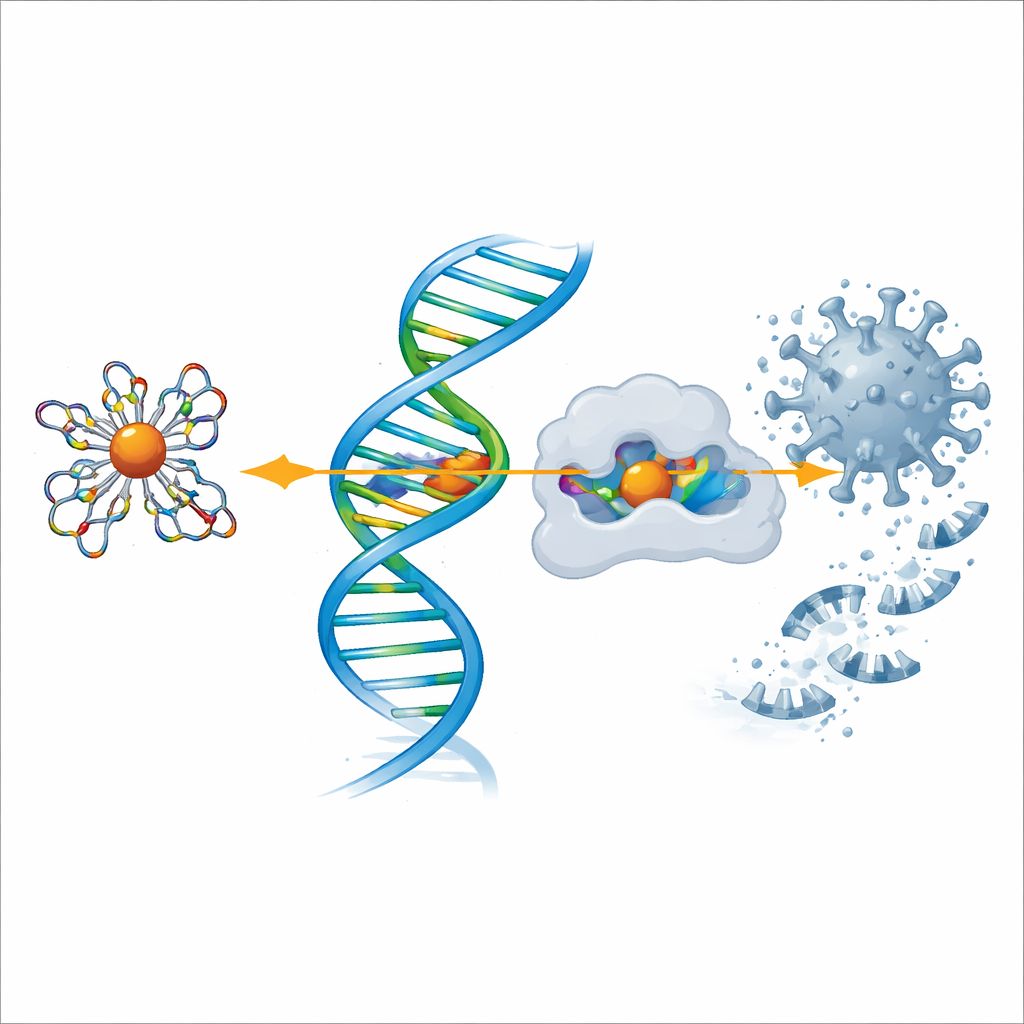
Probing action against COVID‑19 and pain enzymes
To explore antiviral potential, the team used computer modeling to dock the ligand and each metal complex into the three‑dimensional shapes of two key proteins: the main protease of SARS‑CoV‑2, which the virus uses to cut its polyproteins, and cyclooxygenase‑2 (COX‑2), an enzyme involved in pain and inflammation. The simulations showed that the complexes, especially those of copper and zinc, fit snugly into the active pockets of these proteins and formed multiple stabilizing interactions. The more negative the calculated binding energy, the stronger the predicted binding, and again the copper complex stood out, followed closely by zinc and nickel, supporting the laboratory findings on anti‑inflammatory and antimicrobial behavior.
What this could mean for future medicines
Put together, the work shows that a familiar over‑the‑counter drug can be turned into a multi‑purpose chemical tool by fusing it to a carefully chosen organic partner and the right metal ion. Among the four metals tested, copper produced the most promising candidate, with strong antibacterial, antifungal, anticancer, anti‑inflammatory, and computationally predicted anti‑SARS‑CoV‑2 activities, while nickel and zinc offered slightly milder but still notable effects. Although these results are still at the laboratory and computer‑model stage, they point toward a strategy in which everyday medicines are “upgraded” into targeted metal complexes that may work at lower doses and on a broader range of diseases than the original drugs.
Citation: Abdel-Rahman, L.H., El-ezz, D.A., Abdel-Mawgoud, AM.M. et al. Synthesis and biological evaluation of ibuprofen/o-vanillin Schiff base complexes with anti-inflammatory, anti-proliferative and anti-SARS-COV-19 activities. Sci Rep 16, 8501 (2026). https://doi.org/10.1038/s41598-026-38270-8
Keywords: ibuprofen derivatives, metal drug complexes, anticancer agents, antimicrobial therapy, COVID-19 protease