Clear Sky Science · en
Clinically interpretable nomogram combining body composition and clinicopathological features for one year survival prediction in advanced solid tumors
Why your body makeup may shape cancer treatment success
Immunotherapy drugs have changed the outlook for many people with advanced cancers, yet only a portion of patients truly benefit from them. This study explores a simple but powerful idea: that the way fat and muscle are distributed in the body, together with routine clinical information, can help doctors estimate a patient’s chances of being alive one year after starting immunotherapy. Turning complex measurements into an easy-to-use bedside tool, the researchers propose a visual scorecard that could guide conversations about treatment choices and follow-up care.
Looking beyond the tumor to the whole person
Most current tests used to predict who will respond to immune checkpoint inhibitors focus on properties of the tumor itself, such as specific proteins on cancer cells or how many mutations the cancer carries. But immune responses are also shaped by broader factors, including other illnesses, medications, and overall nutrition. Body mass index (BMI) has been widely studied and sometimes appears to be linked to better outcomes in heavier patients, a puzzle often called the “obesity paradox.” However, BMI is a crude measure: it cannot distinguish between fat and muscle, or between different types of fat, which may have very different effects on the immune system.
Turning scans into clues about survival
The researchers examined 146 adults with advanced solid tumors treated with immune checkpoint inhibitors at a single Italian center. Before treatment, each patient had a routine CT scan used to stage the cancer. From a single slice through the abdomen, radiologists measured how much muscle and fat lay in different compartments, such as deep abdominal fat around the organs and fat under the skin. They also collected standard clinical information, including cancer type, treatment details, basic blood tests related to nutrition and inflammation, and common conditions like diabetes, high blood pressure, and heart disease.
Building a combined risk score
Using machine-learning methods designed for survival data, the team first tested how well usual clinical factors alone could forecast overall survival. These features—such as cancer type, treatment regimen, smoking history, a blood-based inflammation ratio, and a nutrition score—showed some predictive power but struggled to generalize to new patients. Next, they evaluated each body composition measure on its own. The ratio between deep abdominal fat and under-the-skin fat emerged as the single best imaging-based predictor, yet it was still not strong enough by itself. The researchers then created new composite scores that mathematically combined several fat and muscle measurements, searching for the combination that best separated patients with longer and shorter survival.
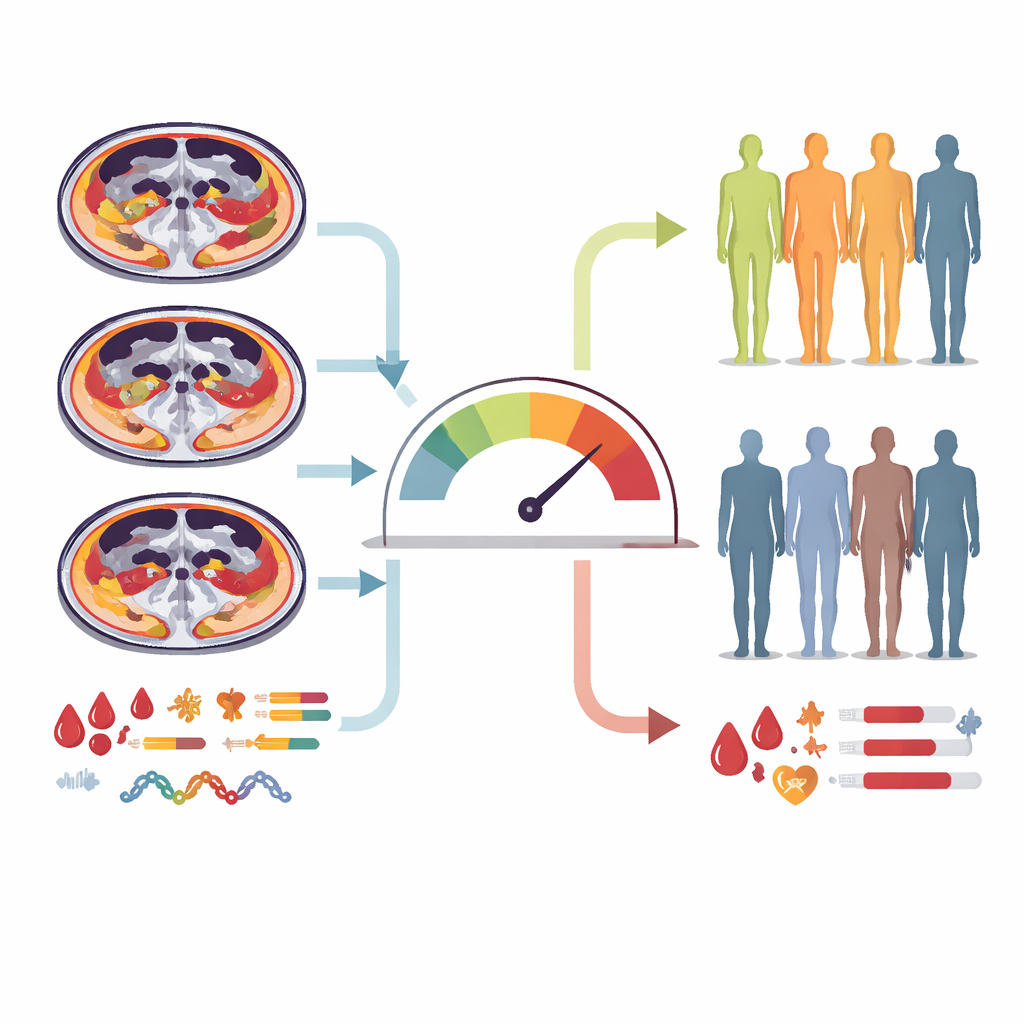
A simple chart to sort patients into risk groups
The most informative combination turned out to be a body composition score built from three CT-derived measurements: fat inside muscles, the amount of deep abdominal fat, and the ratio of deep to subcutaneous fat. When this score was added to 12 routinely collected clinical features, the resulting model clearly outperformed models based only on clinical data or imaging alone. Patients whose body composition score exceeded an optimized cutoff had a median survival of about five months, compared with nearly three years for those below the cutoff. This strong difference held up even when focusing only on people with advanced lung cancer. From this model, the authors constructed a nomogram—a visual chart where a clinician can align a patient’s values for each factor, add up points, and read off the estimated chance of being alive at one year.
What this means for patients and doctors
For people facing advanced cancer, this work suggests that information already hidden in everyday scans and blood tests can be combined to give a clearer picture of likely outcomes with immunotherapy, without needing specialized genetic testing. The proposed chart is not yet ready to change practice on its own: the study is retrospective, comes from a single center, and involves a modest number of patients, so its predictions need to be confirmed in larger, independent groups. Still, it highlights how a patient’s overall body makeup and health—rather than the tumor alone—may crucially influence treatment results, and it points toward more personalized decisions about who might gain the most from powerful but costly and sometimes toxic immunotherapies.
Citation: Bruschi, G., Paoloni, F., Pecci, F. et al. Clinically interpretable nomogram combining body composition and clinicopathological features for one year survival prediction in advanced solid tumors. Sci Rep 16, 13200 (2026). https://doi.org/10.1038/s41598-026-37510-1
Keywords: immunotherapy, body composition, cancer survival, risk prediction, machine learning