Clear Sky Science · en
Impacts of variable operating conditions on flux and energy efficiency of air gap membrane distillation for brine management
Turning Salty Wastewater into a Resource
Modern desalination plants are solving water shortages but creating a new problem: huge volumes of leftover super-salty wastewater, or brine, that are hard to dispose of without harming the environment. This study explores a promising way to squeeze more fresh water out of that brine using a technique called air gap membrane distillation, while keeping an eye on how much energy it uses and how quickly the equipment wears out. The work shows where the sweet spot lies between getting a lot of clean water, keeping energy costs in check, and protecting the filter material inside the system.
How the Heat-Driven Filter Works
Air gap membrane distillation can be thought of as a gentle, guided evaporation process. Warm salty water flows on one side of a thin, water-repelling sheet, while cold water flows on the other side, separated by a small pocket of air. Because of the temperature difference, water molecules evaporate from the hot salt solution, pass through the membrane and air gap as vapor, and then condense as nearly pure water on the cold side. Most of the salt stays behind in the original stream, turning it into a more concentrated brine. This setup can handle salt levels that are too high for many common desalination methods, making it attractive for treating waste brine from existing plants.
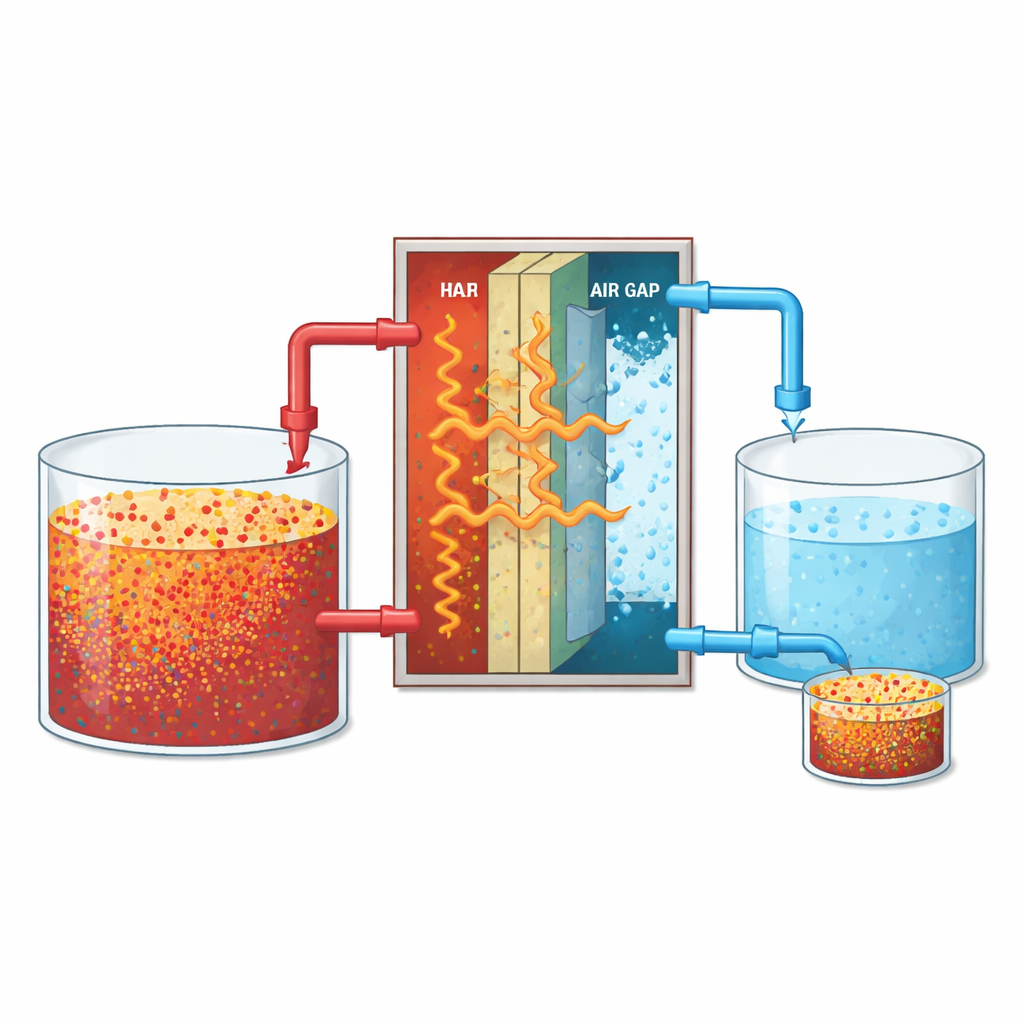
Testing Different Speeds and Salt Levels
To see how best to run this process, the researchers systematically varied two simple knobs an operator can control: how salty the incoming brine is and how fast it flows through the unit. They tested three salinity levels typical of concentrated brine (45, 55, and 65 grams of salt per liter) and flow rates from a slow trickle to a brisk stream. For each condition they measured how much clean water came through the membrane (flux), how effectively the salt was held back (salt rejection), and how much heat energy was needed per volume of fresh water produced. At the same time, they limited each run to six hours so they could study the early stages of buildup and clogging on the membrane surface without confusing them with long-term damage.
Finding the Best Working Point
The results reveal a familiar engineering trade-off. Pushing the brine faster generally boosted the rate of fresh water production, because the hot liquid near the membrane stayed well mixed and could keep evaporating. But this improvement came at a cost: at the highest flow and salt levels, more salt slipped through and the purity of the product water dropped. Energy use also shifted in a non-intuitive way. At very low flow, energy per liter was high because not much water was produced. As flow increased, energy use per liter dropped to a minimum and then began to rise again as pumping demands and other losses grew. The most balanced operation emerged at a moderate flow of about 2.0 liters per minute and salinity up to 55 grams per liter, where the system delivered a healthy water yield, removed more than 98 percent of the salt, and kept heat demand in a more reasonable range for this small-scale setup.

What Happens to the Filter Surface
Beyond numbers on a chart, the team wanted to know what was physically happening to the membrane material under these conditions. Using electron microscopy, they compared unused filters with ones that had run for 72 hours. The fresh material showed a neat web of pores, while the used samples displayed twisted pathways and tiny salt crystals lodged in the openings, clear signs of fouling and partial blockage. A separate chemical fingerprinting technique confirmed that new compounds and deposits had formed on the surface. These changes help explain why extremely high flows and salinities eventually hurt performance: as crystals accumulate and pores begin to wet, it becomes easier for salty water to creep through, lowering the purity of the output.
Why This Matters for Future Desalination
Taken together, the study shows that air gap membrane distillation can be tuned to turn hard-to-handle brine into additional clean water, but only if it is run within a careful window of conditions. Too gentle, and the process wastes energy; too aggressive, and the filters foul and let more salt slip into the product water. The authors argue that operating at moderate flow with mid-range salinity offers a practical compromise today, while future systems could use smarter designs, heat recovery, and digital monitoring to push performance further. For non-specialists, the key message is that there is still untapped fresh water hidden in the waste streams of today’s desalination plants, and with thoughtful engineering, it can be recovered in a way that is both efficient and kinder to the environment.
Citation: Mohamed, E.S., Azzam, A.M., Mohamed, A.T. et al. Impacts of variable operating conditions on flux and energy efficiency of air gap membrane distillation for brine management. Sci Rep 16, 12028 (2026). https://doi.org/10.1038/s41598-026-36621-z
Keywords: brine desalination, membrane distillation, water treatment, energy efficiency, fouling