Clear Sky Science · en
Quinazolinone derivatives as suitable mitigator for corrosion inhibition of carbon steel in hydrochloric acid environment
Why Protecting Everyday Metals Matters
From car bodies and storage tanks to oil pipelines and heat exchangers, many of the metal structures that keep modern life running are made from inexpensive, strong carbon steel. Yet when this steel meets harsh acids used for cleaning and processing, it can corrode rapidly, weakening equipment and driving up replacement and maintenance costs. This study explores two newly designed organic molecules that act like microscopic shields, forming a protective film on steel surfaces to dramatically slow down this type of damage.
How Acid Eats Away at Steel
Corrosion is essentially metal slowly returning to its original, ore-like state. In acidic solutions such as hydrochloric acid, this happens through tiny electrical reactions on the steel surface: some spots give up metal atoms, while others help form hydrogen gas. Industrial processes often rely on strong acids to strip off rust and scale, but that same acid also attacks the steel underneath. Left unchecked, this leads to thinning walls, leaks, and even catastrophic failures in industrial plants, with serious safety and economic consequences.
New Molecules That Act Like a Protective Skin
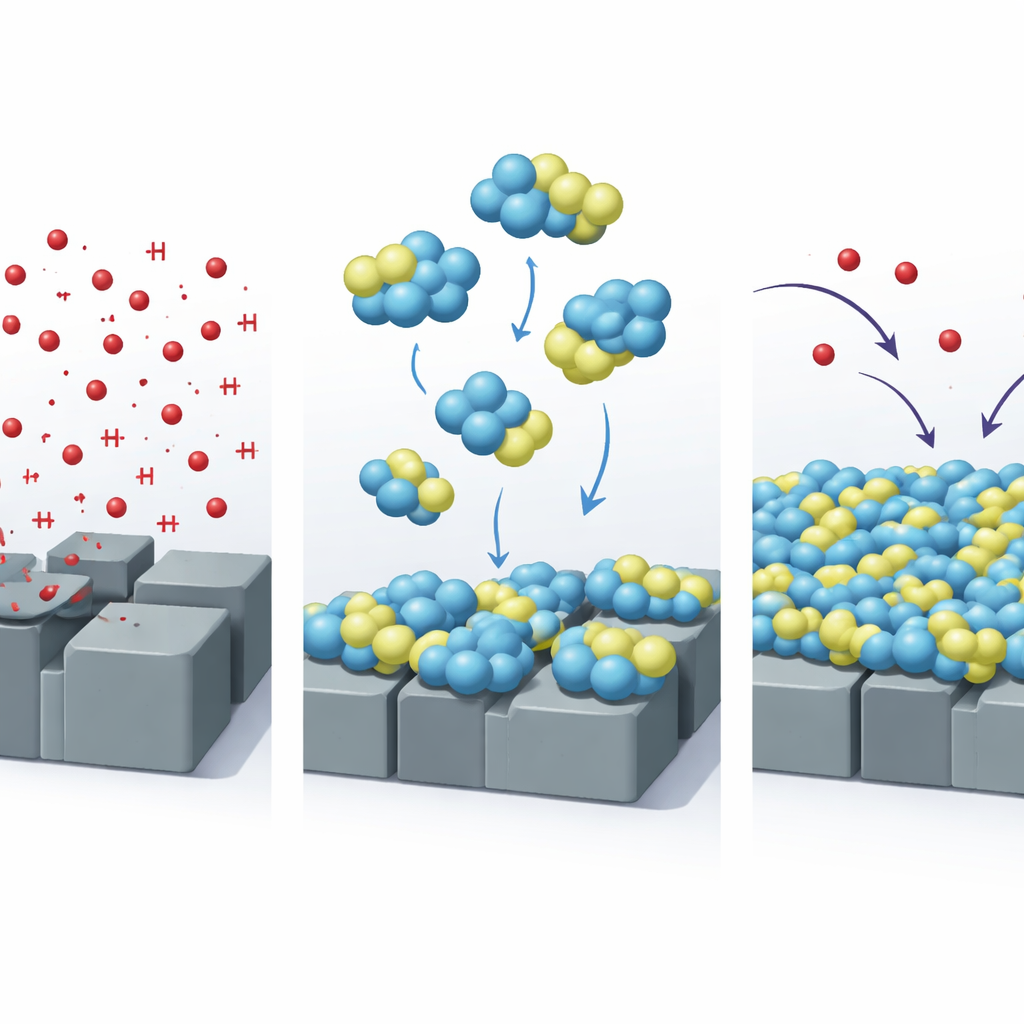
The researchers focused on two related organic compounds, called 4-OPB and 4-HPB, based on a chemical framework known as quinazolinone. These molecules contain several atoms such as nitrogen, oxygen, and sulfur that can cling strongly to iron in steel. When added in very small amounts to hydrochloric acid, they move from the liquid onto the metal surface and spread out into a thin, tightly packed layer. Weight-loss tests, where steel samples are weighed before and after immersion, showed that these compounds can cut the amount of metal lost by more than 90 percent at room temperature when used at the highest tested concentration.
Seeing the Shield in Action
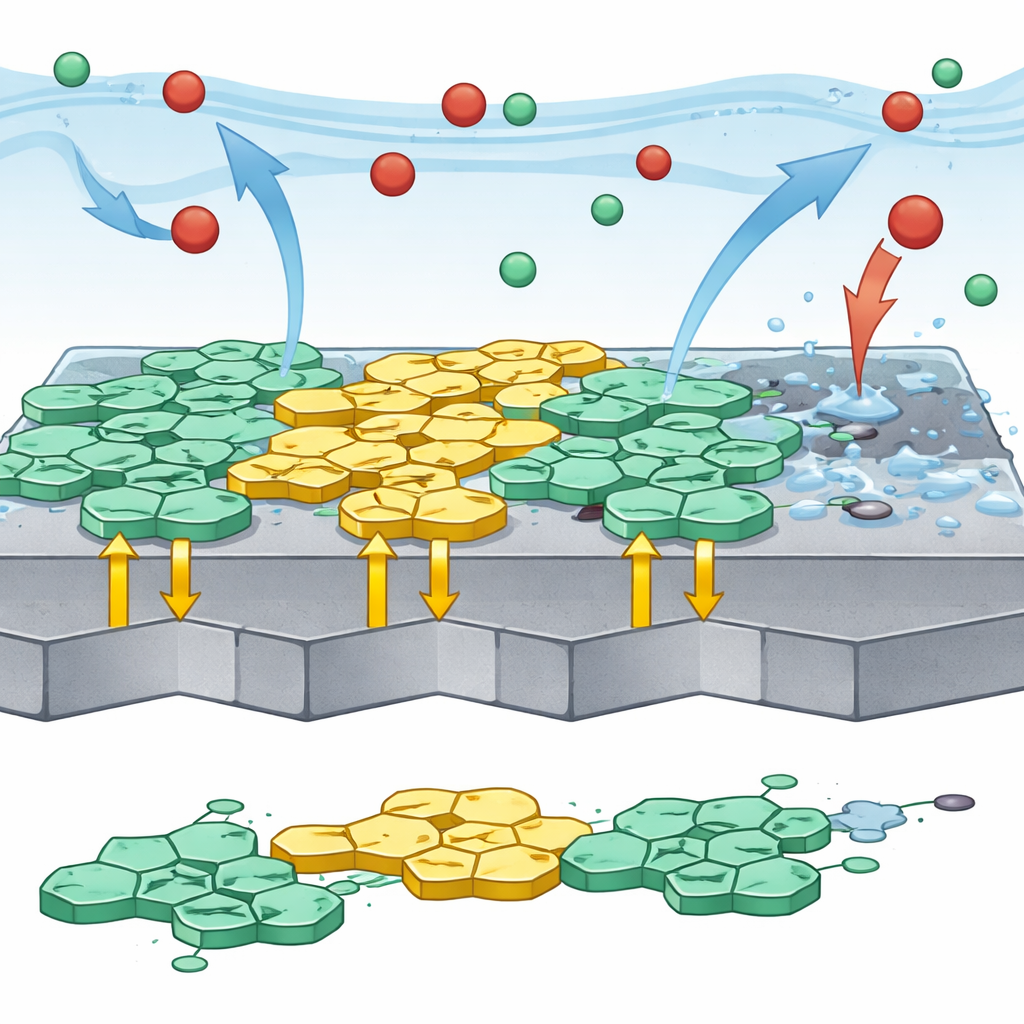
To understand how well this microscopic armor works, the team used electrochemical techniques that track how easily electrical currents associated with corrosion flow at the steel surface. Both 4-OPB and 4-HPB greatly reduced these currents, confirming that they hinder both the metal-dissolving and hydrogen-forming reactions. Imaging methods such as scanning electron microscopy and atomic force microscopy gave a visual before-and-after comparison: steel kept in bare acid appeared rough, cracked, and heavily pitted, while steel protected by the inhibitors looked far smoother, with fewer defects. Chemical analysis of the surface showed signals from elements in the inhibitor molecules, further evidence that a protective film had formed.
What Makes These Shields Stick So Well
Beyond experiments, the scientists turned to computer simulations to probe how the molecules interact with steel at the atomic level. Quantum chemical calculations suggested that key parts of the molecules can donate electrons to the metal, while also accepting some electron density back, creating a strong, chemically bonded layer rather than just a weak physical coating. The way the molecules lie flat and cover the surface, predicted by Monte Carlo simulations, matches the high protection seen in the lab. One of the molecules, 4-HPB, has an extra hydroxyl group that increases its electron density, helping it bind even more strongly and making it slightly more effective than 4-OPB.
What This Means for Real-World Equipment
The study shows that carefully tailored organic molecules can protect carbon steel from aggressive acids by forming a robust, self-assembled barrier only a few molecules thick. In practical terms, using tiny amounts of 4-OPB or 4-HPB could extend the lifetime of acid-washed steel equipment, reduce unplanned shutdowns, and lower costs. Because these compounds work mainly by strong chemical attachment to the steel surface and follow a well-understood adsorption pattern, they also offer a blueprint for designing next-generation, more environmentally friendly corrosion inhibitors that are both efficient and easier to integrate into existing industrial processes.
Citation: Al-Surmi, A.A., Shaaban, M.S., El-Mekabaty, A. et al. Quinazolinone derivatives as suitable mitigator for corrosion inhibition of carbon steel in hydrochloric acid environment. Sci Rep 16, 14152 (2026). https://doi.org/10.1038/s41598-025-33549-8
Keywords: carbon steel corrosion, acid corrosion inhibitors, protective molecular films, quinazolinone compounds, industrial metal protection