Clear Sky Science · en
Magnetic BiFeO3 nanoparticles as a sustainable and efficient catalyst for the green synthesis of highly substituted imidazole derivatives
Cleaner Paths to Important Medicines
Chemistry quietly underpins many of the medicines, crop protectants, and materials we rely on every day. But making these complex molecules can generate a lot of waste and use harsh solvents. This paper explores a smarter way to build an important class of drug-like molecules, called imidazoles, using tiny magnetic particles as a reusable, low-waste helper. The work shows how a carefully designed nanomaterial can make reactions faster and cleaner, while being easy to pull out of the mixture with a simple magnet.
Why These Ring-Shaped Molecules Matter
Imidazoles are small ring-shaped molecules that turn up everywhere: in natural substances in our bodies and in many medicines on the pharmacy shelf. Members of this family help fight fungi, bacteria, parasites, and inflammation, and they also appear in drugs for stomach acid and other conditions. Because these rings can grip metals and form hydrogen bonds, chemists use them as versatile building blocks when designing new therapies and functional materials. Finding ways to make imidazoles quickly, cheaply, and with less environmental impact is therefore a high priority.
One-Pot Cooking for Molecules
The researchers focus on a streamlined recipe called a three-component reaction. Instead of several separate steps, three simple starting ingredients are brought together in a single small flask: a two-carbon unit with two oxygen atoms, an aromatic aldehyde related to benzaldehyde, and a source of ammonia. When mixed under the right conditions, these pieces assemble into a highly substituted imidazole ring, decorated with multiple carbon-based groups that can tune biological activity. Doing this in one pot saves time and materials and fits well with the ideas of green chemistry, which aims to minimize waste and avoid unnecessary purification steps.
Tiny Magnetic Helpers at Work
The heart of the study is a new solid catalyst based on bismuth ferrite, a compound of bismuth, iron, and oxygen engineered into nanoparticles. The team prepared these particles by heating metal nitrates with glycine, producing a porous, rod-like structure with a high surface area and strong magnetic response. Detailed imaging and measurements confirmed their composition, crystal structure, porosity, and magnetism. When just a few milligrams of this powder were added to the three-component mixture and heated gently without any added solvent, the reaction proceeded rapidly, delivering a wide range of imidazole derivatives in high to excellent yield.
Fast Reactions, Less Waste, Easy Recovery
Systematic tests showed that without the catalyst, the reaction barely proceeded, while with bismuth ferrite nanoparticles under solvent-free conditions at about 80 °C, products formed in minutes. The method worked for many different aldehydes, including ones carrying electron-rich, electron-poor, and heteroaromatic groups, demonstrating broad scope. The magnetic nature of the particles is crucial in practice: after the reaction, the solid can be pulled out simply by holding a magnet to the flask, then washed, dried, and reused. The catalyst maintained its performance over at least five cycles with only a small drop in yield, highlighting its stability and potential for scale-up.
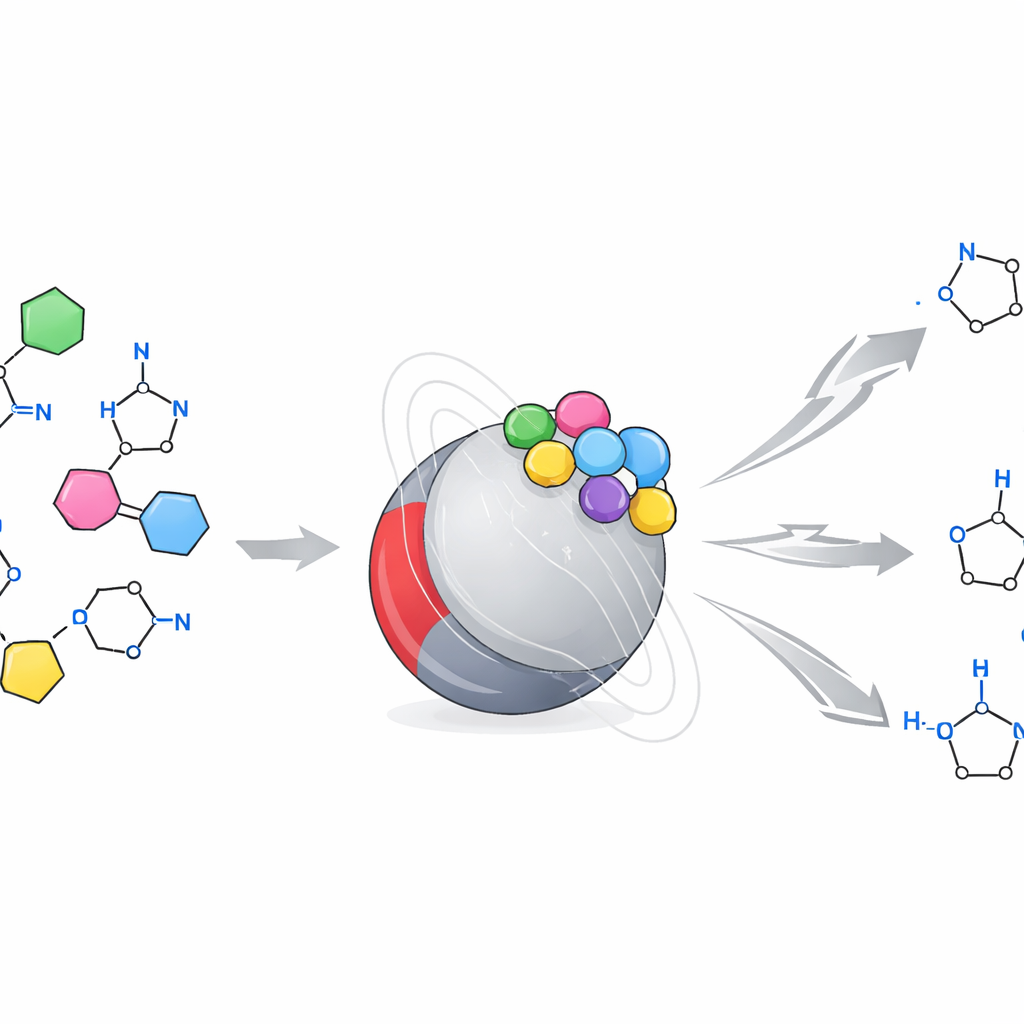
How the Surface Guides the Build
At the molecular level, the authors propose that positively charged sites containing bismuth and iron on the nanoparticle surface temporarily hold and “activate” the aldehyde and related intermediates. This activation makes it easier for ammonia and the carbonyl components to link up, lose water, and cyclize step by step into the imidazole ring. The porous structure provides many accessible sites, while surface hydroxyl groups assist with proton shuttling and removal of water formed during the reaction. Experiments in which the solid catalyst was removed mid-reaction showed that once the particles are gone, the reaction slows dramatically, supporting the idea that most of the work is done on the solid surface rather than by dissolved metal ions.
What This Means Going Forward
In plain terms, this study shows that a handful of specially designed magnetic nanoparticles can help snap together valuable imidazole rings quickly and cleanly, without the need for extra solvent or large amounts of catalyst. Because the particles can be fished out with a magnet and reused multiple times, they cut down on both cost and chemical waste. Compared with many existing methods, this approach offers shorter reaction times, high yields, and gentler conditions, pointing toward more sustainable manufacturing routes for imidazole-based medicines and related fine chemicals.
Citation: Hanifi, S., Dekamin, M.G. & Eslami, M. Magnetic BiFeO3 nanoparticles as a sustainable and efficient catalyst for the green synthesis of highly substituted imidazole derivatives. Sci Rep 16, 10535 (2026). https://doi.org/10.1038/s41598-025-32749-6
Keywords: green chemistry, nanocatalyst, imidazole synthesis, magnetic nanoparticles, heterocyclic drugs