Clear Sky Science · en
Experimental and computational evaluation of ZIF-67 based nanoadsorbents for efficient removal of acid red 37 from wastewater
Turning Dirty Water into a Cleaner Resource
Bright synthetic dyes make our clothes and fabrics appealing, but once they wash out of factories, they can turn rivers into long‑lasting chemical stews. This study tackles one of those stubborn dyes, Acid Red 37, by designing tiny, sponge‑like particles that can pull it out of water. Using both lab experiments and computer simulations, the researchers show how a new hybrid material can capture more dye, work quickly, and be reused many times, pointing toward more sustainable ways to clean industrial wastewater.
Why Colored Wastewater Is Hard to Clean
Industrial dyes are engineered to be vivid, stable, and resistant to fading—exactly the opposite of what we want once they end up in rivers and lakes. Acid Red 37, widely used in leather and textile processing, is not only eye‑catching but also potentially harmful to ecosystems and human health. Conventional treatment methods such as filtration, chemical oxidation, and sedimentation often struggle with these molecules, especially at low concentrations. Adsorption—where pollutants stick to the surface of a solid—has emerged as a promising approach, but traditional adsorbents can saturate quickly or be hard to regenerate. The challenge is to build a material that offers many accessible binding sites, selectively grabs dye molecules, and withstands repeated use.
Building a Smarter Dye Sponge
The team started with ZIF‑67, a metal‑organic framework made from cobalt ions and small organic linkers that arrange into a porous, crystal‑like network. This material already has a large internal surface area, making it a good starting point for adsorption. To boost its performance, the researchers created a composite by physically attaching very small copper and cobalt oxide particles to the ZIF‑67 structure, forming a hybrid called CuO/Co3O4NP@ZIF‑67, or CCZ. Microscopy and surface measurements confirmed that these oxide nanoparticles decorate the outer surfaces and partly fill the pores of ZIF‑67. Interestingly, even though this reduces the overall pore volume, it introduces new chemically active spots on the surface. X‑ray and spectroscopic analyses further showed mixed oxidation states of copper and cobalt, hinting at a redox‑active interface that can interact strongly with dye molecules.
How Well the New Material Grabs Dye
In batch tests, both ZIF‑67 and the CCZ composite were shaken with Acid Red 37 solutions while the researchers varied pH, contact time, dye concentration, and adsorbent dose. The composite consistently outperformed the original material, reaching a higher maximum dye uptake (about 66 milligrams of dye per gram of adsorbent, versus about 59 milligrams for ZIF‑67) and achieving up to 97% removal under optimized acidic conditions. Both materials captured most of the dye within tens of minutes, and mathematical fits to the time‑course data showed that a so‑called pseudo‑second‑order model best described the process, indicating strong affinity between dye and surface sites. Temperature‑dependent measurements revealed that adsorption is favorable and becomes more efficient at higher temperatures, with ZIF‑67 showing an intermediate energy profile and the composite behaving more like a classic physical sorbent. In practical terms, CCZ could be regenerated and reused for at least five cycles with only about a one‑percent drop in performance, an important feature for cost‑effective water treatment.
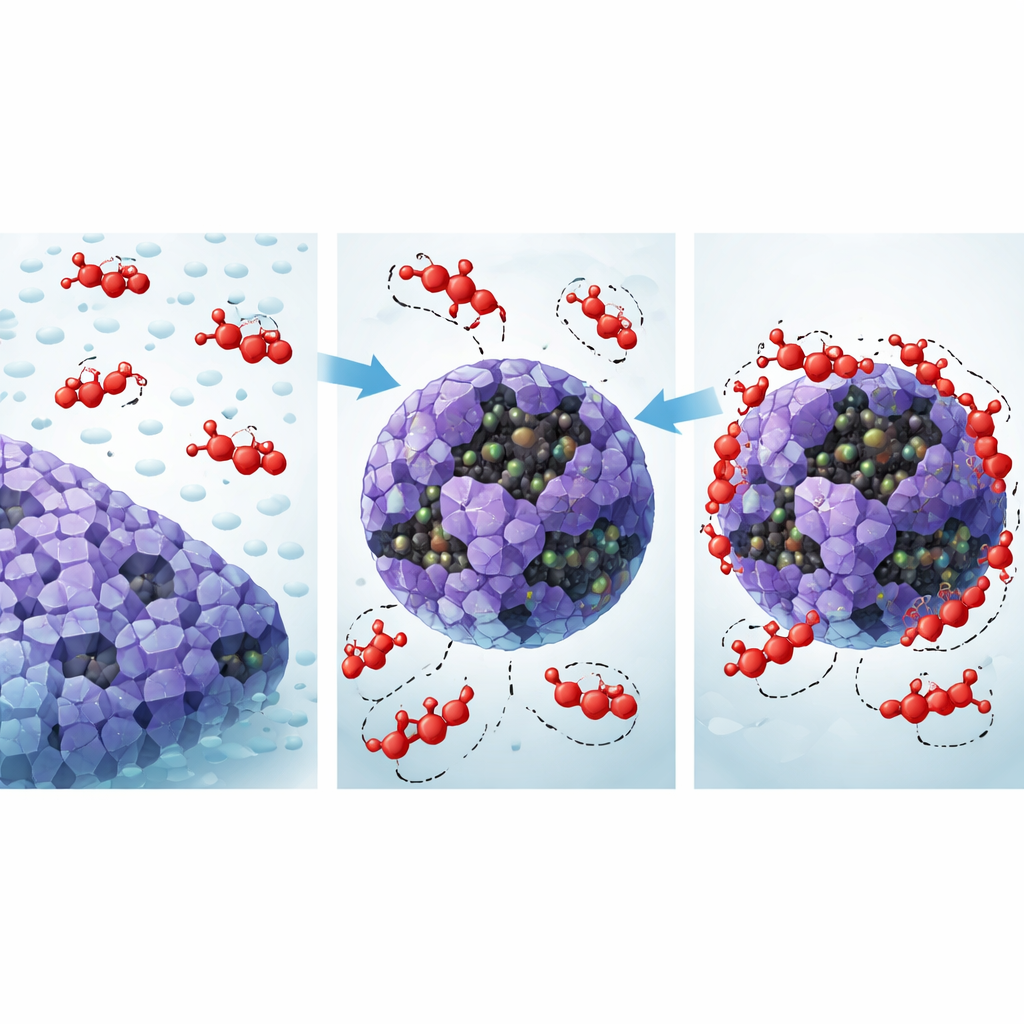
Peering into the Molecular Grip
To understand why the composite behaves so well, the authors turned to computer modeling. They optimized the structures of the dye, the ZIF‑67 framework, and the metal‑oxide layers, and then ran molecular dynamics simulations to see how the systems evolve over time in a virtual environment. These simulations confirmed that the composite remains structurally stable and that dye molecules settle into energetically favorable positions on its surface. Adsorption locator calculations, which search for the best binding arrangements, showed that Acid Red 37 anchors itself through a mix of hydrogen bonds, van der Waals contacts, and stacking interactions between its aromatic rings and those of the framework. The sulfonate and azo groups on the dye tend to cluster near oxygen‑rich and metal‑rich regions on the composite, exploiting the mixed copper and cobalt oxidation states as strong yet non‑permanent binding centers. These interactions explain how the composite can act like a powerful “physical” sponge while still benefiting from chemically tuned attraction.
What This Means for Cleaner Water
For non‑specialists, the main message is that the new CCZ material offers a smarter way to mop up a problematic red dye from water. By combining a porous framework with reactive metal‑oxide particles, the researchers created a hybrid adsorbent that soaks up more dye, works faster, shrugs off competing ions commonly found in wastewater, and can be reused multiple times with minimal loss of efficiency. The combined experimental and computational evidence shows that the dye is held firmly but not irreversibly through many small, cooperative interactions rather than a few strong chemical bonds. This concept of “chemically enhanced physisorption” could guide the design of future materials that are both highly effective and energy‑efficient, helping to keep industrial color out of our rivers and bring us closer to safer, cleaner water.
Citation: Ali, A.ES., El-Dissouky, A., Elbadawy, H.A. et al. Experimental and computational evaluation of ZIF-67 based nanoadsorbents for efficient removal of acid red 37 from wastewater. Sci Rep 16, 14396 (2026). https://doi.org/10.1038/s41598-025-32600-y
Keywords: wastewater treatment, dye removal, metal-organic frameworks, nanocomposite adsorbents, computational modeling