Clear Sky Science · en
Evaluating microbial community profiles of Chile peppers grown on the International Space Station provides implications for fruiting crops
Fresh Peppers in Space
As humans look toward living on the Moon and Mars, we will need more than packaged meals. Fresh fruits and vegetables can boost nutrition, morale, and long-term health for astronauts. This study follows an ambitious experiment: growing chile peppers to full maturity on the International Space Station (ISS) and carefully checking the tiny life forms—microbes—living on the plants, their roots, and the surrounding hardware to make sure the food is safe and to understand how plant–microbe relationships work in space.
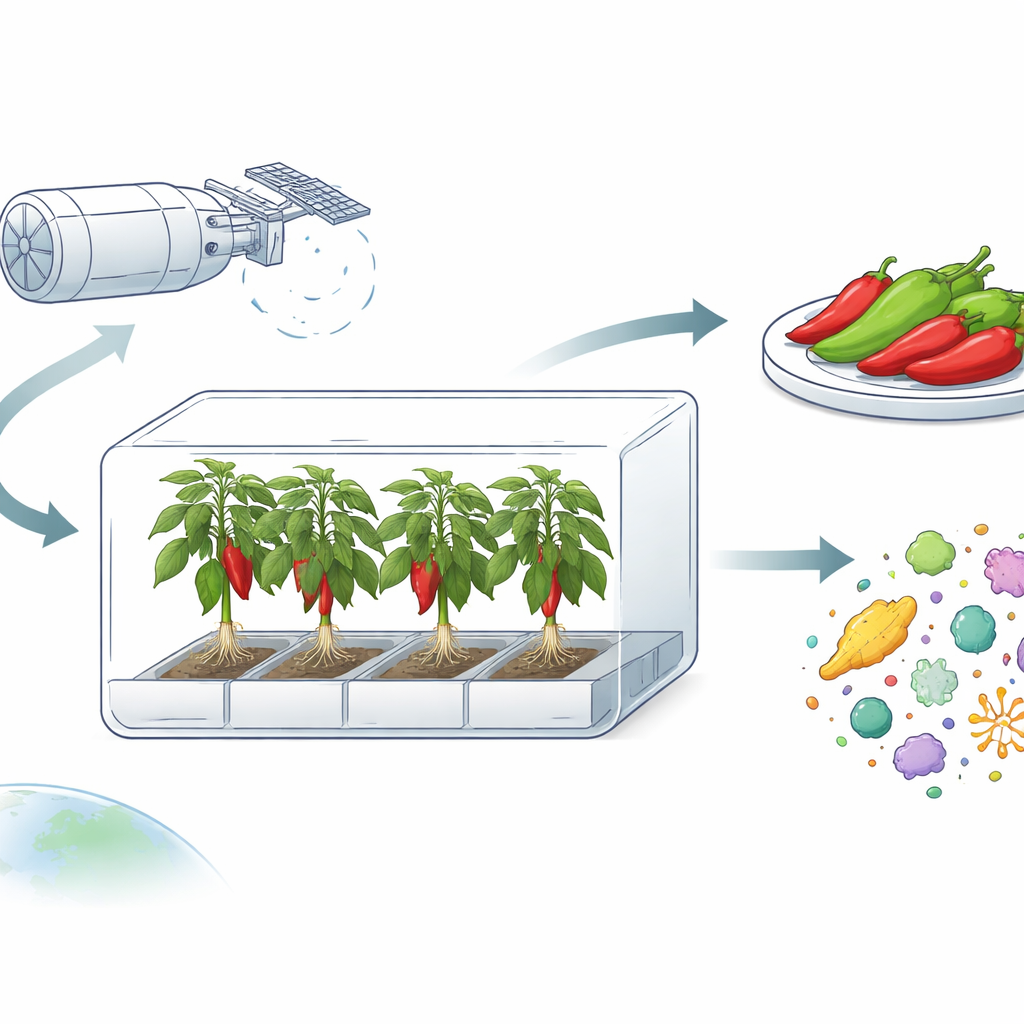
How Peppers Grow in Orbit
The peppers were grown inside the Advanced Plant Habitat, a tightly controlled, box‑shaped growth chamber on the ISS. This chamber lets scientists adjust light, temperature, humidity, carbon dioxide, and even airflow from Earth, while shielding the plants from the rest of the station’s environment. The team chose a compact Hatch chile variety that could fit the chamber yet still produce enough fruit to eat and to study. Seeds were carefully cleaned on the ground to strip away surface microbes, planted in sterile ceramic granules mixed with slow‑release fertilizer, and then launched to the station. Once installed in orbit, the peppers grew for 137 days—longer than any previous edible crop experiment on the ISS—producing 26 fruits, half eaten by the crew and half frozen for later analysis.
A Tiny World Around Roots and Leaves
Even after surface cleaning and sterile planting, plants inevitably gather microbes from their surroundings. On the ISS, these can come from cabin air, water lines, equipment surfaces, and the crew. To map this hidden community, the researchers sampled nearly every part of the system after the peppers were harvested. They swabbed surfaces and foam, collected the wicks that carried water, scooped up root‑zone granules, and dissected roots, stems, leaves, fruits, and seeds. They counted bacteria and fungi by growing them on nutrient gels and also read the genetic fingerprints of microbial communities by sequencing a commonly used “ID tag” in microbial DNA. This let them compare how many kinds of microbes were present and how similar or different the communities were across plant tissues and hardware.
Food Safety and Friendly Microbes
From a food safety standpoint, the news was reassuring. Microbial counts on the peppers were extremely low, sometimes below detectable levels, and no tested foodborne pathogens such as Salmonella, E. coli, or Staphylococcus aureus were found. Most of the microbial activity was concentrated in the hidden parts of the system: the moist wicks, the root zone, and the ceramic substrate, where nutrient‑rich conditions support more diverse life. Here, the researchers found a mix of bacterial groups, including several also known from the ISS drinking water system and air. Some of these, such as Sphingomonas and Novosphingobium, are often associated with plants on Earth and may help with growth or stress tolerance. Others, like Pseudomonas and Burkholderia, are more complicated: they can form stubborn films on surfaces or act as opportunistic pathogens under the wrong conditions, but they can also promote plant growth in some situations.
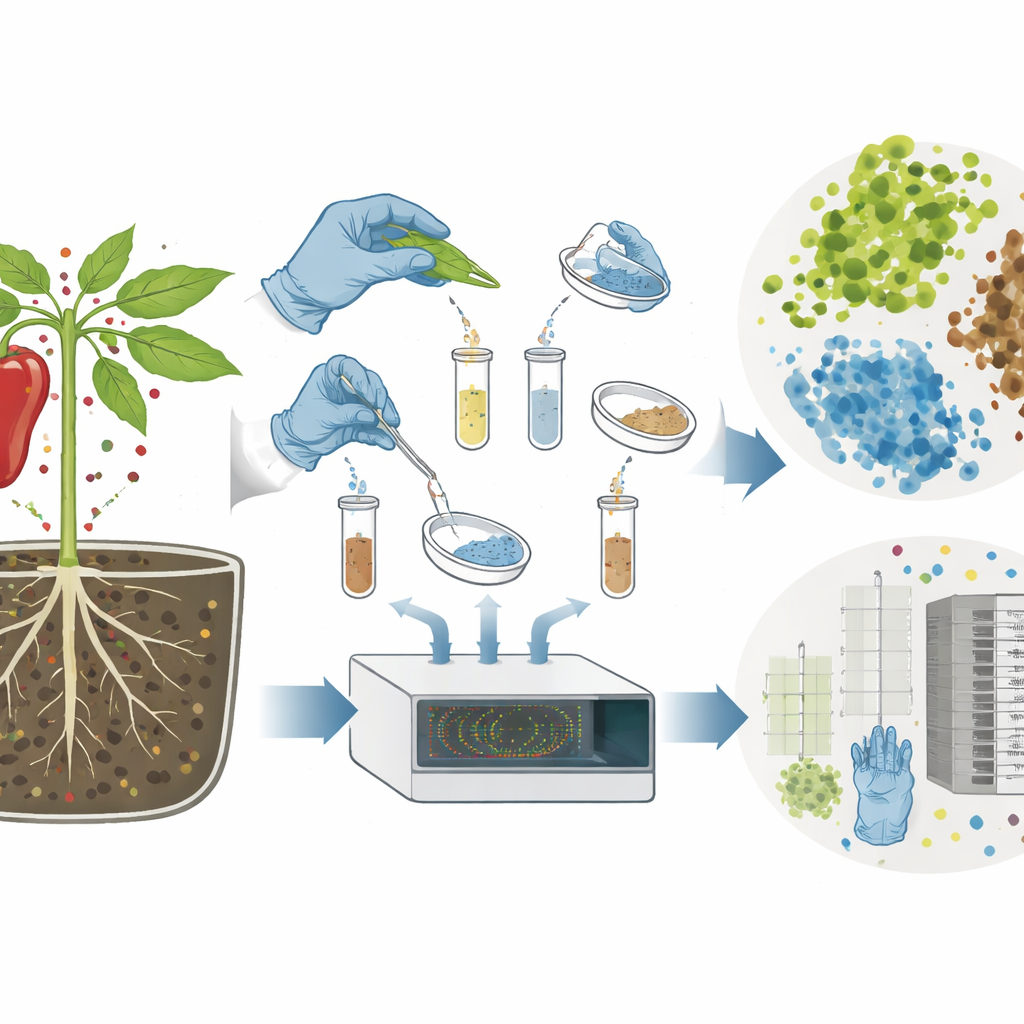
Patterns in the Invisible Garden
By comparing microbial DNA from above‑ground tissues (leaves, stems, fruits, seeds), below‑ground tissues (roots and the nearby granules), and the hardware pieces, the team found clear patterns. Above‑ground plant parts hosted relatively simple communities with fewer types of microbes. Below the surface, near the roots and in the substrate, microbial diversity roughly doubled, reflecting the richer and more stable environment around the root zone. The hardware pieces that touched water or the root zone showed their own characteristic mixes. Statistical tests suggested that where a sample came from—leaf versus root versus foam versus wick—mattered more than which quadrant of the tray it occupied. Yet a small set of bacterial groups appeared across seeds, plants, substrate, water, and hardware, forming a proposed “core microbiome” that persists throughout the mini‑ecosystem despite careful cleaning and physical separation.
What This Means for Future Space Farms
For non‑specialists, the key takeaway is twofold. First, it is possible to grow a long‑lived, fruiting crop like chile peppers in orbit and safely eat the harvest; the peppers met NASA’s microbial safety guidelines and added a welcome fresh, spicy food to the crew’s menu. Second, even in a tightly controlled, mostly closed system, plants develop complex microbial partners and hitchhikers, especially around their roots and in damp hardware. Understanding who these microbes are, where they come from, and how they behave will be essential for designing reliable space greenhouses that can support crews far from Earth. This study provides an important early map of that invisible ecosystem and a baseline for engineering healthier, more resilient crops in future space habitats.
Citation: Khodadad, C.L.M., Dixit, A.R., Hummerick, M.E. et al. Evaluating microbial community profiles of Chile peppers grown on the International Space Station provides implications for fruiting crops. Sci Rep 16, 12863 (2026). https://doi.org/10.1038/s41598-025-20440-9
Keywords: space agriculture, microbiome, International Space Station, chile peppers, food safety