Clear Sky Science · en
Assembling a chromosome-level genome for the Microtus fortis using PacBio HiFi and Hi-C technologies
A Small Mammal with Big Scientific Potential
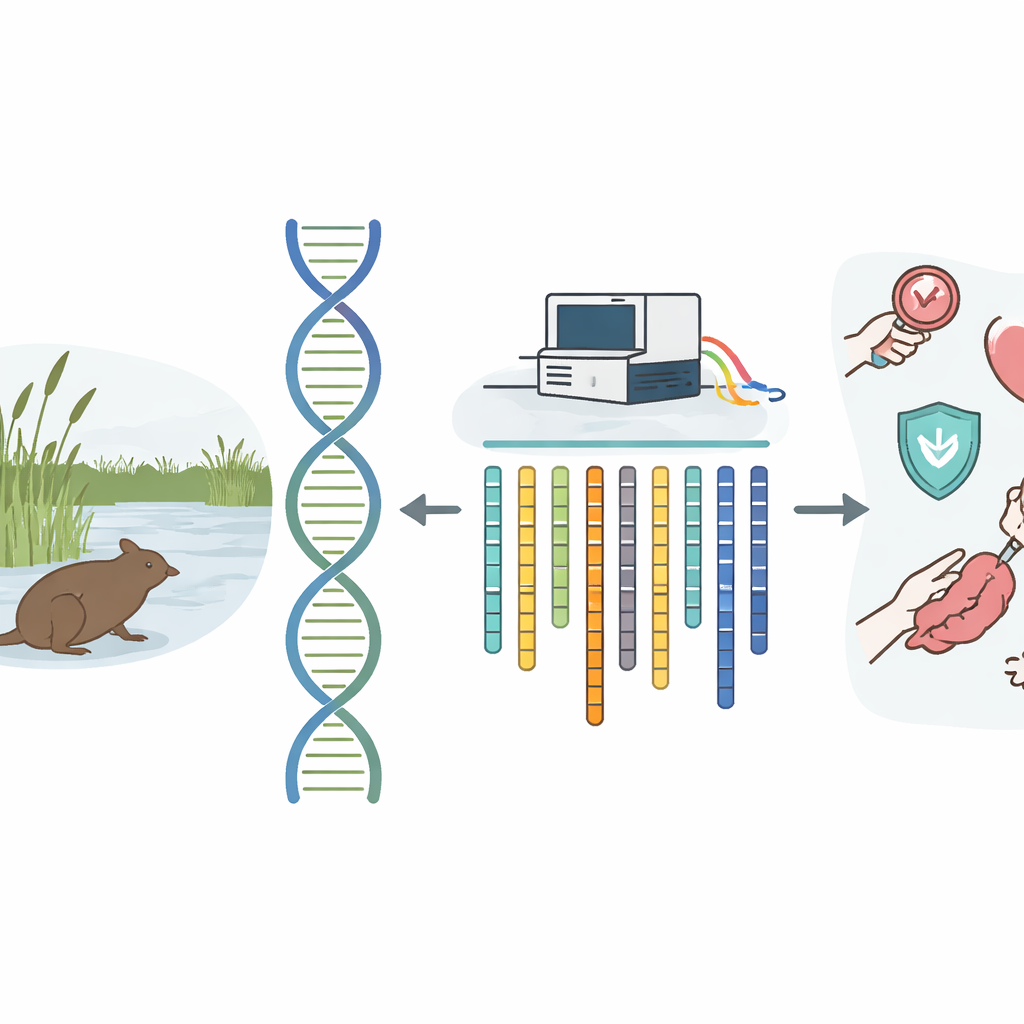
The reed vole, a mouse-sized rodent that lives in wetlands of China, turns out to be much more than a field-dwelling nibble of fur. It naturally resists a serious human parasite, develops certain cancers on its own in ways that resemble human disease, and thrives on a tough, high-fiber diet. All of these quirks make it an unusually valuable lab animal. Until now, however, scientists lacked a complete genetic blueprint for this species, limiting what they could learn from it. This study delivers that missing blueprint: a detailed, chromosome-level map of the reed vole’s DNA.
Why a Genome Map Matters
A high-quality genome is like a reference atlas for biology. Instead of scattered notes about individual genes, researchers get a carefully organized set of “chromosome-length” chapters that show how genes are arranged and how they may work together. For the reed vole (Microtus fortis), this is especially important. Its natural resistance to the parasitic worm Schistosoma japonicum, which causes schistosomiasis in humans, has long intrigued scientists. The vole is also used to study digestion, behavior, and ovarian cancer. Without a solid genome map, it has been hard to pinpoint which stretches of DNA shape these unusual traits or to compare its genes cleanly with those of mice, rats, or people.
How the Genetic Blueprint Was Built
The team started with tissues from a single healthy male vole to keep the genetic background as simple as possible. They extracted high-quality DNA and fed it into two modern sequencing technologies that excel at different jobs. PacBio HiFi sequencing produces long, highly accurate stretches of DNA, which are ideal for piecing together the basic draft of the genome. Hi-C sequencing captures which parts of the DNA sit close together inside the cell nucleus, giving clues about how smaller pieces should be linked into full chromosomes. The researchers also sequenced RNA from muscle, liver, and ovary to see which genes are actually switched on in different tissues, helping them find and confirm gene locations.
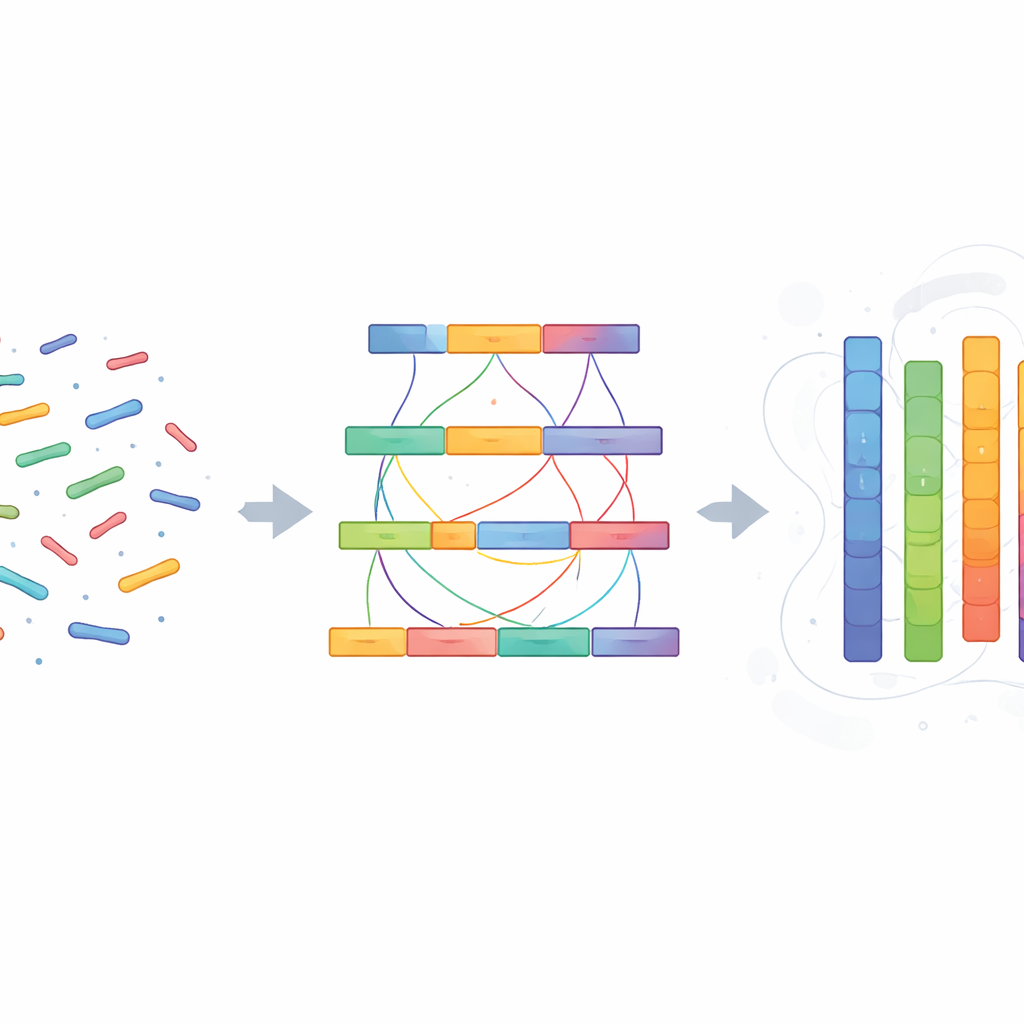
From Raw Reads to Full Chromosomes
Using specialized software, the scientists first stitched the long DNA reads into large blocks called contigs, then removed duplicate fragments arising from two copies of the same chromosome. Next, they used the Hi-C data—patterns of physical contact between distant DNA segments—to arrange and orient these blocks into 26 chromosome-length pieces that match the known karyotype, or chromosome count, of the reed vole. The final assembly is about 2.29 billion DNA letters long and remarkably continuous, with nearly all of it assigned to chromosomes. Quality checks showed that more than 96 percent of a standard set of important mammal genes are present and complete, a strong sign that the assembly is both comprehensive and accurate.
What the Genome Reveals Inside
With the assembled genome in hand, the team turned to its contents. They identified 23,678 protein-coding genes and could assign likely functions to almost all of them by comparing their sequences with public databases and searching for known protein building blocks. They also cataloged many kinds of noncoding DNA, such as mobile elements and repetitive sequences, which together make up nearly 42 percent of the genome, as well as thousands of RNA genes involved in protein synthesis and gene regulation. The researchers further built and checked a complete mitochondrial genome—the small, circular DNA molecule that powers cells—ensuring that future work can trace both nuclear and mitochondrial contributions to the vole’s biology.
A Foundation for Future Discoveries
By turning a once-fragmented picture of the reed vole’s DNA into a high-resolution, chromosome-level map, this study equips scientists with a powerful new tool. Researchers can now search systematically for the genes and genomic patterns that give the vole its natural resistance to schistosome parasites, its distinctive digestive system, and its value as a cancer and behavior model. The genome also provides a key reference for comparing rodents to one another, sharpening our understanding of how their bodies and defenses evolved. In practical terms, this work lays the groundwork for more targeted experiments and, ultimately, for insights that could inform human health, from infectious disease control to cancer research.
Citation: Zhang, D., Hu, Q., He, T. et al. Assembling a chromosome-level genome for the Microtus fortis using PacBio HiFi and Hi-C technologies. Sci Data 13, 447 (2026). https://doi.org/10.1038/s41597-026-06813-3
Keywords: reed vole genome, chromosome-level assembly, PacBio HiFi and Hi-C, schistosome resistance, rodent model organism