Clear Sky Science · en
Rapid temporal processing in the olfactory bulb underlies concentration-invariant odor identification and signal decorrelation
How the Brain Keeps Smells Stable
Walking past a bakery, the scent of fresh bread is recognizable whether it’s faint from down the street or intense at the door. Yet the nose’s chemical sensors are highly sensitive to how strong an odor is. This paper asks a simple question with deep implications: how does the brain recognize “the same smell” across a wide range of concentrations, and do this fast enough to guide behavior in just a fraction of a second?
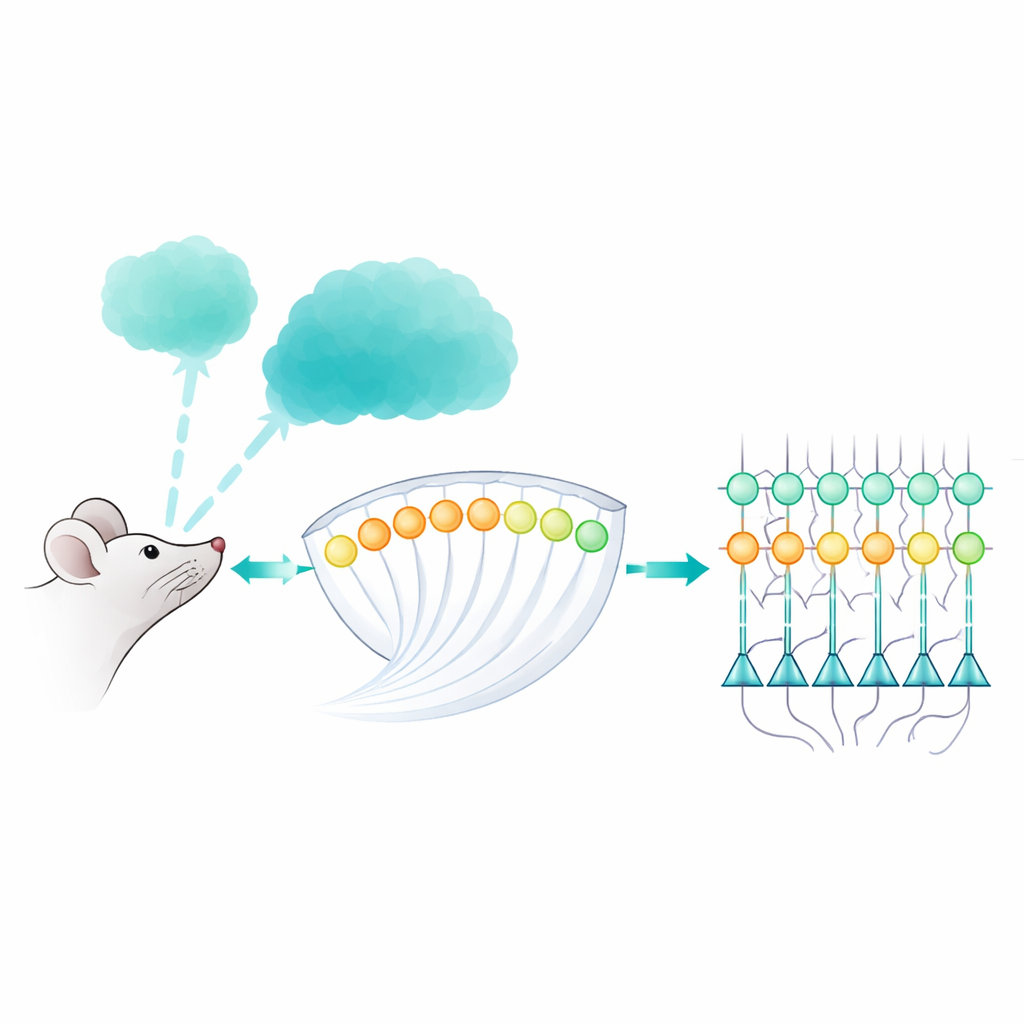
The First Stop for Smell Signals
When we sniff, airborne molecules bind to millions of receptor cells high inside the nose. Each type of receptor sends its signals to a dedicated cluster of nerve endings in the brain called a glomerulus, and thousands of these glomeruli tile the surface of a structure known as the olfactory bulb. From there, output neurons called mitral and tufted cells carry the information deeper into the brain. The authors took advantage of this tidy wiring to watch, in awake mice, how patterns of activity flow from glomeruli to mitral and tufted cells as odors of many types and strengths are inhaled.
Lighting Up and Probing the Circuit
To do this, the team built an all-optical set-up. They genetically equipped mice so that the olfactory receptors could be activated by light and the downstream neurons would flash when active. Using fast two-photon microscopy, they monitored hundreds of glomeruli and mitral/tufted cells at once. At the same time, a digital micromirror projector delivered pinpoint flashes of blue light to selected glomeruli, effectively letting the researchers “dial” input into specific channels. This combination allowed them to map which mitral and tufted cells were directly driven by a given glomerulus, and then see how those cells behaved when real odors, or mixtures of odors, arrived with each sniff.
The Power of Being Early
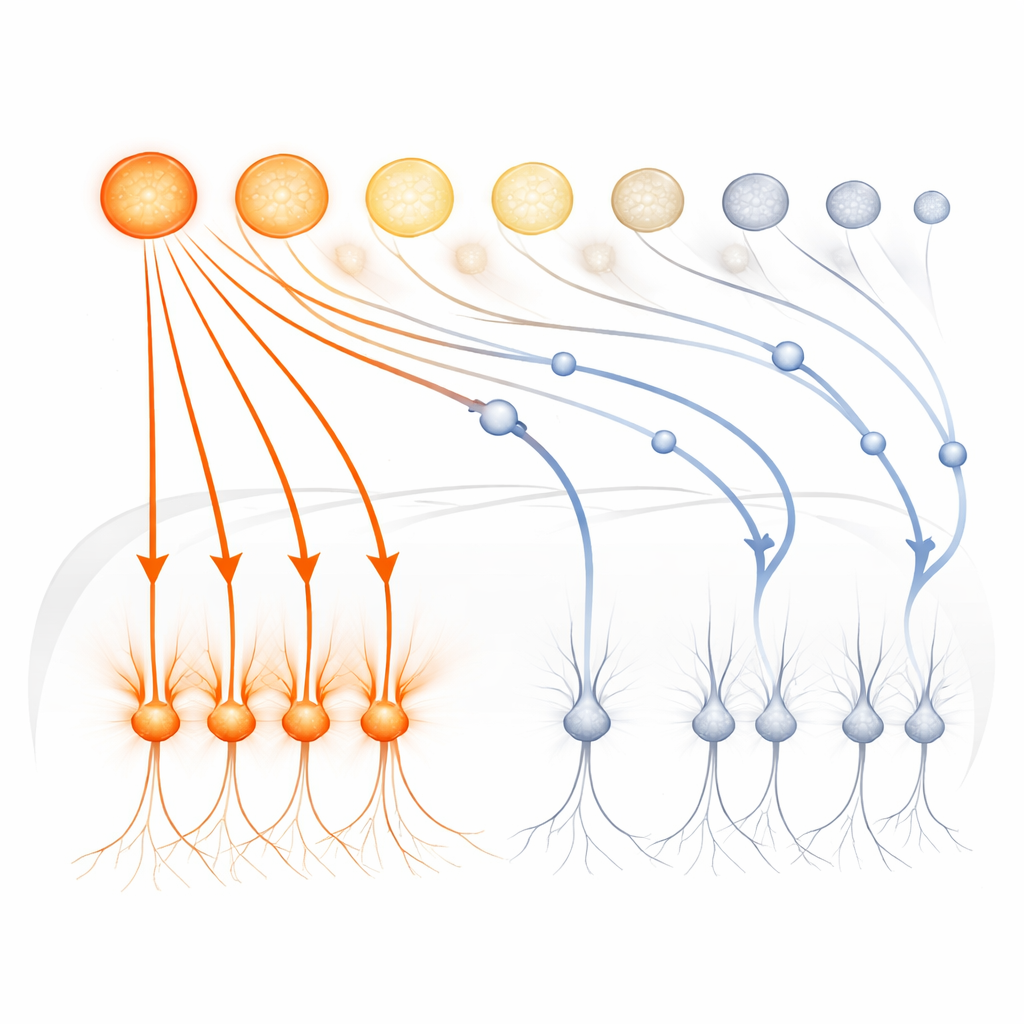
The results revealed that timing is everything. For any given odor, glomeruli did not all respond at once; instead they fired in a sequence, with some activating just after inhalation begins and others joining later. Crucially, the very first glomeruli to respond did so at nearly the same moments across low and high odor concentrations. Their associated mitral and tufted cells produced strong, stereotyped excitatory responses that were also remarkably stable across concentrations. In contrast, cells linked to later-responding glomeruli showed responses that changed dramatically with concentration and were often dominated by inhibition rather than excitation. This means that the earliest slice of activity in the bulb reliably signals odor identity, while later activity is more malleable and context-dependent.
A Brief Window and Strong Inhibition
To understand why late-arriving signals were so weak, the authors used light to poke single glomeruli at different times during a sniff. When a glomerulus was stimulated on an otherwise blank background, its partner mitral and tufted cells responded similarly no matter when in the sniff it was pulsed. But in the presence of an odor that already activated other glomeruli, the picture changed sharply: only pulses delivered in the first few tens of milliseconds after inhalation produced strong responses. Pulses arriving later were strongly suppressed for about 200 milliseconds. Similar behavior appeared when the team used odor mixtures instead of light. Together, these findings indicate that early-activated glomeruli recruit inhibitory circuits that slam shut a brief “excitability window,” preventing later inputs from effectively driving the bulb’s output.
Sharpening Smell and Separating Odors
This rapid temporal filter has two key consequences. First, because the same sensitive glomeruli tend to be the first to activate at both low and high concentrations, their mitral and tufted cell partners carry a concentration-invariant signature of odor identity to higher brain areas. Second, odors that might initially produce overlapping glomerular patterns are pulled apart as signals travel through the bulb: early channels are amplified, late ones are suppressed, and the resulting output patterns for different odors become less correlated. The study thus shows that the olfactory bulb is not just a relay, but an active processor that uses time and inhibition to stabilize what we smell and to keep similar scents distinct.
Why This Matters for Understanding Smell
To a lay observer, the main conclusion is that the brain solves a tricky problem—recognizing a smell quickly and reliably, regardless of how strong it is—by paying special attention to the first signals that arrive with each sniff and rapidly damping down everything that follows. This simple timing rule not only explains how odor identity can remain stable across large changes in concentration, but also how the system can separate similar smells in a fraction of a second. In short, the olfactory bulb uses a fast “first-come, first-served” filter to decide what information about an odor is worth sending on to the rest of the brain.
Citation: Karadas, M., Gill, J.V., Ceballo, S. et al. Rapid temporal processing in the olfactory bulb underlies concentration-invariant odor identification and signal decorrelation. Nat Neurosci 29, 1109–1121 (2026). https://doi.org/10.1038/s41593-026-02250-y
Keywords: olfactory bulb, odor concentration, temporal coding, lateral inhibition, sensory processing