Clear Sky Science · en
Distinct radial glia subtypes regulate midbrain dopaminergic neuron development
Why this brain story matters
Parkinson’s disease robs people of movement because a very specific set of brain cells, dopamine-producing neurons in the midbrain, gradually die off. One of the most promising treatment ideas is to replace these cells using stem cell–derived neurons grown in the lab. This paper asks a deceptively simple question with huge practical consequences: during normal development, which neighboring cells build, time and protect these dopamine neurons, and can we borrow their tricks to make better replacement cells?
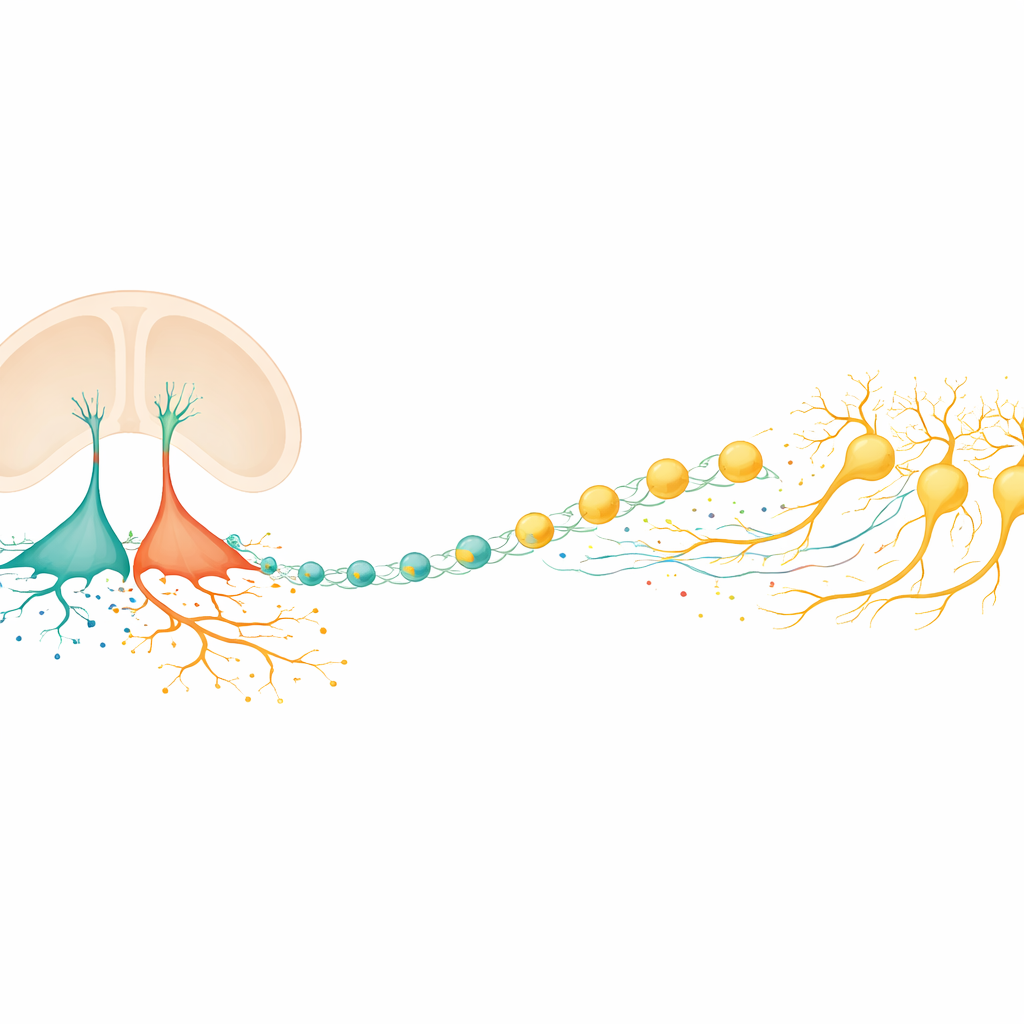
The brain’s hidden builders and helpers
In the embryonic brain, a special class of cells called radial glia act both as stem cells and as structural scaffolds. In the ventral midbrain region where dopamine neurons arise, earlier work had suggested there were at least three distinct radial glia subtypes, but their individual jobs were unclear. The authors combined large-scale gene-expression measurements from bulk tissue and single cells in mice and humans to tease this apart. They discovered that two subtypes in the floor of the midbrain take on especially important roles: one (called Rgl1) behaves as the main "starter" population that generates dopamine neuron precursors, while another (Rgl3) specializes in sending signals and shaping the surrounding molecular environment.
Mapping signals in a developing neighborhood
To understand how Rgl3 talks to other cells, the team built an expanded catalog of known signaling partnerships—pairs of secreted molecules and their receptors—and overlaid this on single-cell datasets from the developing mouse and human midbrain. Computational analyses showed that Rgl3 has more outgoing communication lines than almost any other cell type in the region. It sends out classic developmental signals that influence stem cells and young neurons, as well as guidance cues that help growing axons find their way. Rgl3 also turns out to be a major contributor to the extracellular matrix, the protein-rich mesh that surrounds cells, supplying distinct components that are poised to influence dopamine neuron survival and wiring.
Borrowing nature’s recipes to grow better neurons
Armed with a prioritized list of Rgl3-made molecules, the researchers moved to human stem cell cultures that were being steered toward a midbrain dopamine fate. They added selected signaling proteins and matrix components during the critical window when Rgl3 would naturally appear in vivo. Some factors—particularly two extracellular matrix proteins known for guiding nerve fibers—substantially boosted the proportion of dopamine-like neurons and, importantly, protected them from cell death without simply forcing more cell division. In contrast, blocking a receptor for one Rgl3-derived signal made dopamine neurons more likely to die, hinting that this pathway normally acts as a survival support line. These experiments show that copying the niche created by Rgl3 can noticeably improve the yield and robustness of lab-grown dopamine neurons.
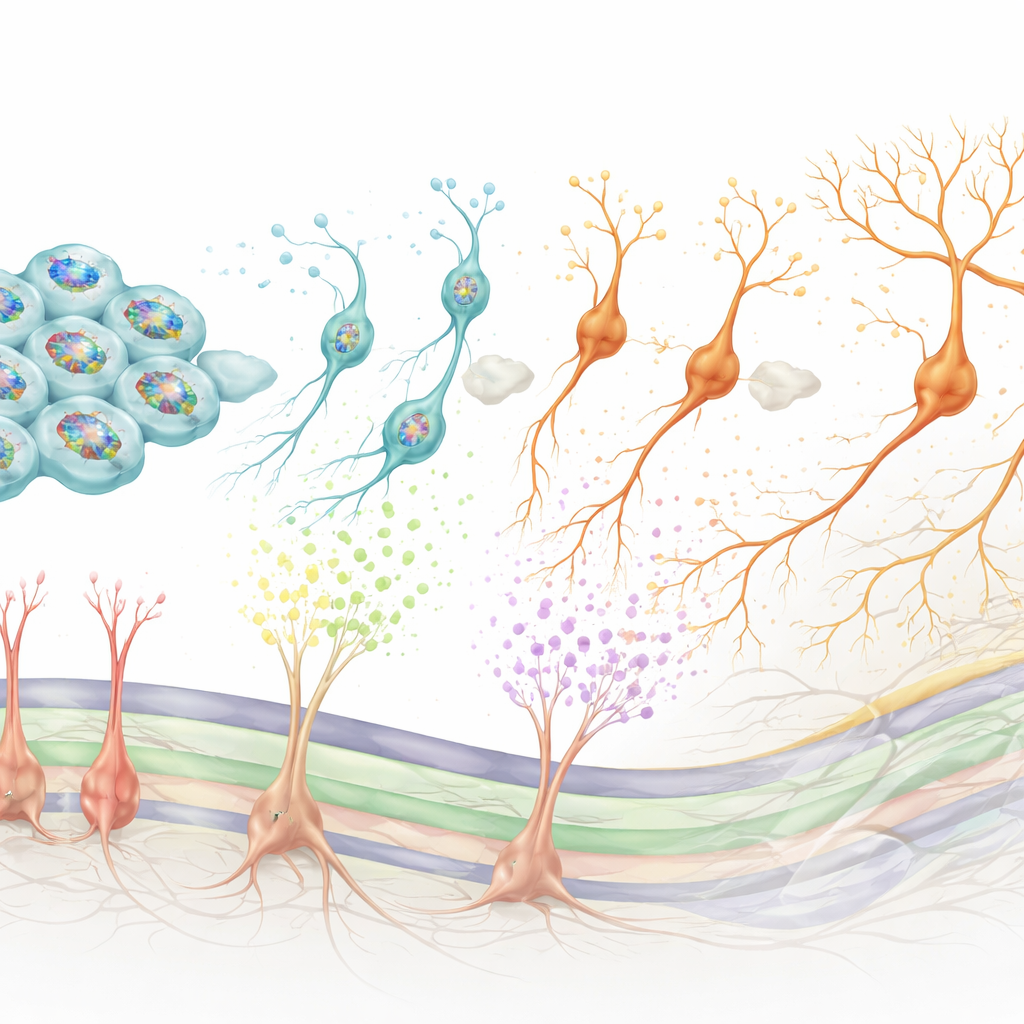
Timing the birth of neurons from the inside
While Rgl3 shapes the environment, Rgl1 controls when and how new dopamine neurons are born. By examining which control genes are active together in Rgl1, the authors identified a core regulatory network centered on BMAL1, a protein best known for keeping circadian clocks running. In human midbrain-like stem cell models, turning BMAL1 up at the right developmental moment caused progenitor cells to divide more and then enter the dopamine neuron pathway, increasing the final number of dopamine neurons. Knocking BMAL1 down had the opposite effect, pushing cells to exit the cycle too early and changing the timing of neuron birth. Additional tests linked BMAL1 activity to Wnt signaling, a key pathway for stem cell maintenance and differentiation, and showed that BMAL1 helps set the balance between staying a progenitor and committing to a dopamine neuron fate.
Tracing family trees in the developing midbrain
To connect these roles to real lineage relationships, the team used a barcoding strategy in human stem cell cultures. They isolated cells resembling Rgl1, tagged them with unique DNA barcodes, and then followed their descendants over time using single-cell sequencing. This revealed that individual Rgl1 cells can give rise both to dopamine neuron precursors and to Rgl3-like support cells, effectively generating both the main players and the caretakers in the same neighborhood. The reconstructed developmental “family trees” also aligned well with patterns seen in natural human fetal midbrain tissue, strengthening the case that these stem cell cultures faithfully recapitulate in vivo development.
What this means for future Parkinson’s therapies
Put together, the study shows that midbrain radial glia are not a uniform pool, but include at least one subtype that builds dopamine neurons and another that furnishes and protects their niche. Rgl1, through a BMAL1-centered control network and Wnt-linked signals, times when new dopamine neurons are produced. Rgl3, through a rich set of secreted factors and matrix proteins, improves their guidance, maturation and survival. By identifying the key molecules in these processes and demonstrating their effects in human stem cell systems, the work offers a roadmap for making dopamine neurons that are closer to their natural counterparts—more numerous, more stable and potentially better suited for transplantation in Parkinson’s disease.
Citation: Ásgrímsdóttir, E.S., Bassini, L.F., Sun, T. et al. Distinct radial glia subtypes regulate midbrain dopaminergic neuron development. Nat Neurosci 29, 810–824 (2026). https://doi.org/10.1038/s41593-026-02200-8
Keywords: midbrain dopaminergic neurons, radial glia, Parkinson's disease, stem cell therapy, neurodevelopment