Clear Sky Science · en
Mechanically driven Li dendrite penetration in garnet solid electrolyte
Why Cracks in Solid Batteries Matter
Next-generation solid-state batteries promise electric cars and devices that are safer and store more energy. A key ingredient is a hard ceramic that conducts lithium ions while blocking dangerous short circuits. Yet experiments keep revealing a puzzling failure: soft, metallic lithium grows as needle-like “dendrites” that somehow force their way through this hard ceramic, cracking it and causing the very shorts engineers are trying to avoid. This study digs into that paradox at the nanoscale and shows that the culprit is not chemistry alone but intense mechanical pressure that lithium builds up as it plates inside tiny cracks.
The Strange Case of Soft Versus Hard
Engineers use a lithium metal anode together with a stiff, garnet-type ceramic electrolyte to make all-solid-state batteries. In theory, the solid should act like armor, keeping the soft metal contained. Instead, thin lithium filaments can pierce the ceramic, eventually connecting the two sides of the battery and causing a short. Earlier ideas fell into two camps: either lithium inside existing filaments pressurizes and cracks the solid, or stray electrons leak through grain boundaries and seed many small metal islands that later connect. Distinguishing these pictures requires actually seeing where lithium sits and how the ceramic breaks, right where the damage occurs.
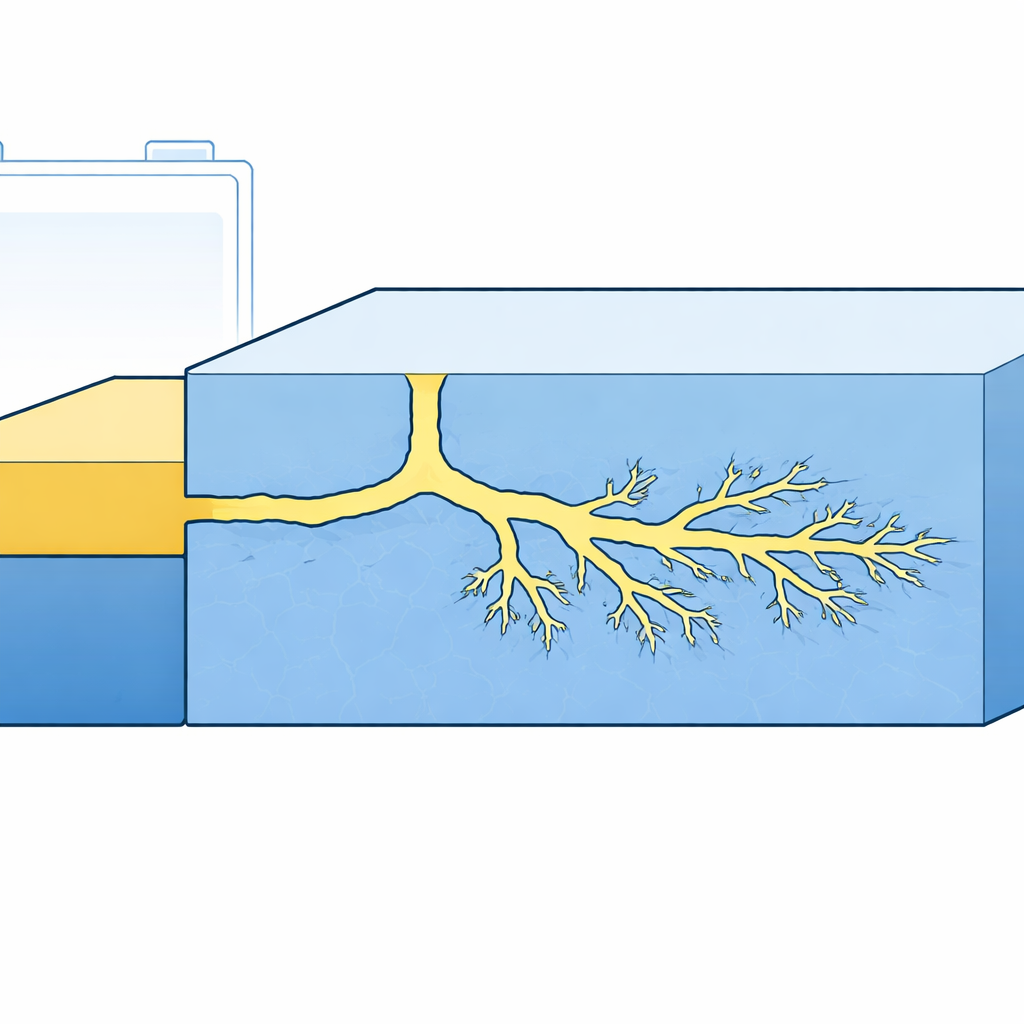
Watching Cracks Grow in 3D
The researchers built specially designed cells with a thinned garnet electrolyte so that a single lithium filament would grow in a controlled, easily imaged direction. Using cryogenic electron microscopes and focused ion beams, they reconstructed the crack networks in three dimensions at extremely low temperatures to preserve the fragile lithium. They found that the crack paths inside the ceramic are highly tortuous, sometimes slicing through grain boundaries between crystals and sometimes cutting straight through the crystal grains themselves. Importantly, they saw that nanoscale crack tips are completely filled with metallic lithium, whereas regions just ahead of the advancing tip showed no detectable lithium accumulation, even along grain boundaries that are often thought of as preferred growth sites.
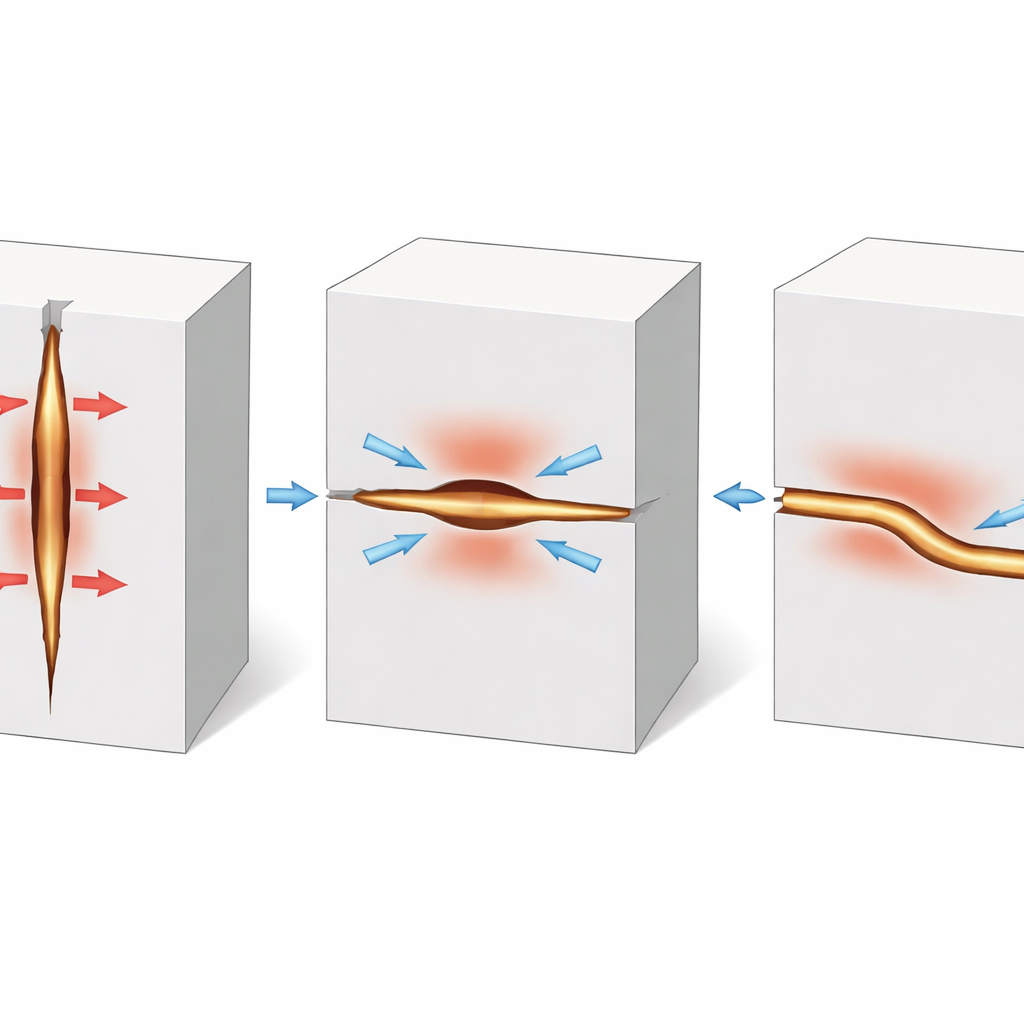
Pressure, Not Plastic Flow
To understand why a soft metal can break a brittle ceramic, the team mapped the internal crystal orientations of the lithium dendrites trapped inside microcracks. If the lithium were flowing and deforming plastically, its crystal lattice would show strong rotations and distortions. Instead, they observed only tiny orientation changes near the ceramic interface and almost none in the dendrite interior. This points to a state where the lithium is squeezed almost evenly in all directions—high hydrostatic pressure—rather than being heavily sheared. Advanced computer models that couple mechanics and fracture supported this view: as lithium plates into a confined crack, its internal pressure can climb to hundreds of megapascals, transferring strong tensile stresses to the surrounding ceramic and driving further crack advance, even when the lithium itself deforms very little.
Guiding Dendrites Away from Danger
Armed with the insight that crack-driven, pressure-loaded growth governs the “soft penetrates hard” behavior, the researchers tested whether they could steer dendrites away from catastrophic paths. They deliberately introduced rows of controlled surface indents that generate pre-existing transverse cracks, acting as mechanical “guard rails” inside the electrolyte. In operando observations showed that when a lithium filament encountered these engineered cracks, it turned and propagated sideways along them rather than continuing straight toward the opposite electrode. Simulations comparing different void shapes confirmed that elongated, transverse voids effectively redirect growth by reshaping the stress field, whereas round voids allow dendrites to continue straight through.
Designing Safer Solid-State Batteries
This work shows that lithium dendrite penetration in garnet electrolytes is a mechanically driven cracking problem: lithium fills existing flaws, builds up high internal pressure, and pries the brittle ceramic open. There is little evidence for isolated metal islands forming ahead of the tip under normal operating voltages. For practical batteries, this points to three key strategies: strengthen grain boundaries so cracks cannot easily deflect along them, toughen the ceramic so it can better dissipate stress, and deliberately engineer weak, transverse features that pull dendrites sideways before they reach the far electrode. Together, these approaches translate a nanoscale understanding of crack mechanics into clear guidelines for making solid-state batteries safer and more reliable.
Citation: Zhang, Y., Motahari, S., Woods, E.V. et al. Mechanically driven Li dendrite penetration in garnet solid electrolyte. Nature 652, 912–918 (2026). https://doi.org/10.1038/s41586-026-10415-9
Keywords: solid-state batteries, lithium dendrites, garnet electrolyte, battery safety, fracture mechanics