Clear Sky Science · en
Thymic health and immunotherapy outcomes in patients with cancer
Why a Small Organ Matters for Cancer Treatment
Most people have never heard of the thymus, a thumb‑sized organ tucked behind the breastbone. Yet this quiet structure trains the very immune cells that modern cancer immunotherapies try to unleash. This study reveals that the hidden condition of the thymus in adults strongly tracks with how well patients respond to immunotherapy, suggesting that our immune system’s “schoolhouse” may be just as important as the tumor itself when deciding who benefits from these powerful drugs.
Looking Beyond the Tumor
Immunotherapy drugs, especially immune checkpoint inhibitors, can lead to long‑lasting control of advanced cancers. Still, only a fraction of patients gain major benefit, and current tests mostly examine features of the tumor, such as how many mutations it carries or whether it displays certain surface molecules. These tumor‑focused tests miss a basic question: is the patient’s immune system healthy enough to mount a strong attack? The thymus plays a central role here by producing new T cells and keeping their repertoire diverse, but because it naturally shrinks with age, it has often been dismissed as unimportant in adults. The authors of this paper challenged that assumption, asking whether the visible state of the thymus in routine scans could predict how well patients fare on immunotherapy.
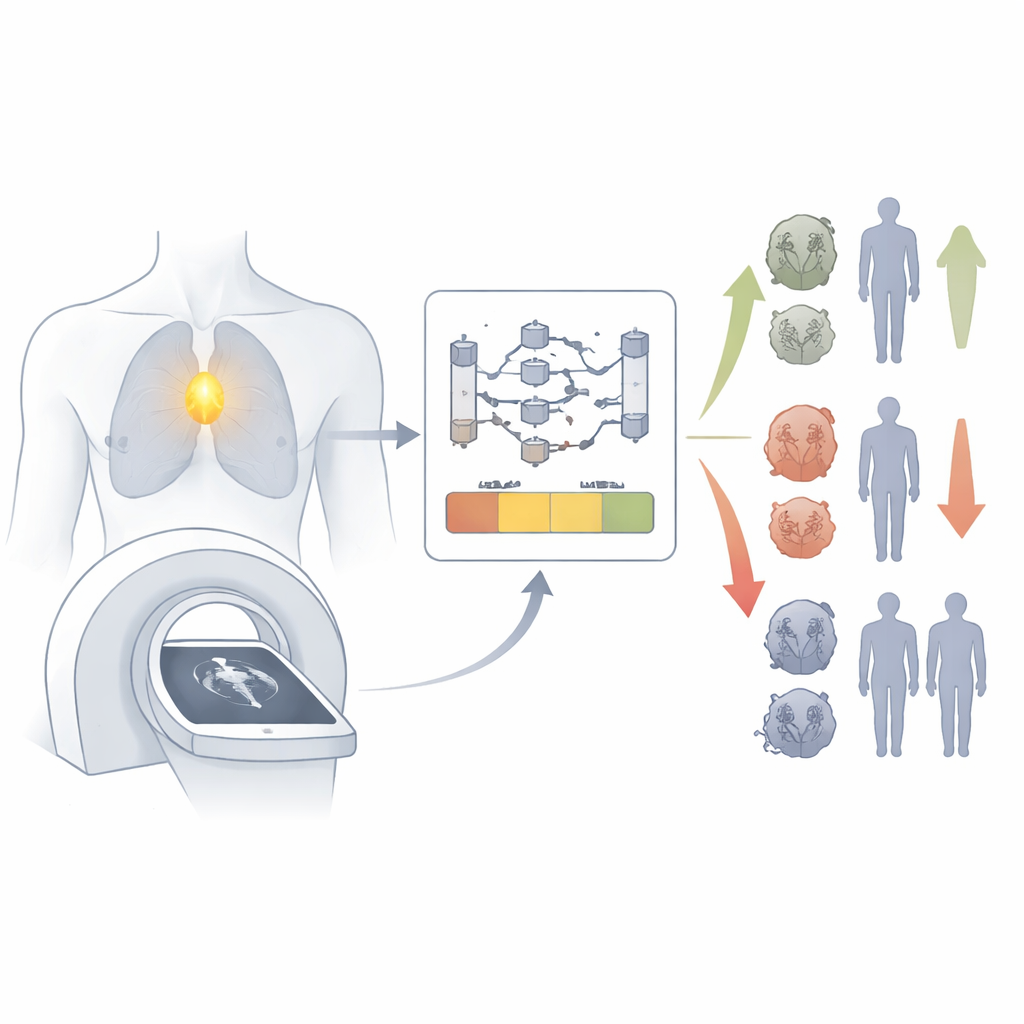
Turning Chest Scans into an Immune Health Score
The researchers built a deep‑learning system that analyzes standard chest CT scans—images almost every cancer patient already receives—to derive a continuous “thymic health” score. Using thousands of scans from 3,476 patients treated with checkpoint inhibitors across many cancer types, the system automatically located the thymus and translated its appearance into a number from low to high health. Patients were then grouped into low, average, or high thymic health based on where they fell in the overall distribution. This approach required no extra tests, no injections, and no new imaging—only clever use of data already collected in routine care.
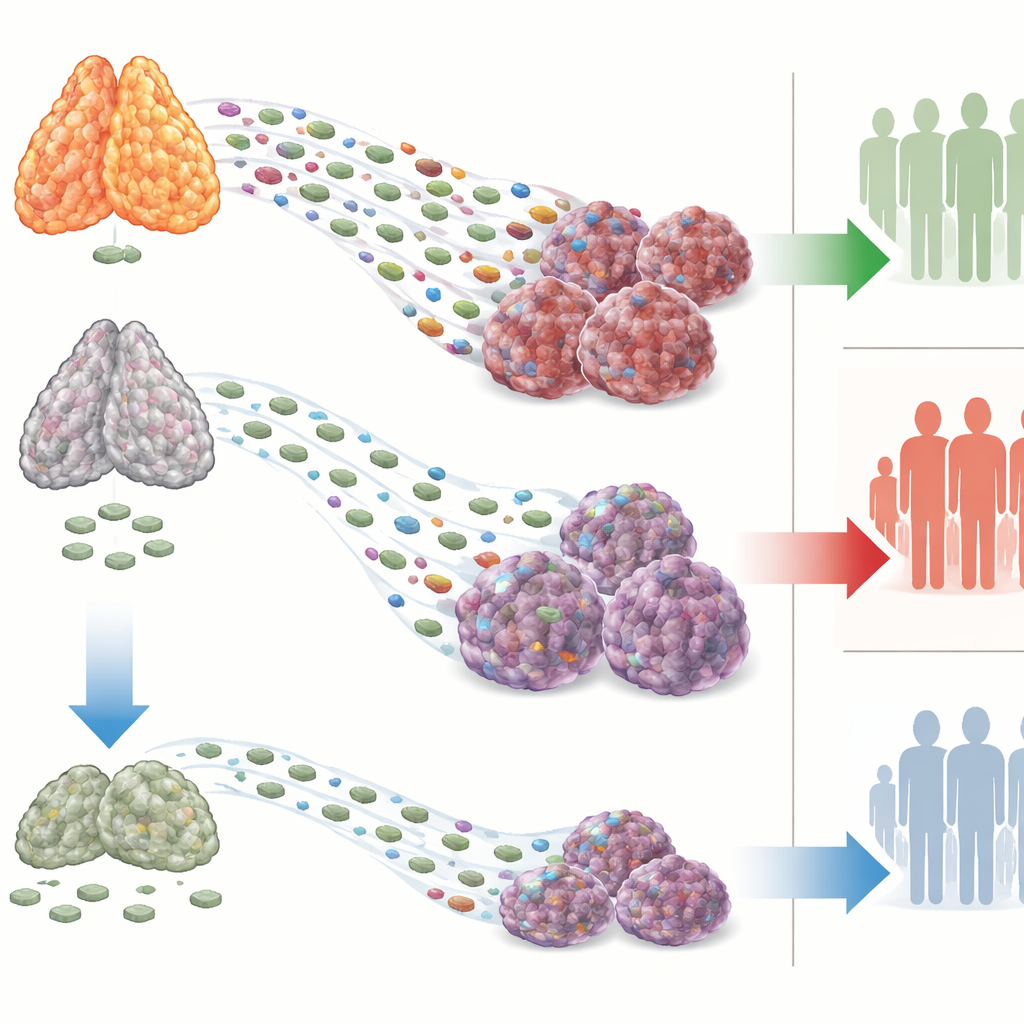
Stronger Thymus, Better Outcomes
In patients with advanced non‑small cell lung cancer, those with average or high thymic health had substantially lower risks of their cancer worsening or of dying compared with those with low thymic health. This pattern held whether immunotherapy was given alone or alongside chemotherapy, and it remained after accounting for age, sex, tumor type, smoking history, and established biomarkers such as PD‑L1 levels and tumor mutation burden. Importantly, thymic health mattered even in patients whose tumors looked unfavorable by traditional tests—for example, those with very low PD‑L1. Similar links between healthier thymus and longer survival appeared in patients with melanoma, kidney, and breast cancers, as well as in a pooled set of other tumor types, suggesting that this organ’s influence crosses disease boundaries.
Connecting Imaging to Immune Biology
To test whether the imaging‑based score truly reflected immune strength, the team turned to a carefully followed lung cancer trial called TRACERx. In this group of earlier‑stage patients, higher thymic health on CT scans coincided with several hallmarks of a vigorous immune system: more recent thymic output measured by DNA circles produced during T cell formation, greater diversity of T cell receptors in the blood and inside tumors, and higher estimated numbers of circulating and tumor‑infiltrating T cells. Blood protein patterns linked to a healthy thymus were enriched for pathways involved in adaptive immunity. Taken together, these findings show that what the algorithm sees in the thymus is not cosmetic—it mirrors genuine biological activity important for fighting cancer.
Implications for Personalized Care
Because thymic health can be read from routine scans, it has the potential to become a quick, non‑invasive measure of a patient’s immune readiness. The authors argue that, alongside tumor‑focused markers, this host‑focused indicator could help doctors better identify who is likely to benefit from immunotherapy, who might need alternative or additional treatments, and when treatment should begin—ideally before chemotherapy or other stresses further weaken the immune system. The work also raises practical questions for surgeons and radiation oncologists, who may need to reconsider removing or heavily irradiating the thymus in adults if it remains crucial for long‑term immune resilience.
What This Means for Patients
For patients, the message is that successful cancer immunotherapy depends not only on how the tumor looks under the microscope, but also on the hidden state of their own immune machinery. A healthier thymus appears to support a richer pool of T cells that can recognize and attack cancer when checkpoint inhibitors remove the brakes. While more studies are needed before thymic health is routinely used in the clinic, this research positions the thymus as a key, previously overlooked player in modern cancer care and suggests that protecting and possibly rejuvenating this small organ could one day improve outcomes for many people facing cancer.
Citation: Bernatz, S., Prudente, V., Pai, S. et al. Thymic health and immunotherapy outcomes in patients with cancer. Nature 652, 995–1003 (2026). https://doi.org/10.1038/s41586-026-10243-x
Keywords: thymus, cancer immunotherapy, T cell diversity, deep learning imaging, immune biomarkers