Clear Sky Science · en
Contrasting single-molecule magnet behaviour in dysprosium and terbium bis(stannolediide) complexes
Memory in a Single Molecule
Today’s hard drives and memory chips rely on tiny magnetic domains made of billions of atoms. Chemists are now pushing this idea to the extreme: can a single molecule remember a magnetic state and potentially store one digital bit? This paper explores a new family of such “molecular magnets” built from rare-earth metals and tin-containing rings, revealing how subtle changes in their structure can dramatically change how well they hold on to magnetic information.
Building Tiny Magnetic Sandwiches
The researchers focus on single-molecule magnets, special molecules that can keep their internal magnet pointing “up” or “down” even after an external magnetic field is switched off. This behavior makes them candidates for ultra‑dense data storage and elements in quantum technologies. The team works with two rare-earth metals—dysprosium and terbium—that are known for strong magnetic properties. They surround each metal ion with two flat, ring-shaped ligands containing tin atoms, forming a sandwich-like structure. These rings carry a high negative charge, which creates a very directional (“axial”) magnetic environment that, in principle, helps lock the molecular magnet in place.
Making and Tuning the Magnetic Molecules
To build these sandwiches, the authors first prepare a highly charged tin-based ring unit, then react it with terbium or dysprosium salts to give complexes called bis(stannolediides). A positively charged potassium ion initially sits between the tin rings, helping to assemble the structure but not strongly bonding to the rare-earth metal. Using a crown-shaped additive, 18‑crown‑6, they can remove the potassium ion. With terbium, this simply yields a cleaner negatively charged sandwich molecule. With dysprosium, the potassium removal triggers an internal electron shift, converting the metal from a +3 to a +2 state and producing a different, doubly charged sandwich. Careful X‑ray measurements reveal that all these molecules are almost linear stacks of rings around the metal, a geometry known to favor strong magnetic directionality.
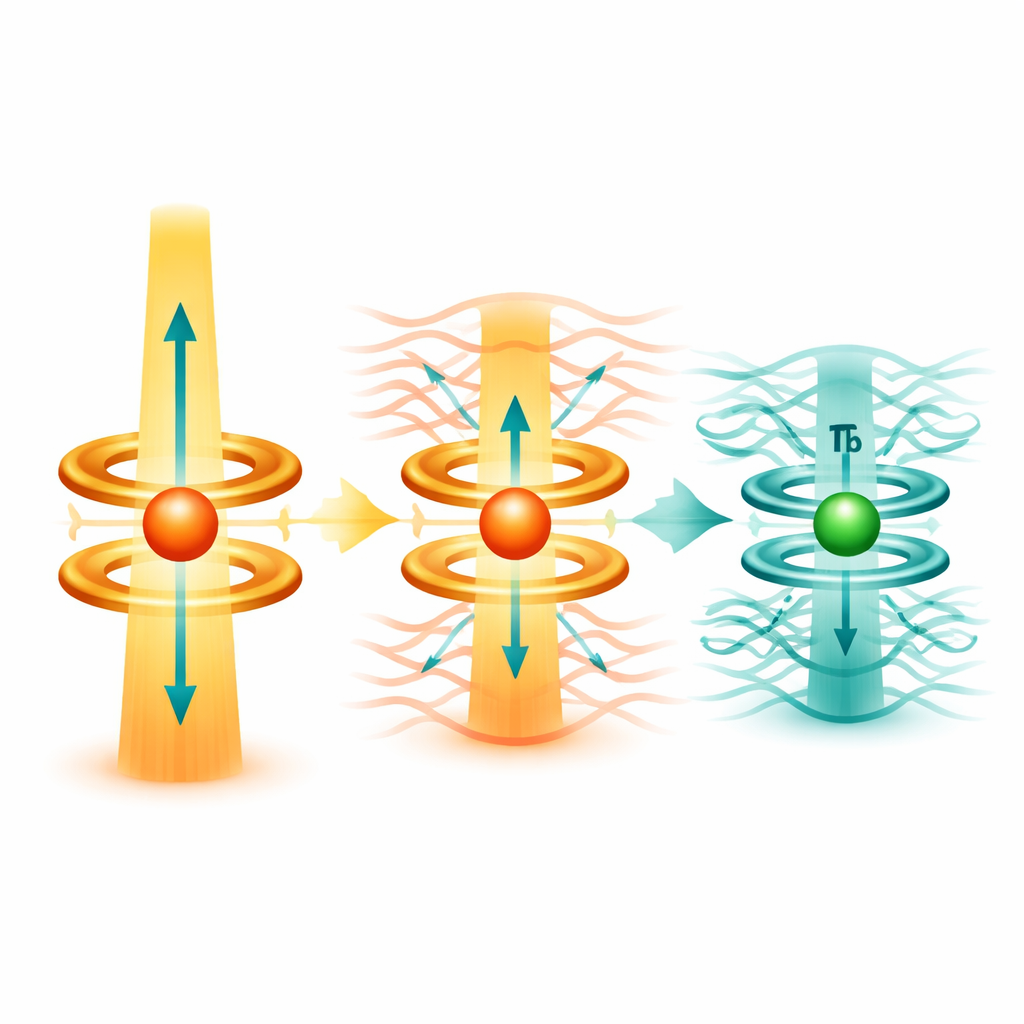
How the Molecules Behave as Magnets
The team then measures how the molecules respond to changing magnetic fields and temperatures. The dysprosium(III) sandwich stands out: it shows very slow magnetic relaxation and keeps its magnetization up to about 55 kelvin—far above the temperature of liquid nitrogen. The energy barrier that must be overcome to flip its magnetic direction is around 1,500 kelvin, indicating a very stable magnetic state within the molecule. In contrast, the terbium(III) sandwiches do behave as single-molecule magnets, but their barriers are lower and they lose their magnetization much more quickly, especially at higher temperatures. By applying a modest steady magnetic field, the researchers can suppress fast relaxation pathways, revealing the underlying energy barriers and showing that vibrations of the molecular framework strongly influence how fast the magnetization decays.
When an Extra Electron Spoils the Order
The dysprosium(II) sandwich, formed after potassium removal, offers a surprising twist. Structurally it looks nearly ideal: a perfectly linear stack of rings, which might suggest excellent magnetic performance. Yet magnetic measurements show that it has only weak directionality and loses its magnetization quickly. Quantum chemical calculations explain why: an extra electron occupies a more extended orbital that mixes with tin-based orbitals on the rings. This interaction generates a magnetic environment that is less focused along one axis and more spread out, effectively erasing the sharp directionality that single-molecule magnets need to work well.
Why These Tiny Magnets Matter
Taken together, the results show that tin-containing ring ligands can create powerful, highly directional environments around rare-earth ions, producing robust single-molecule magnets—especially with dysprosium(III). At the same time, the comparison between terbium(III), dysprosium(III) and dysprosium(II) highlights how delicately magnetic behavior depends on both the charge state of the metal and the way its electrons interact with surrounding atoms. By learning how vibrations, subtle bonding changes and geometry control the “magnetic memory” of a single molecule, chemists move closer to designing custom molecular bits for future data storage and quantum devices.
Citation: Sun, X., Hinz, A., Maier, S. et al. Contrasting single-molecule magnet behaviour in dysprosium and terbium bis(stannolediide) complexes. Nat. Chem. 18, 872–881 (2026). https://doi.org/10.1038/s41557-026-02114-9
Keywords: single-molecule magnets, lanthanide chemistry, molecular data storage, magnetic anisotropy, quantum materials