Clear Sky Science · en
Generating extended foldamer dye stacks and unravelling their evolving exciton dynamics
Why longer dye chains matter
Smartphones, solar cells and sensors all rely on how efficiently materials can absorb and emit light. Chemists often predict these behaviors using very simple models that consider only pairs of light-absorbing units, called dimers. This paper shows that such pair-based models can be misleading. By building much longer, precisely ordered chains of dye molecules, the authors reveal that new, brighter light-emitting states appear only once the chain reaches a certain length, changing how we should think about designing future optical and electronic materials.
Building molecular “toy trains” of dyes
The researchers use a popular organic dye known as perylene bisimide and connect these dyes into rigid, stacked chains, called foldamers. Instead of linking dyes one by one all the way to fourteen units, they devise a block-based strategy, much like snapping together prefabricated train cars. Small central blocks and end pieces are prepared separately and then fused, making it possible to access chains from a single dye all the way up to a fourteen-dye stack. These chains are remarkably well defined, comparable in size and precision to small proteins or short DNA strands, but made entirely of light-absorbing dyes rather than natural building blocks.

Proving the stacks are orderly
To confirm that these dyes really sit in a tight, orderly stack rather than in a loose jumble, the team uses high-resolution nuclear magnetic resonance experiments in a hot, viscous solvent. Subtle shifts and cross-signals between hydrogen atoms show that the dyes lie almost directly on top of one another with a small, regular offset, like a neatly slid deck of cards. Computer calculations back this up, predicting close contact between the flat dye surfaces and strong overlap of their electronic clouds. Interestingly, the different types of electronic couplings between dyes largely cancel each other, leading to overall “null-like” coupling that disguises the complexity of the underlying interactions.
Watching light energy move and settle
When these dye stacks are excited with light, their absorption and glow (fluorescence) patterns change systematically with chain length. Simple dimers behave one way: they show a mix of a normal bright state and a more complex “multiexciton” state that holds two linked excitations and can either split into two long-lived dark states or fuse back to give light. As the stacks grow to four, five and six dyes, this multiexciton character strengthens, the glow bands become narrower and more intense, and the overall light-emission efficiency climbs sharply—from about half for the dimer to three-quarters for the fourteen-unit chain. Ultrafast laser experiments reveal that, in longer stacks, the complex state forms very quickly and the resulting excitation gradually localizes in the more rigid central part of the chain, rather than wandering along its entire length.
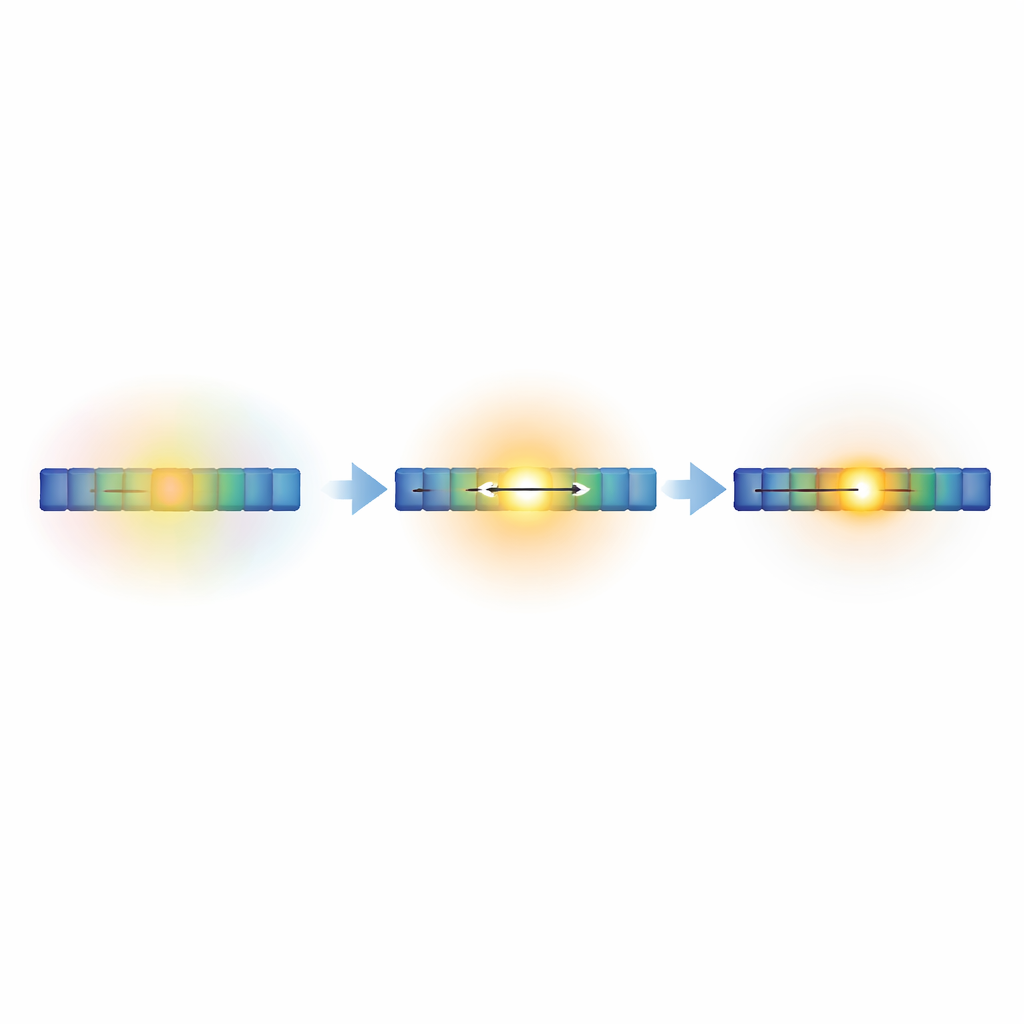
How the environment stops quenching the light
The surrounding liquid normally gives excitations many ways to lose energy without emitting light. In short dye chains, the multiexciton state remains exposed to this environment, and a significant fraction of excitations escape by forming dark, long-lived triplet states. In longer chains, however, the central dyes become more structurally locked and better shielded from solvent motion. Measurements show that non-radiative loss pathways are strongly suppressed, while light emission from the multiexciton state becomes dominant, especially beyond six dyes. This means that, past a certain length, the chain effectively creates a protected “bright zone” in its middle, where excitations can shine rather than fade away.
Rethinking simple models of light in materials
Overall, the study demonstrates that a pair of dyes is not enough to represent how real dye-based materials behave. Only when at least six dyes are tightly stacked do the desirable bright, long-lived states fully emerge, much like how protein helices or DNA double strands require a minimum length to form stable structures. For designers of organic electronics, lasers and light-harvesting systems, this work argues that larger, precisely built stacks are essential for predicting and tuning performance. Extended dye foldamers like these could serve as model “wires” for charge and energy flow and as building blocks for next-generation nanoelectronic and nanophotonic devices that are both brighter and more efficient.
Citation: Ernst, L., Hong, Y., Song, H. et al. Generating extended foldamer dye stacks and unravelling their evolving exciton dynamics. Nat. Chem. 18, 923–930 (2026). https://doi.org/10.1038/s41557-026-02082-0
Keywords: perylene dye stacks, multiexciton dynamics, organic optoelectronics, foldamer materials, exciton localization