Clear Sky Science · en
Electron hopping in conjugated molecular wires with application to solar cells
Wires Too Small to See
Imagine being able to control how single electrons move across a device the way traffic engineers manage cars on a highway. This study zooms in to the nanometre scale, where chemists build invisible “wires” made of molecules to guide charges inside solar cells. By understanding and reshaping how these tiny currents flow, the researchers show a new strategy to squeeze more power from next-generation, lead-free solar cells.
Tiny Bridges Between Light and Power
At the heart of the work are molecular wires: chains of linked rings and bonds only one to three nanometres long, thousands of times thinner than a virus. One end of each wire is fixed to a transparent conducting material called indium tin oxide, widely used in touchscreens and solar cells. The other end carries a special iron-based unit called ferrocene that easily gives and takes electrons. When these wires form an ultrathin, ordered layer on the electrode surface, they act as custom-built bridges that connect the electrode to other parts of an electronic or solar device, while also serving as a well-defined playground for studying how electrons move across interfaces.
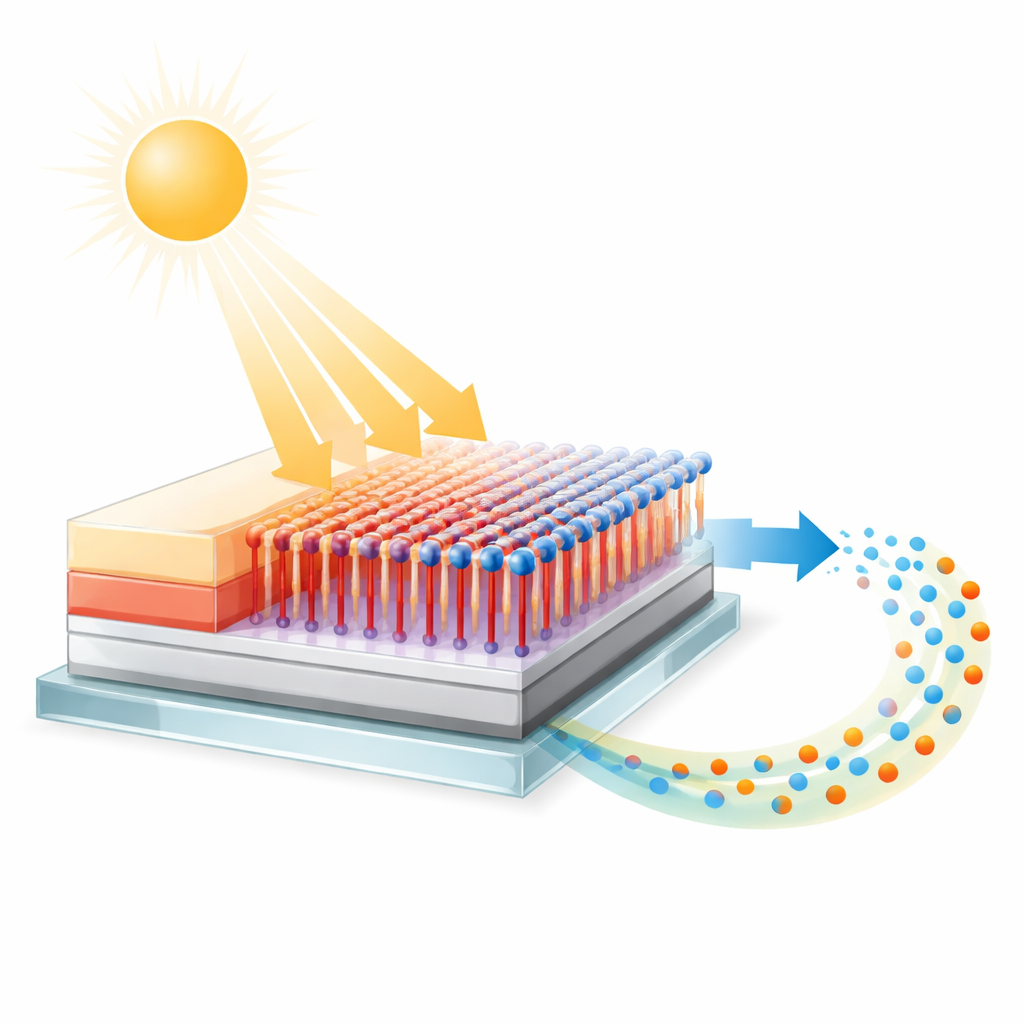
Watching Electrons Move Step by Step
To probe what the electrons actually do, the team turned to electrochemistry, using voltage sweeps to push electrons back and forth between the ferrocene ends and the indium tin oxide surface. From the shape and timing of the electrical signals, they extracted how fast electrons transfer and how that rate changes as the wire gets longer or the temperature varies. Surprisingly, even their shortest wire—only about one nanometre long—did not behave like a simple quantum tunnel, where the chance of an electron appearing on the other side drops off steeply with distance. Instead, the electron transfer slowed only gently as the wire length increased and became faster at higher temperatures, signatures of a “hopping” process in which charge moves in small steps along the wire rather than leaping straight across.
Why This Electrode Makes Hopping Easy
The key to this unusual behaviour lies in how the energies of the materials line up. The researchers compared the energy level at which electrons sit in indium tin oxide with that of the molecular wires when the ferrocene end is oxidized. They found that these levels are very closely matched, more so than is typical for gold, a common metal in single-molecule electronics. This small energy gap means that it costs relatively little energy for an electron to hop from the electrode into the wire and then on to the ferrocene unit. Calculations show that as more building blocks are added to the wire, the electron-rich orbitals spread out along the backbone and closer to the surface, further encouraging stepwise hopping. Together, the gentle distance dependence, the thermal activation, and the near-perfect energy match point to hopping as the dominant pathway, even over ultrashort distances where tunnelling is usually assumed to rule.
Plugging Molecular Wires into Solar Cells
Armed with this mechanistic insight, the team asked whether their best-performing wire could improve a real device. They attached the shortest ferrocene-terminated wire to smooth indium tin oxide and then grew a thin film of a tin-based perovskite, a promising lead-free light absorber, on top. In these solar cells, the molecular wire layer plays the role of a “hole-extraction” contact, pulling positive charges away from the perovskite after light absorption and sending them into the external circuit. Compared with standard hole-transport layers used in tin perovskite cells, devices using the molecular wire achieved a higher voltage and better current, reaching a power conversion efficiency of about 9.5 percent. Control devices that used a similar molecule without the ferrocene end group performed far worse and showed higher internal resistance, underscoring the importance of the redox-active terminus for swift charge transfer.
From Fundamental Insight to Future Devices
For non-specialists, the main message is that by carefully matching energies and structures at the atomic scale, chemists can coax electrons to move in ways that were once thought unlikely—here, making a hopping pathway dominate even over nanometre distances. This not only deepens our understanding of how electrons cross the boundary between a solid electrode and a molecular layer but also offers a new tool for engineering interfaces in solar cells and other optoelectronic technologies. As molecular design and device fabrication continue to advance hand in hand, such custom-built wires could help make thin, flexible, and more sustainable solar technologies a practical part of everyday life.
Citation: Fang, F., Li, A., Geoghegan, B.L. et al. Electron hopping in conjugated molecular wires with application to solar cells. Nat. Chem. 18, 756–764 (2026). https://doi.org/10.1038/s41557-025-02034-0
Keywords: molecular wires, electron transfer, indium tin oxide, perovskite solar cells, ferrocene