Clear Sky Science · en
Aluminum nanoparticle enhanced TiO2 photocatalysis of organic pollutants under solar and UV-B irradiation
Turning Sunlight into Safer Drinking Water
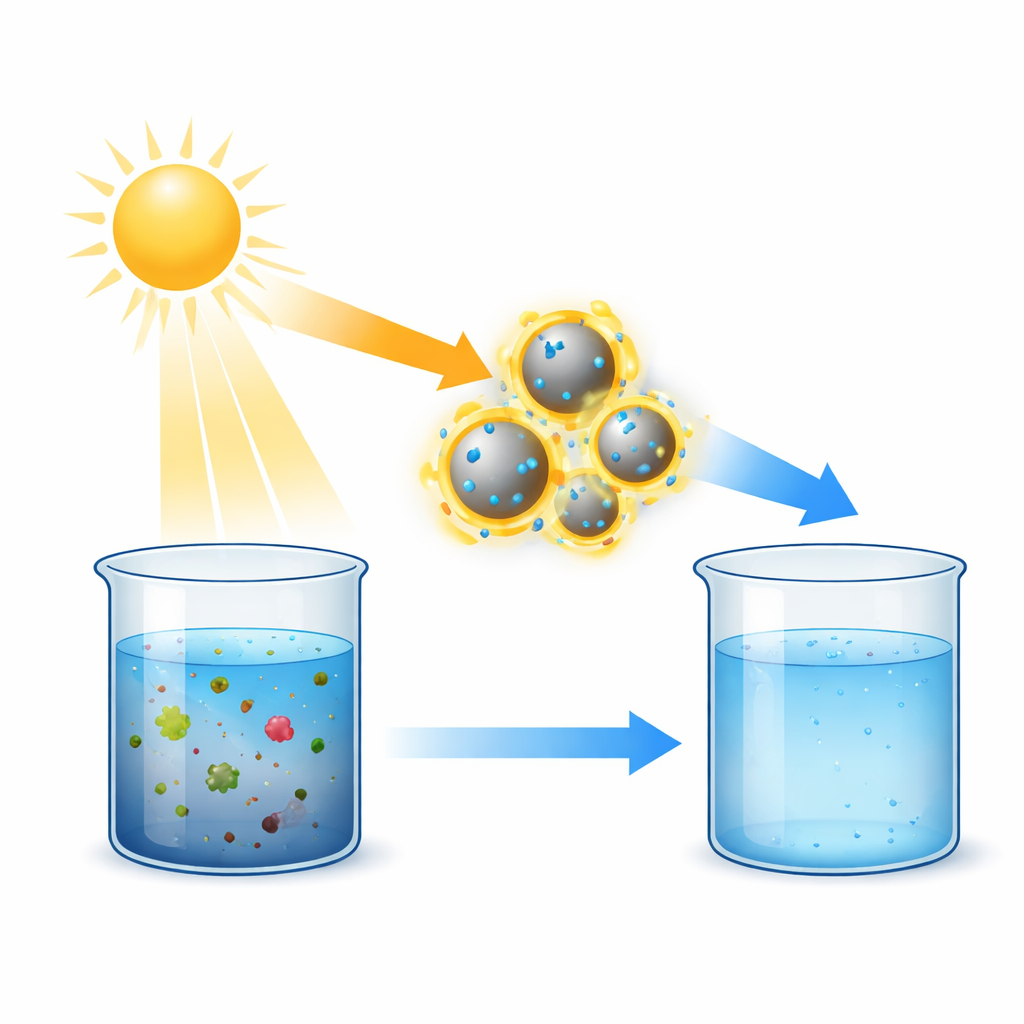
Billions of people still lack access to reliably clean drinking water, especially in regions where electricity and chemical supplies are scarce. This study explores a way to use ordinary sunlight and tiny engineered particles to help break down stubborn chemical contaminants in water. By combining two low-cost materials—aluminum and a common white pigment called titanium dioxide—the researchers show it is possible to clean water more efficiently without adding extra chemicals or consuming much energy.
Why Tiny Particles Matter
The work builds on a technology called photocatalysis, in which light-activated materials help destroy unwanted chemicals. Titanium dioxide is a workhorse in this field because it is cheap, stable, and already used in many products. However, it mainly responds to ultraviolet light, which makes up only a small slice of sunlight. To make better use of the full solar spectrum, the team paired titanium dioxide with ultra-small aluminum particles. These aluminum particles act like miniature antennas for light: when they are hit by the right wavelengths, the electrons inside them move collectively, concentrating energy at their surface and helping trigger chemical reactions nearby.
Building a Stable Cleaning Team
Simply mixing the two materials is not enough; the particles must stay well-dispersed in water and in close contact to transfer energy efficiently. The researchers therefore crafted a “heterostructure,” a combined particle where aluminum nanoparticles sit on the surface of titanium dioxide. Each aluminum particle has a metallic core and a very thin shell of aluminum oxide that naturally forms in air. To keep everything stable in water, the team used small bridging molecules, including one called cysteine, which can grab onto both aluminum and titanium dioxide. Advanced imaging and spectroscopy confirmed that these bridges successfully link the materials together without significantly changing the underlying light-absorbing properties of titanium dioxide.
How Light Helps Break Down Dyes
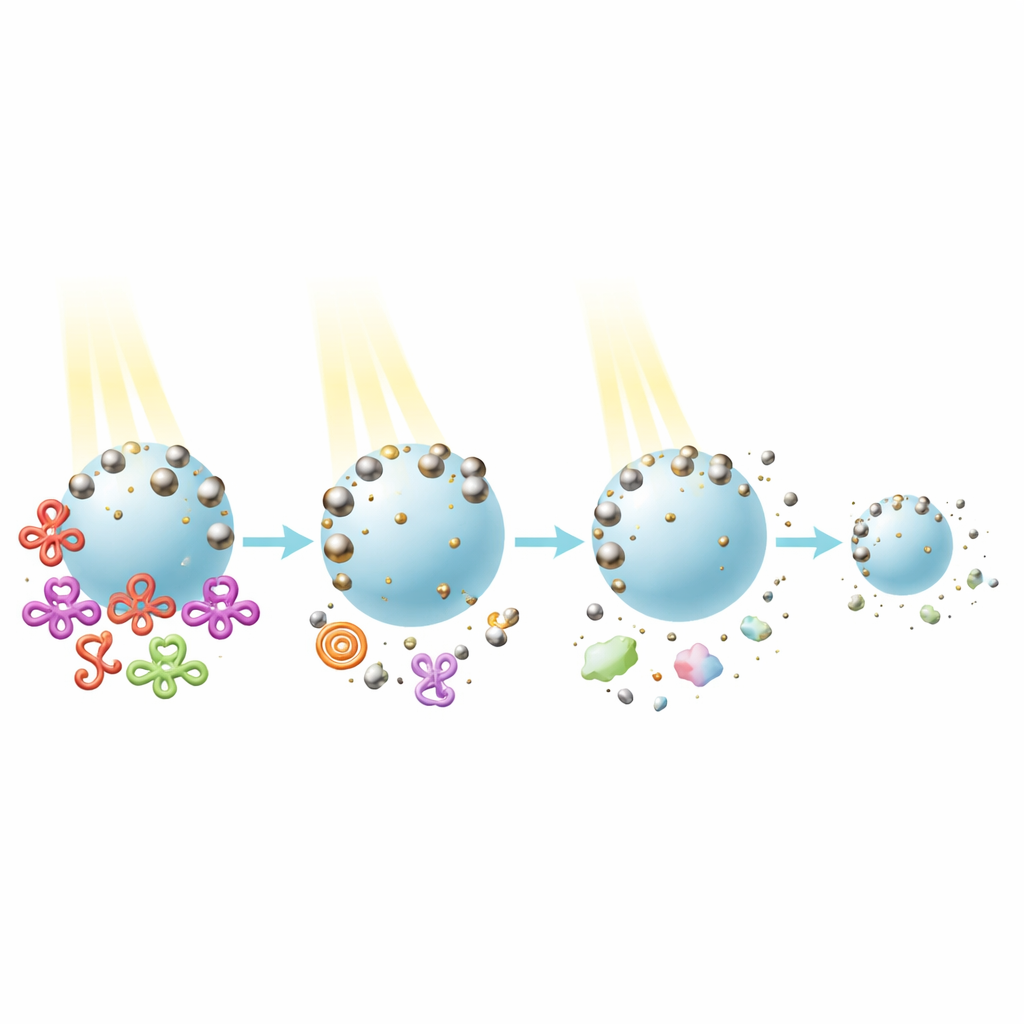
The team then tested how well their new particles could remove two common types of water pollutants: a brightly colored dye called amaranth, which carries a negative charge, and phenol, a neutral industrial chemical. Under a lamp that mimics sunlight, the cysteine-linked aluminum–titanium dioxide particles broke down amaranth about 60 percent faster than standard titanium dioxide alone. Under a narrower ultraviolet lamp, the improvement was smaller but still present. The results suggest that the aluminum core concentrates light energy and can feed energetic electrons into the titanium dioxide, while the surrounding aluminum oxide shell helps attract dye molecules close to the reactive sites and slows down wasteful recombination of charges inside the catalyst.
Selective Action and Lasting Power
Interestingly, the boosted performance depended on the type of pollutant. The new particles were much more effective for the negatively charged dye than for neutral phenol, indicating that how pollutants interact with the catalyst surface strongly affects cleaning efficiency. Trapping experiments showed that positively charged “holes” formed in the material, along with some highly reactive oxygen-containing species, are the main actors in destroying the dye molecules. Just as important for real-world use, the aluminum–titanium dioxide particles remained stable through multiple cleaning cycles under simulated sunlight. They did not require complicated washing or drying between cycles, and they consistently outperformed the standard material.
A Step Toward Affordable Solar Water Treatment
Overall, this study shows that pairing abundant aluminum nanoparticles with titanium dioxide can substantially improve solar-driven breakdown of certain organic pollutants in water, without relying on rare precious metals or added chemicals. While more work is needed to understand all the microscopic steps and to test a wider variety of contaminants, the approach points toward compact, low-cost water treatment systems that could one day help deliver safer drinking water in areas where conventional infrastructure is difficult to build or maintain.
Citation: Wasim, S., Loeb, S.K. Aluminum nanoparticle enhanced TiO2 photocatalysis of organic pollutants under solar and UV-B irradiation. npj Clean Water 9, 29 (2026). https://doi.org/10.1038/s41545-026-00560-z
Keywords: solar water treatment, photocatalytic nanoparticles, aluminum titanium dioxide, organic pollutant degradation, clean drinking water