Clear Sky Science · en
An innovative nasal nanovaccine against SARS-CoV-2 induces systemic and upper airway immunity controlling viral replication
Why a nose-based COVID-19 vaccine matters
Most COVID-19 shots do a good job of keeping people out of the hospital, but they are far less effective at blocking the virus where it first enters the body: the nose and upper airways. That is why people can still get infected and spread the virus even after being vaccinated. This study describes an experimental nasal “nanovaccine” that aims to build a sturdy immune shield right at the doorway of infection, while also strengthening protection throughout the body.
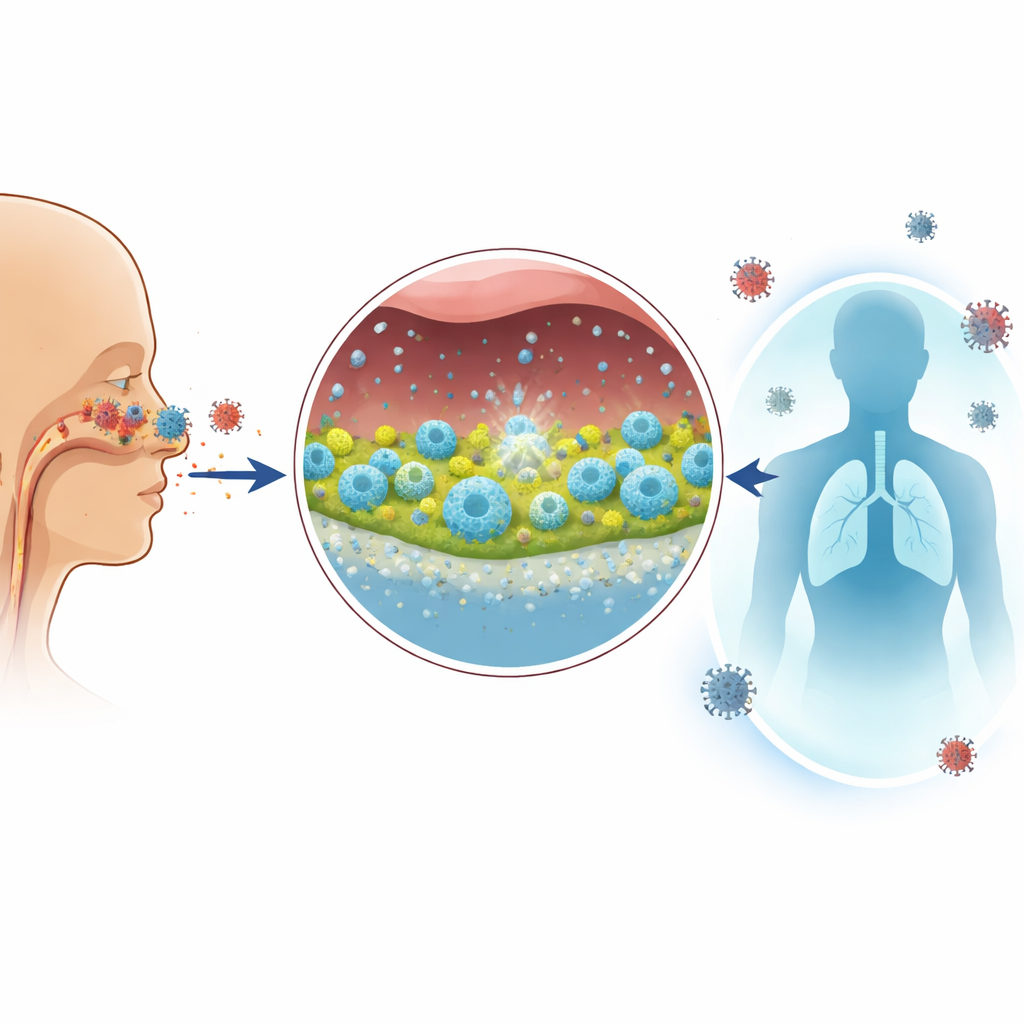
A new way to deliver protection through the nose
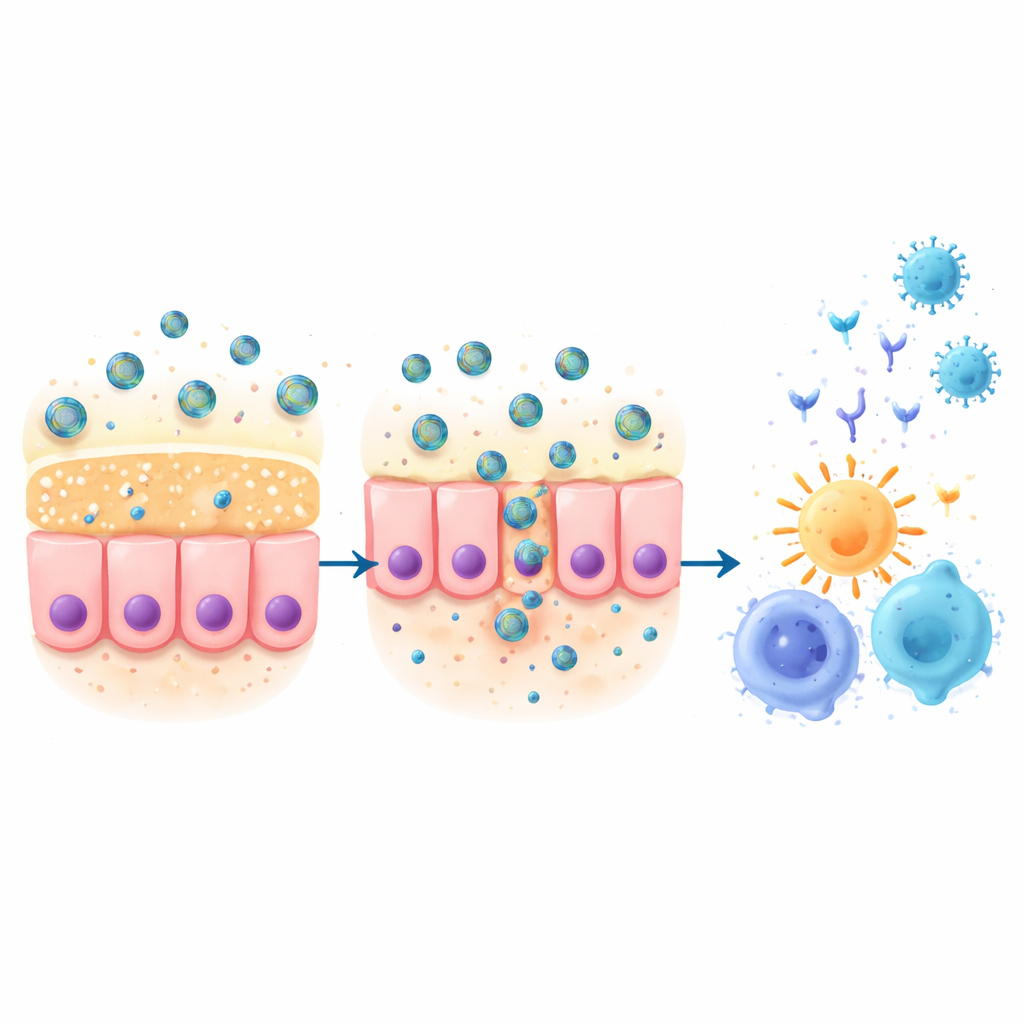
The researchers designed a tiny delivery system built from solid silica nanoparticles—hard, spherical particles much smaller than a virus. These particles were coated with a mucoadhesive cyclodextrin-based polymer that helps them stick to the slippery mucus lining inside the nose and slowly move through it. Attached to the particles were key pieces of the coronavirus spike protein, including the receptor-binding domain, plus carefully chosen short fragments that train both helper and killer T cells. A separate immune-boosting molecule was included to wake up local defenses. The goal was to combine sticking power and penetrating ability so that the vaccine components would stay in the nasal cavity long enough to be taken up by immune cells, without spreading widely through the body.
Staying in place and staying safe
In mice, the team showed that the nanovaccine lingered far longer in the nasal passages than the same viral proteins delivered without the specialized particles. Fluorescent tracking revealed that the coated nanoparticles remained detectable in the nose for hours, while little material accumulated in distant organs and all signal disappeared by 24 hours. Microscopic examination of tissues one day after dosing showed only mild, expected signs of local immune activation in the lungs and lymphoid organs, with no serious damage to the liver, kidneys, brain, or heart. This suggests that the nasal nanovaccine can focus its action where it is needed while keeping overall exposure and toxicity low.
Strong and long-lasting immune responses
When mice received three nasal doses spaced two weeks apart, the combined mucus-penetrating and mucoadhesive formulation triggered remarkably strong antibody responses in the blood. Levels of spike-binding antibodies, including those capable of neutralizing the original Wuhan strain of SARS-CoV-2, rose sharply after the second and third doses and remained high for at least a year. The vaccine also induced antibodies of several types, reflecting a balanced pattern of immune help, and it produced short-lived IgA antibodies in the blood. Importantly, compared with simpler formulations lacking either the particles or the sticky coating, only the full nanovaccine combination produced robust responses. It also generated many interferon‑gamma–producing T cells that recognize both the spike protein and conserved T-cell fragments, showing that cellular defenses were strongly engaged.
Building a shield in the airways
Beyond blood responses, the vaccine’s most distinctive feature was its impact on mucosal immunity. After two and three doses, the complete nanovaccine triggered spike‑specific IgA antibodies in saliva and nasal washings, but not when the same ingredients were given without the silica–polymer system. IgA is the dominant protective antibody on moist surfaces such as the nose and mouth and is closely linked to reduced viral load and milder disease. In the lower airways, the vaccine provoked IgG antibodies and neutralizing activity in lung washings, likely reflecting spillover from the bloodstream. While antibodies induced against the ancestral spike protein were less effective at neutralizing the Omicron XBB.1.5 variant, the drop in potency was smaller in nasal fluids than in serum, hinting that mucosal IgA may better recognize new variants.
Putting the nanovaccine to the test
To see whether these immune responses translated into real protection, the researchers challenged humanized mice—genetically engineered to be highly susceptible to SARS‑CoV‑2—with a lethal dose of the original virus. Animals that had received the nasal nanovaccine lost almost no weight, showed only mild and brief signs of illness, and all survived. In contrast, placebo-treated animals rapidly became sick and died. Swabs from the throat and samples from the lungs of vaccinated mice contained 20‑ to 100‑fold less viral genetic material, and in many animals the virus was undetectable. Lung tissue from vaccinated mice showed far fewer signs of pneumonia, vascular injury, and airway damage than tissue from unprotected animals, underscoring that the nanovaccine not only reduced infection but also prevented severe disease in this model.
What this could mean for future vaccines
For non-specialists, the take‑home message is that this study outlines a promising nose-delivered COVID‑19 vaccine that attacks the virus at its point of entry while also reinforcing whole‑body defenses. By combining nanoparticles that can both cling to and move through nasal mucus with carefully selected viral pieces, the platform produces long‑lasting antibodies and T cells, including IgA in the upper airways that may help curb infection and transmission. Although more work is needed to adapt the formulation for people, update the antigen to current variants, and confirm safety and efficacy in humans, this nanovaccine approach offers a plausible path toward next‑generation vaccines against SARS‑CoV‑2 and other respiratory viruses that spread through the nose.
Citation: Pagni, R.L., Cunha-Neto, E., Silva Santos, Y.d. et al. An innovative nasal nanovaccine against SARS-CoV-2 induces systemic and upper airway immunity controlling viral replication. npj Vaccines 11, 82 (2026). https://doi.org/10.1038/s41541-026-01407-x
Keywords: intranasal vaccine, nanoparticles, mucosal immunity, SARS-CoV-2, COVID-19 boosters