Clear Sky Science · en
Electrochemical investigation of platinum electrode corrosion under neurostimulation protocols
Why this matters for brain and hearing implants
Modern brain and hearing implants can restore movement, hearing, or mood by sending tiny electrical pulses into nerves. These devices must work safely for decades inside the body, often starting in childhood. In this study, researchers asked a simple but crucial question: do the very pulses that make these implants work slowly eat away at the metal contacts that deliver them, and if so, under which conditions does this hidden damage become dangerous?
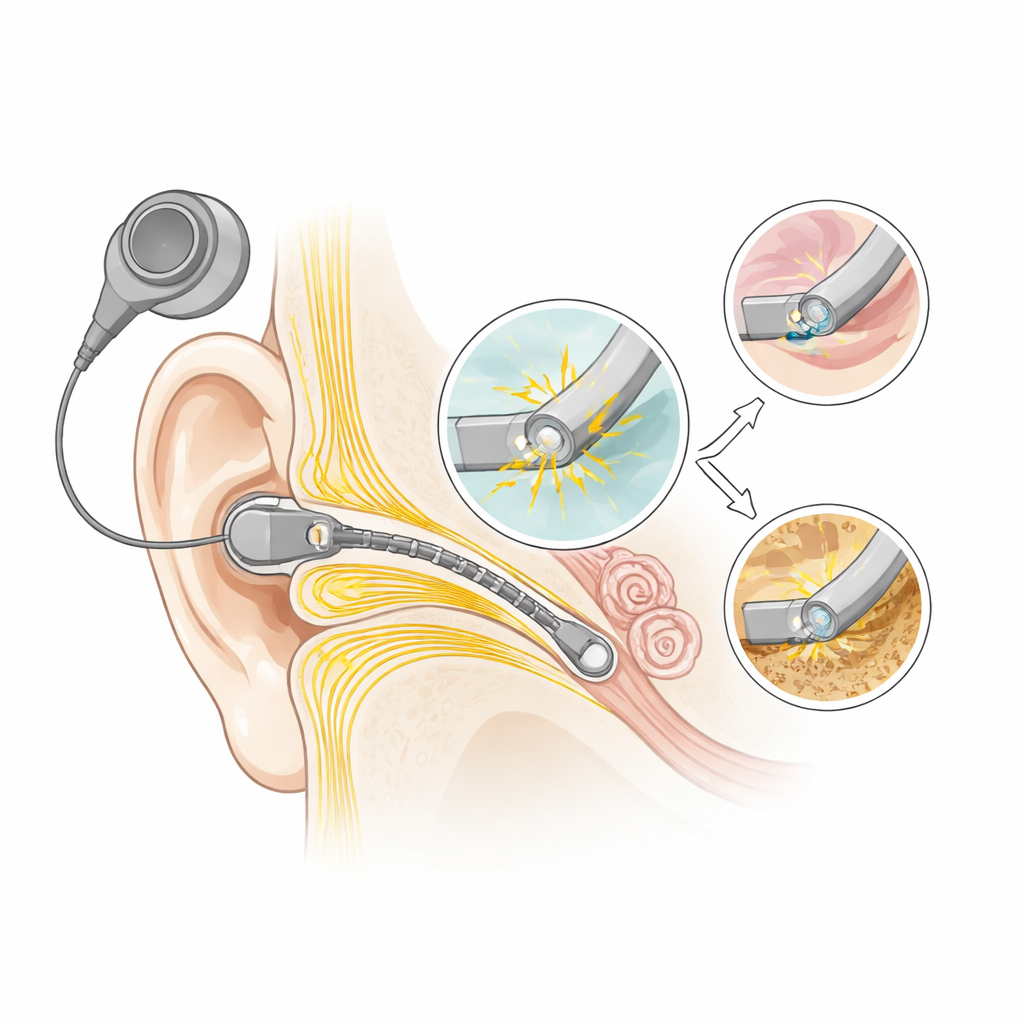
How tiny metal contacts keep nerves talking
Most clinical stimulators, from cochlear implants to deep brain stimulators, use platinum contacts to pass current into nearby nerve cells. Platinum is chosen because it is stable and conducts electricity well. Doctors design stimulation patterns so they stay within a supposedly “safe” electrical range, based mainly on avoiding gas bubbles and tissue injury. But past work has shown that platinum can still slowly dissolve, even at neutral pH similar to body fluids. Until now, however, most measurements compared the electrode only before and after long experiments, leaving the actual damage process over billions of pulses largely unseen.
A new way to watch electrodes age in real time
The authors built an automated test bed that mimics how a cochlear implant stimulates in animal and human use. A commercial neurostimulator sent fast, charge-balanced pulses through thin-film platinum contacts immersed in a salt solution like body fluid. At set intervals, electronic switches connected the same contacts to a sensitive electrochemistry instrument and an oscilloscope. This allowed the team to track the exact voltage of the electrode relative to a reference scale and to measure how much of its surface was still active for key reactions such as oxygen reduction. Because the platinum films were only 100 nanometers thick, mechanical profilometry could detect material loss with nanometer precision, and microscopes and electron microscopes revealed how the surface shape changed over time.
What really drives the metal to crumble
Following individual electrodes through billions of pulses, the researchers saw a characteristic four-stage life cycle. Early on, the apparent active surface area actually increased, likely due to gentle roughening and cleaning of the metal. Later, material began thinning from the edges and across the surface while electrical tests still looked fairly normal. Once the remaining platinum film was almost consumed, the surface suddenly reorganized: parts ballooned and roughened, holes opened down to the titanium underlayer, and the voltages during pulses shot beyond the safe “water window,” where gas bubbles of hydrogen and oxygen form. At that point the electrode was no longer usable. A key insight was that the worst damage occurred when each pulse repeatedly grew a thin platinum-oxide layer and then stripped it away again. This chemical cycling dramatically accelerated loss of metal.

Pulses that look similar on paper can age electrodes very differently
The team then compared different ways of arranging the same overall charge in the stimulation pulses. They varied whether the negative or positive phase came first, and whether the stimulator actively shorted the electrodes between pulses to force them back to a common starting voltage. Surprisingly, just changing the phase order and discharge behavior had a much larger effect on corrosion than modest changes in charge density per pulse. Two protocol types, which repeatedly pushed the electrode high enough to form oxide and then low enough to remove it, led to severe roughening and eventual failure. Two other protocol types, with the same charge per phase but voltage histories that either only formed oxide or only reduced it, showed no measurable material loss or surface roughening over the tested period. Overall, lifetime under current-controlled conditions was highly variable, and simple charge-based safety rules did not reliably predict when electrodes would fail.
Rethinking what “safe stimulation” means
This work shows that keeping stimulation within traditional charge limits is not enough to guarantee long-lived electrodes. What matters most is how the voltage of the platinum surface moves during and between pulses, especially whether it cycles through the oxide formation and removal range. By combining real-time voltage monitoring with repeated surface measurements, the authors demonstrate a framework that can reveal when a clinically used protocol quietly drives corrosion long before obvious failure. For future implants, this means that pulse shape, phase order, and how the device lets electrodes relax between pulses should be optimized not only for nerve response and tissue safety, but also to avoid the damaging chemical cycles that slowly consume the metal making stimulation possible.
Citation: Reinelt, S., Doering, M., Weltin, A. et al. Electrochemical investigation of platinum electrode corrosion under neurostimulation protocols. npj Mater Degrad 10, 49 (2026). https://doi.org/10.1038/s41529-026-00789-6
Keywords: neurostimulation electrodes, platinum corrosion, cochlear implants, electrode longevity, brain implants