Clear Sky Science · en
Thermophysical properties and solidification behavior of liquid Vit106a in microgravity
Why space helps us understand special metals
Some metals can be frozen into a glass-like state, giving them unusual strength and toughness that engineers hope to use in everything from spacecraft to medical devices. But making large pieces of this “metallic glass” is tricky, because the molten metal must be cooled fast enough to avoid forming crystals. This paper reports how scientists used microgravity on the International Space Station to carefully measure how a promising alloy, called Vit106a, behaves as a liquid and how it solidifies—key knowledge for manufacturing strong metallic glass parts at larger scales.
A metal that wants to become glass
Bulk metallic glasses are metal alloys whose atoms are frozen into a disordered arrangement, more like window glass than a typical crystal. This structure can make them very strong, elastic, and resistant to corrosion. Vit106a is a zirconium‑based alloy specially designed to form metallic glass without using toxic elements like beryllium. On Earth, small spheres of Vit106a only a few millimeters across can be cooled into glass at relatively modest cooling rates, suggesting it might be suitable for large parts. However, to truly control industrial casting, engineers need accurate data on how the molten alloy flows, radiates heat, and stores energy over a wide temperature range—data that are hard to obtain on Earth because gravity and container walls disturb the liquid.
Letting metal float in space
To overcome these limitations, the team processed a 6.5‑millimeter Vit106a sphere in an electromagnetic levitator aboard the International Space Station. Powerful coils held the droplet in mid‑air and heated it without any container touching it. In this nearly weightless environment, the researchers gently shook the droplet to measure how quickly surface ripples moved and faded, revealing its surface tension and viscosity (a measure of how runny or sluggish the liquid is). They also used a carefully modulated heating signal to determine how efficiently the droplet radiated heat and how much energy was needed to raise its temperature, which provided its emissivity and specific heat capacity.
What the liquid metal revealed
The measurements showed that Vit106a’s surface tension stays almost constant over the explored high‑temperature range and is very similar to that of other zirconium‑rich alloys, implying only weak surface‑driven flows in the melt. The viscosity data showed that, at high temperatures, the liquid behaves as a relatively “fragile” fluid whose resistance to flow changes rapidly with temperature. When these data were combined with earlier low‑temperature measurements from other groups, the analysis indicated that Vit106a undergoes a transition from more fragile to more “strong‑like” behavior as it cools toward the glass transition—an effect linked in other alloys to subtle rearrangements in the liquid’s atomic structure. The specific heat capacity of the fully molten alloy was found to be slightly higher than some ground‑based estimates, refining the thermodynamic picture needed for casting simulations.
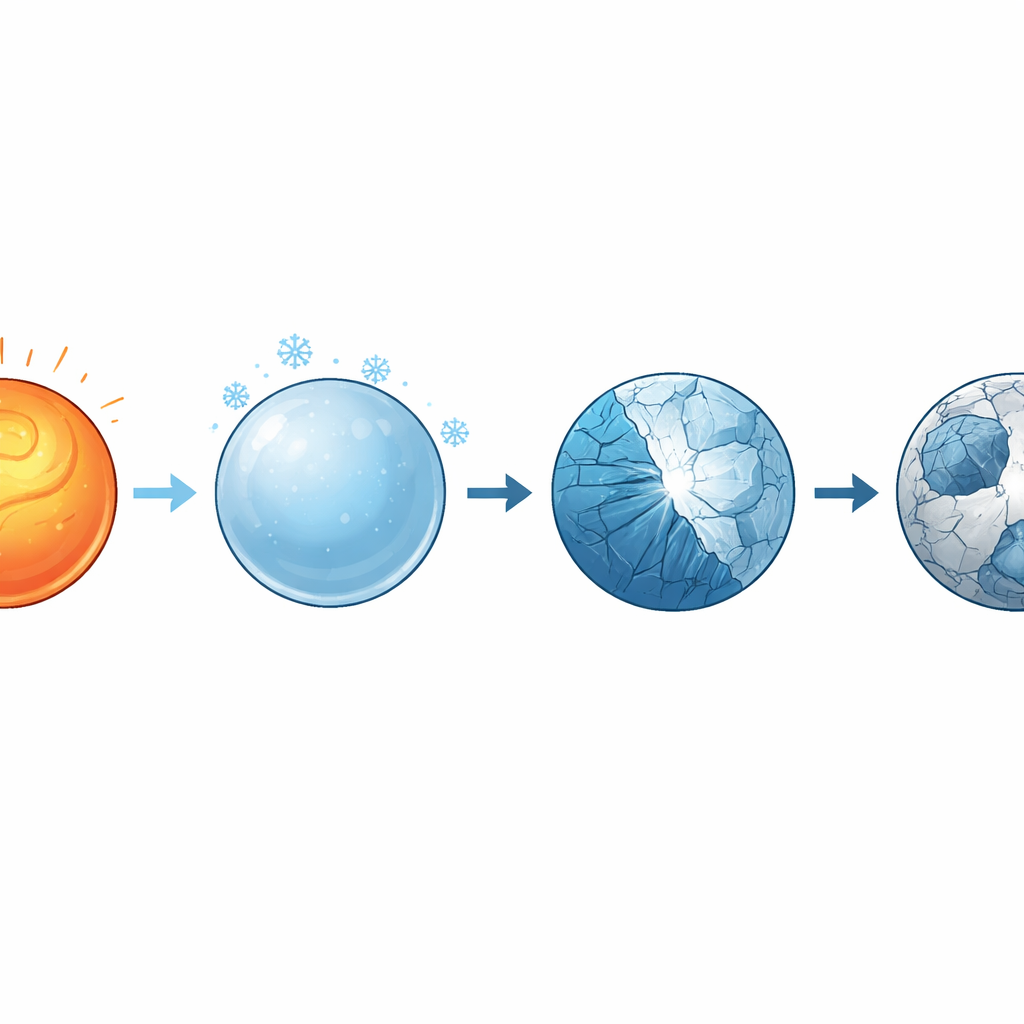
When cooling is fast but still not fast enough
After the property measurements, the droplet was allowed to cool freely in the levitator at about 16 kelvins per second—much faster than the critical cooling rate previously reported to be sufficient for forming glass in small Vit106a samples. Surprisingly, temperature recordings showed a clear plateau associated with crystallization, and detailed X‑ray diffraction and electron microscopy back on Earth confirmed that the sphere had become fully crystalline, not glassy. The solidified sphere contained several kinds of simple zirconium‑based compounds and large internal voids, suggesting that crystals started at the surface and grew inward, dragging material away from the center. This behavior points to heterogeneous nucleation, in which tiny impurities or structural fluctuations act as starting points for crystals, and raises doubts about how easily Vit106a can form glass in larger castings.
What this means for future metallic glass parts
The study delivers a precise set of thermophysical data for molten Vit106a under near‑ideal microgravity conditions and shows that, despite its reputation as an excellent glass former, it may crystallize more readily in larger volumes than earlier work on tiny samples implied. For engineers, these results highlight that successful large‑scale production will require not only fast enough cooling, but also strict control of oxygen and other impurities, careful management of how hot the melt is before cooling, and realistic expectations about how casting thickness affects glass formation. The new measurements can now feed into computer models that help design casting processes and hardware, bringing the goal of reliable, large metallic glass components one step closer.
Citation: Terebenec, D., Mohr, M., Wunderlich, R. et al. Thermophysical properties and solidification behavior of liquid Vit106a in microgravity. npj Microgravity 12, 26 (2026). https://doi.org/10.1038/s41526-026-00572-6
Keywords: bulk metallic glass, microgravity processing, Vit106a alloy, metal solidification, thermophysical properties