Clear Sky Science · en
Diffusion-Wetting: a universal molecular relation
Why Water on Surfaces Matters
From raindrops rolling off a leaf to blood flowing through medical implants, how water behaves on a surface can make or break a technology. Engineers usually describe this behavior with a single number: the contact angle, which tells you whether water beads up (water-repelling) or spreads out (water-loving). But at the tiny scales inside batteries, catalysts, or living cells, this familiar angle becomes fuzzy and hard to measure. This paper shows that instead of struggling to draw angles on microscopic droplets, we can read a surface’s “love or hate” for water directly from how quickly water molecules move along it.
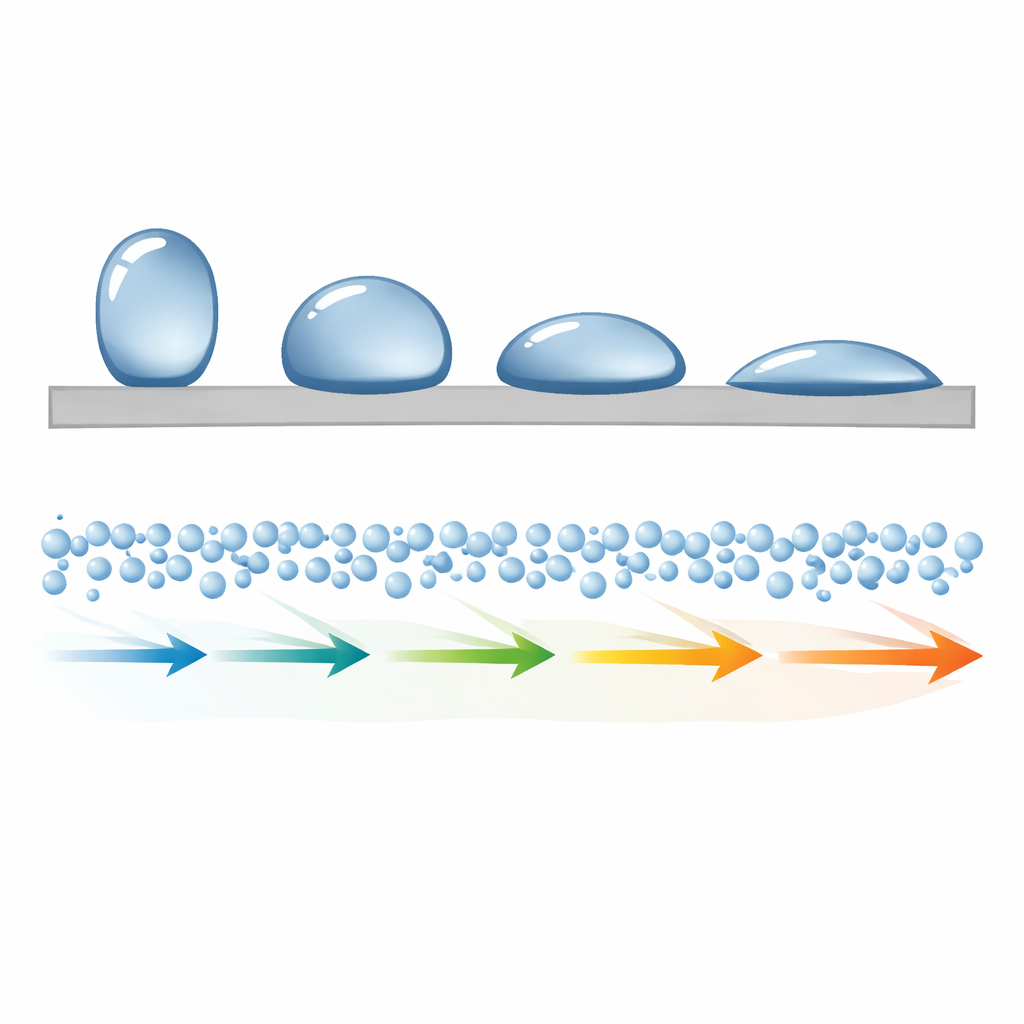
From Big Droplets to Tiny Molecules
Traditionally, wettability is measured by placing a droplet on a flat surface and recording the angle the droplet edge makes with the material. That angle comes from a balance of surface energies between solid, liquid, and air. In practice, however, real surfaces are rough, chemically patchy, and full of tiny defects. These details cause hysteresis, pinning, and other complications that make the measured angle differ from the ideal one. At the nanoscale, the whole idea of a macroscopic droplet may break down entirely, especially in narrow pores, reactive materials, or ultrathin films where droplets simply cannot form.
Looking at Motion Instead of Shape
The authors build on a simple physical intuition: water molecules move differently near a surface than they do in the bulk liquid, and that change in motion reflects how strongly the surface holds them. On water-loving surfaces, strong attractions and hydrogen bonds hold molecules in place and slow their sideways motion. On water-repelling surfaces, weaker interactions let molecules slip and slide more freely. Using molecular dynamics simulations—computer experiments that track individual water molecules—the researchers studied water next to idealized walls whose attraction to water could be finely tuned from very hydrophobic to very hydrophilic.
A Universal Link Between Speed and Spreading
In these simulations, the team measured two things: the contact angle of water droplets on the surface, and the lateral diffusion of water molecules in the first few billionths of a meter next to the wall. They found a remarkably simple relationship: the ratio between the diffusion near the surface and the diffusion in the bulk uniquely determines the contact angle across the full range from strongly water-repelling to strongly water-attracting. By connecting a standard formula for diffusion (which relates mobility to an energy barrier) with the classical contact-angle equation, they derived an analytical law that directly links the contact angle to diffusion in the interfacial layer, without needing to simulate or observe any droplets.
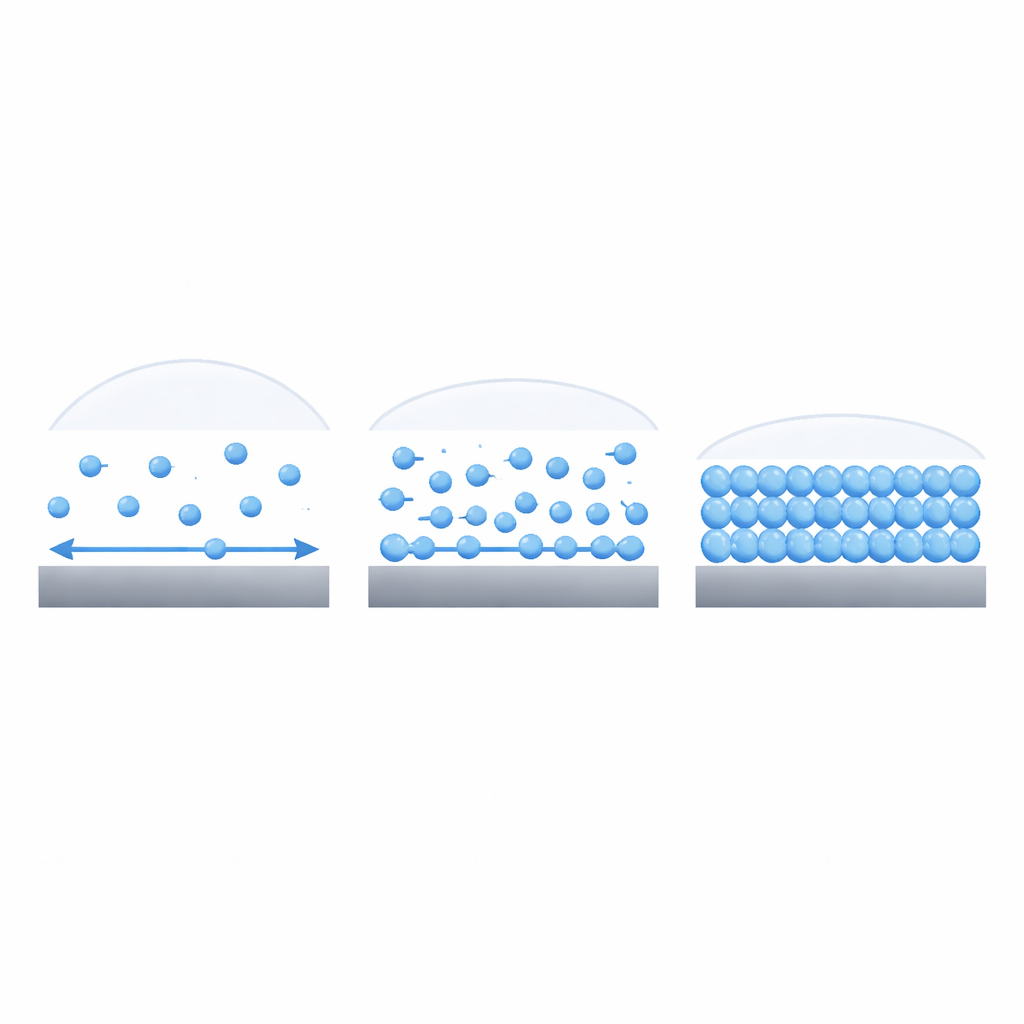
Checking the Rule and Extending Its Reach
The new diffusion–wetting law was tested against a broad set of simulations and matched the computed contact angles with high accuracy in every regime. In the extreme water-repelling limit, where interfacial water moves as freely as in a non-attracting environment, the formula predicts complete non-wetting. As the interfacial diffusion slows relative to the bulk, the predicted angle smoothly decreases, capturing the transition through neutral wetting into highly water-attracting behavior. Because diffusion near surfaces often reaches a steady value in very short simulations—even for expensive quantum-level or machine-learned models—the method is practical for complex materials where traditional free-energy calculations would be prohibitive.
What This Means in Simple Terms
For a lay reader, the main message is that you no longer need to watch how a droplet looks to know whether a surface is water-loving or water-hating. Instead, you can “feel” the surface through the way water molecules move along it. Fast motion signals weak grip and a high contact angle; slow motion signals a strong grip and a low contact angle. This provides a universal, efficient way to predict wettability in confined spaces, rough or reactive materials, and other situations where droplets are hard to define. In other words, the dance of water molecules at the surface becomes a direct, quantitative window into how that surface will behave in real-world applications, from anti-fog coatings to next-generation energy and biomedical devices.
Citation: Agosta, L., Dzugutov, M. Diffusion-Wetting: a universal molecular relation. npj Comput Mater 12, 163 (2026). https://doi.org/10.1038/s41524-026-02079-w
Keywords: wettability, hydrophobic surfaces, hydrophilic surfaces, interfacial diffusion, molecular dynamics