Clear Sky Science · en
Early mitophagy activation by Urolithin A prevents, but late activation does not reverse, age-related cognitive impairment
Why Keeping Our Brain’s Powerhouses Young Matters
As we grow older, many of us notice that names, routes, and recent events are harder to recall. Scientists know that this mental slowdown is closely tied to tiny power plants inside our brain cells called mitochondria. When these structures falter, the brain struggles to produce enough energy and to clear out damaged components. This study asks a simple but crucial question: can a natural compound called Urolithin A, which boosts cellular cleanup, protect memory as we age—and does timing matter?
A Natural Helper Born in the Gut
Urolithin A is not something we eat directly; it is produced when our gut microbes break down certain plant polyphenols found in foods like pomegranates and nuts. Earlier work showed that Urolithin A can trigger “mitophagy,” a housekeeping process that identifies worn-out mitochondria and removes them before they cause trouble. In animal models of Alzheimer’s disease, this compound improves memory and reduces harmful protein build-up. However, whether it can guard a normally aging brain—or even reverse age-related memory loss—has remained unclear. The authors set out to test Urolithin A in two different mouse strains that represent different stages and speeds of brain aging.
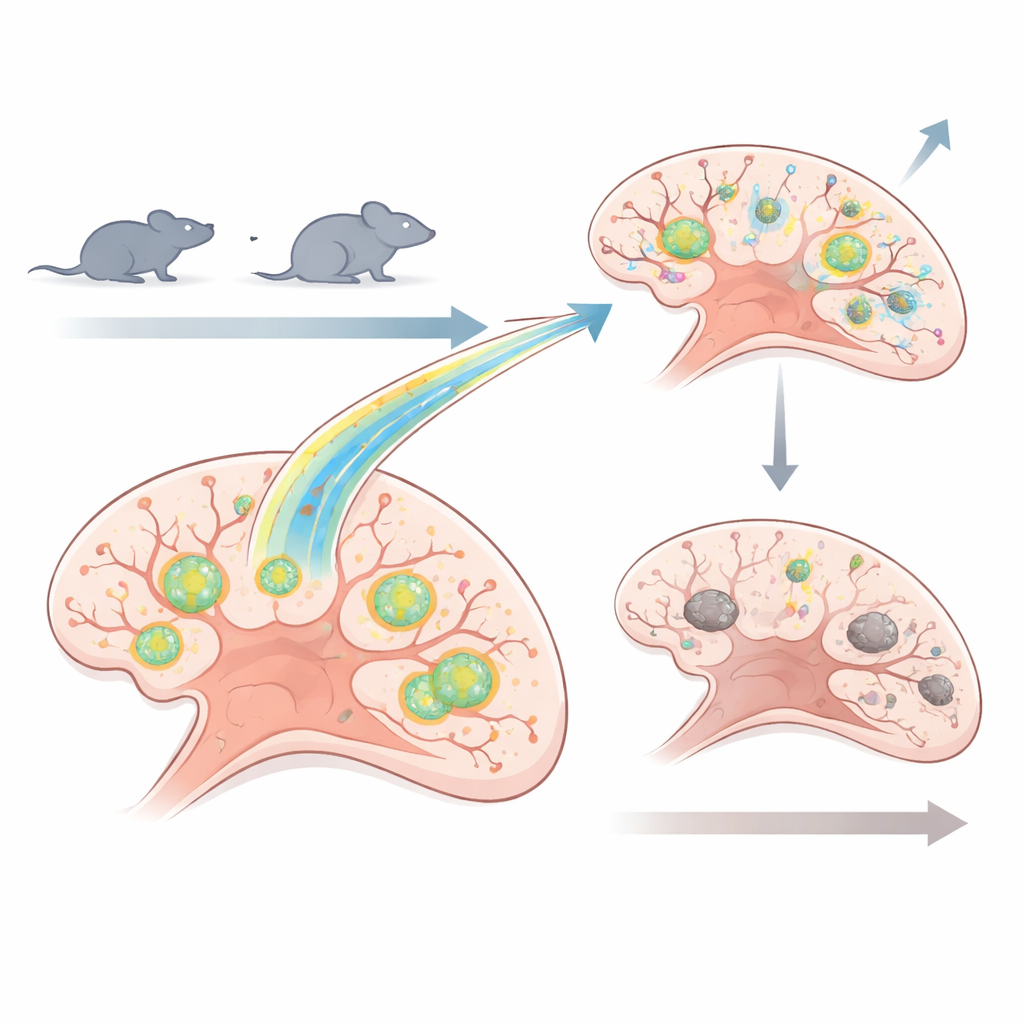
Two Paths of Aging, One Common Target
The researchers focused on the hippocampus, a deep brain region key for learning spatial layouts and recognizing objects. They used naturally aging C57BL/6 mice, treated late in life when memory problems were already established, and SAMP8 mice, a strain that ages unusually fast and begins to show memory decline around six months of age. These SAMP8 mice received Urolithin A earlier, before major memory loss had set in. In both cases, the compound was given for eight weeks. The team then examined energy production (ATP levels), signs of oxidative stress, protein clumps, the balance of mitochondrial renewal and removal, synaptic proteins, and performance in classic memory tests such as the Morris water maze and novel object recognition.
Late Help Boosts Energy but Not Memory
In the older C57BL/6 mice, Urolithin A clearly improved the inner life of hippocampal cells. Mitochondria made more ATP, produced fewer damaging reactive oxygen molecules, and showed signs that cleanup pathways were being engaged, especially through a quality-control route involving the PINK1 and Parkin proteins. Proteins that support the formation of new mitochondria, and those that favor healthy mitochondrial fusion, were also increased. Notably, levels of a particularly harmful form of the tau protein—a version linked to tangle-like structures in aging and Alzheimer’s disease—fell both in the whole hippocampus and within mitochondria themselves. Synaptic proteins important for communication between neurons went up, and the animals moved more and faster. Yet despite this cellular tune-up, their spatial and recognition memory remained poor. Once cognitive decline was firmly in place, two months of treatment were not enough to restore lost function.
Early Help Keeps the System in Shape
The story was very different in the fast-aging SAMP8 mice treated earlier in life. Short-term experiments in hippocampal slice cultures showed that Urolithin A quickly reduced protein misfolding and oxidative stress while boosting markers of mitophagy. In living animals, eight weeks of treatment led to a striking rise in ATP levels in the hippocampus and a drop in overall unfolded protein burden, including the troublesome phosphorylated tau form. One key postsynaptic protein, PSD95, which supports the structural stability of synapses, also increased. In the water maze, treated SAMP8 mice learned the platform location faster and maintained better spatial memory as they aged, while their untreated peers showed the expected decline. Interestingly, after this long treatment, molecular signs of mitophagy and mitochondrial biogenesis actually went down, suggesting that once the mitochondrial pool was healthier, cells dialed back their stress responses and repair machinery.
What This Means for Aging Brains
Taken together, the findings paint a clear picture: activating mitochondrial cleanup early can help prevent or delay age-related memory loss, but the same strategy is far less effective once damage is entrenched. Urolithin A acted like a preventive maintenance plan for the brain’s energy factories—limiting the buildup of defective mitochondria and toxic proteins, preserving synaptic components, and supporting learning and memory in a vulnerable aging model. In contrast, when given after memory decline had already emerged, it improved many cellular markers without restoring cognition. For people, this suggests that future therapies based on boosting mitophagy and mitochondrial health, possibly including Urolithin A or related compounds, are likely to work best if started before serious memory problems appear, as part of an early, preventive approach to healthy brain aging.
Citation: Jara, C., Venegas-Zamora, L., Park-Kang, H.S. et al. Early mitophagy activation by Urolithin A prevents, but late activation does not reverse, age-related cognitive impairment. npj Aging 12, 54 (2026). https://doi.org/10.1038/s41514-026-00351-3
Keywords: mitochondria, brain aging, memory loss, urolithin A, cellular cleanup