Clear Sky Science · en
Population-based genome-wide association study of plasma complex lipid species
Why tiny fats in blood matter for health
The blood is full of thousands of different fat molecules that help build cell walls, store energy and send signals between organs. Subtle imbalances in these fats are increasingly linked to heart disease, diabetes, kidney problems and even brain disorders such as Alzheimer’s disease. This study asks a simple but far-reaching question: how much of this complex "lipid landscape" in our blood is written in our genes, and can that genetic map help us understand who is at risk for which diseases?
Taking a deep look at blood fats
Most previous studies have lumped blood fats into broad groups like “good” and “bad” cholesterol or total triglycerides. Here, researchers in Germany went much deeper. In more than 6,000 adults from the population-based Rhineland Study, they precisely measured 970 individual complex lipid molecules and 267 summary measures of their fatty acid building blocks. These lipids spanned many families, including storage fats, membrane fats and more exotic species such as ceramides. At the same time, participants’ genomes were scanned for millions of common genetic variants, allowing the team to search for stretches of DNA that track with fine-grained differences in the lipid profile.
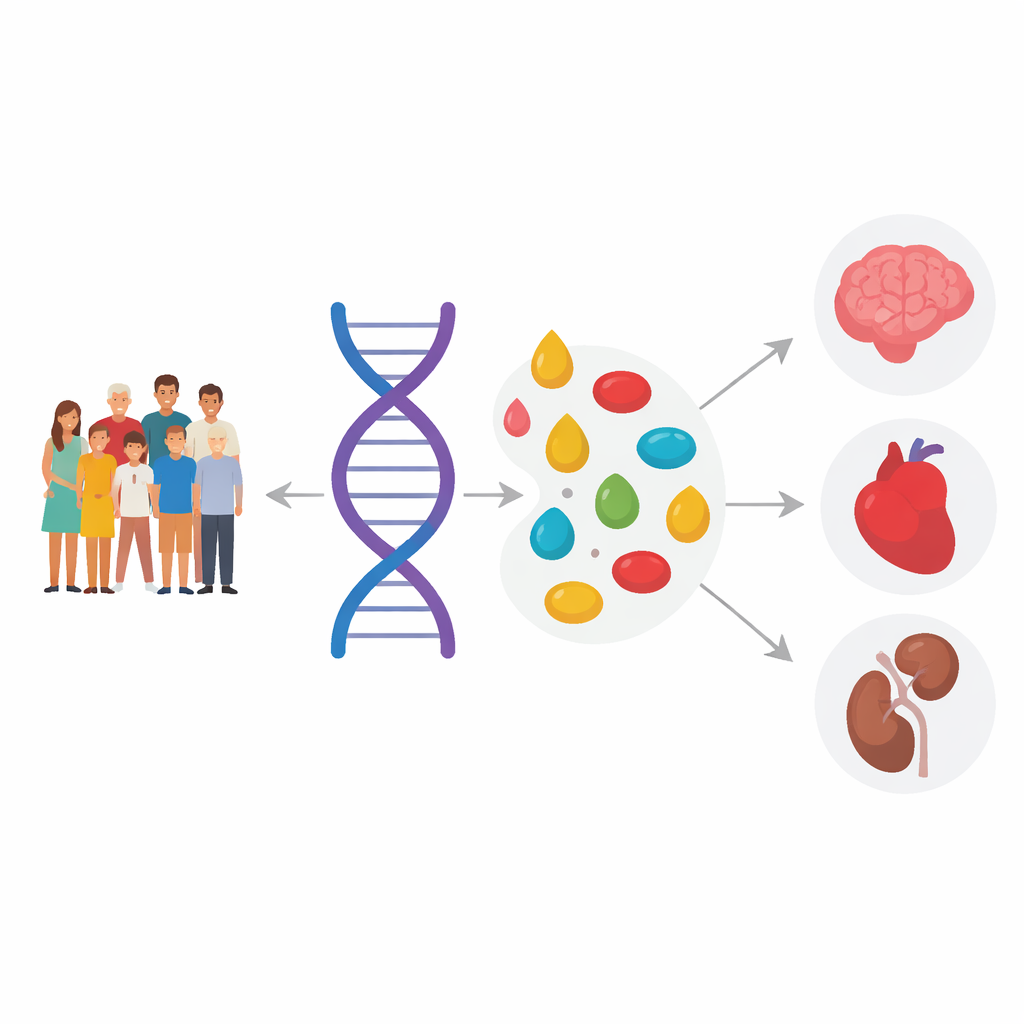
Genes that sculpt the lipid landscape
By systematically scanning the genome, the team found 217 key regions of DNA associated with levels of specific lipid molecules, 136 of which had not been linked to such traits before. Many of these regions sit near genes already known to be involved in fat handling, such as those controlling how unsaturated fats are made, or how fats are transported in the blood. Others, including FDFT1 and ABCA7, point to new players that influence particular lipid species or specific features of their fatty acid “tails”, like chain length and degree of saturation. Some genes acted broadly across many types of lipids, while others tuned just a narrow subset, revealing a surprisingly intricate genetic control of blood fats.
From genes and lipids to disease
Finding genetic links to lipids is only useful if those links matter for health. To test this, the researchers compared their lipid-associated variants against large genetics databases of common diseases, including coronary artery disease, type 2 diabetes, chronic kidney disease and Alzheimer’s disease. They then used statistical tools that mimic randomized trials in nature to ask whether particular lipid changes lie on the causal path to disease. The analyses suggested that certain triacylglycerols are likely involved in heart disease and diabetes, specific cholesteryl esters are tied to kidney disease, and particular complex lipids called lactosylceramides may help connect some genetic risk for Alzheimer’s disease to changes in the brain.
Checking and refining the genetic signals
To ensure the findings were not a fluke of a single study, the team cross-checked their results in two independent groups: more than 7,000 participants from the Finnish FinnGen project and nearly 1,200 from the EPIC-Potsdam cohort, which used the same lipid measurement platform. Most of the strongest gene–lipid links held up across studies. By combining data sets, the researchers uncovered additional genetic regions that would have been hard to detect in any one group alone. They also examined how much of the person-to-person variation in lipid levels could be explained by genetics, showing that some lipid types are strongly heritable while others are more shaped by lifestyle and environment.
What this means for future medicine
To a non-specialist, the central message is that blood fats are not just crude numbers on a lab report, but a rich tapestry of molecules under tight genetic control, with specific patterns that can nudge the body toward or away from disease. By charting which genes influence which detailed lipids, and which of those lipids are most likely to contribute directly to conditions like heart disease, diabetes, kidney disease and Alzheimer’s disease, this work lays groundwork for more precise risk prediction and more targeted therapies. In the future, a person’s genetic and lipid “fingerprints” might help doctors identify who would benefit from particular drugs or lifestyle changes long before symptoms appear.
Citation: Landstra, E.N., Imtiaz, M.A., Talevi, V. et al. Population-based genome-wide association study of plasma complex lipid species. Nat Commun 17, 3984 (2026). https://doi.org/10.1038/s41467-026-72542-1
Keywords: lipidomics, genetics, cardiometabolic disease, Alzheimer’s disease, biomarkers