Clear Sky Science · en
Enhanced elastocaloric cooling beyond Clausius–Clapeyron limits
Why cooling needs a new approach
Air conditioners, supermarket freezers, and data centers all rely on vapor-compression refrigerators that burn a lot of electricity and use gases that can warm the planet. Scientists are searching for “solid-state” cooling systems that swap out gases for solids and work by gently squeezing and releasing special materials. This paper reports a titanium–aluminum–chromium (Ti–Al–Cr) alloy that cools very efficiently over an unusually wide temperature range, pointing toward lighter, greener refrigerators for everything from homes to spacecraft.
Cooling by squeezing metal
The alloy in this study uses the elastocaloric effect: when the metal is stressed, its internal crystal structure changes shape, and when the stress is released, the structure snaps back, absorbing or releasing heat in the process. Unlike ordinary metals that simply bend, this Ti–Al–Cr alloy behaves like a spring on the atomic scale, undergoing a reversible “phase transformation” between two crystal structures. By rapidly unloading the stress under nearly insulated conditions, the researchers directly measured how much the alloy cools itself, similar to watching a stretched rubber band cool when let go, but with far larger and more controllable temperature drops.
A wide window of useful temperatures
Most elastocaloric materials face a tough trade-off: they can either give a big cooling effect over a narrow temperature band, or a smaller effect over a broad band. This limitation is tied to a classical thermodynamic rule called the Clausius–Clapeyron relation, which links how strongly stress depends on temperature to how much entropy—the measure of heat-handling capacity—changes during transformation. The Ti–Al–Cr alloy breaks out of this box. In compression tests on carefully prepared single crystals, the team observed stable, fully reversible superelastic behavior from near absolute zero up to about 460 K. Direct cooling measurements showed a strong cooling response from 97 K to 402 K, a span of 305 K, much wider than theory would normally allow.
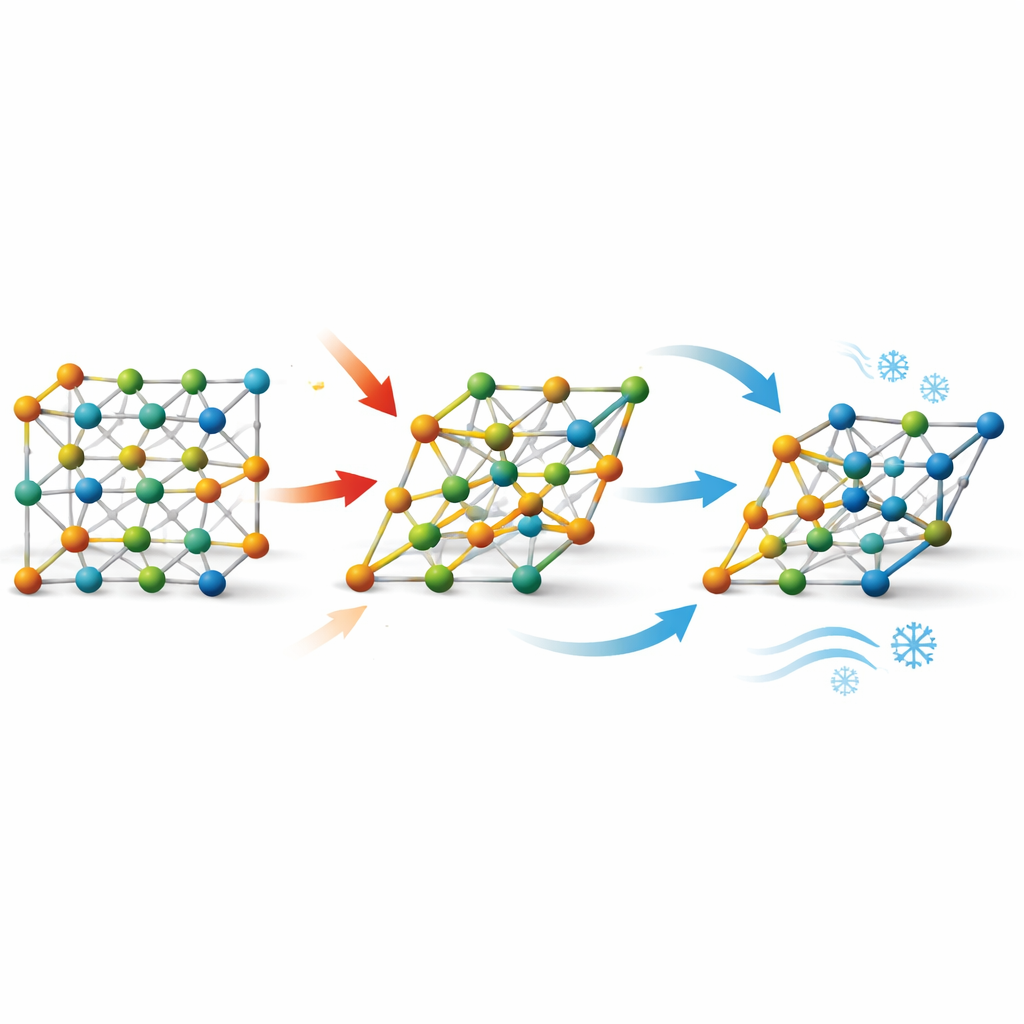
How the crystal structure makes it possible
To see what happens inside the metal while it is being squeezed, the researchers used in-situ neutron diffraction, which lets them watch atomic planes move under load. They found that the alloy switches cleanly between a simple cubic-like “B2” structure and a more distorted “B19” structure, with no messy intermediate phases. About two-thirds of the recoverable strain comes from this transformation, and one-third from ordinary elastic stretching, and the change is fully reversible. This clean, two-phase behavior allows the thermodynamics of the transformation—especially the heat and entropy changes—to be described reliably from structural data, providing a firm foundation for evaluating and predicting cooling performance.
Beating classic thermodynamic limits
The authors combined calorimetry (precise heat measurements) with mechanical tests to calculate the effective entropy change and total cooling capacity, taking into account real-world energy losses due to hysteresis. At room temperature, the alloy shows an adiabatic temperature drop of about 10 K and a cooling output of 5.76 J per gram, with a material coefficient of performance of 4.6—competitive with leading commercial elastocaloric alloys. More strikingly, when they compared the measured performance to what would be expected from the Clausius–Clapeyron relation, they found that both the working temperature span and the total cooling capacity exceeded the predicted “upper bounds” by roughly 20–30%. This unusual behavior stems from an anomalous stiffening of the parent crystal structure at low temperatures, which flattens the usual link between stress and temperature and lets strong cooling persist even when a key thermodynamic parameter is near zero.
What this could mean for future cooling
Because titanium alloys are relatively light, this Ti–Al–Cr material offers a rare combination of large cooling power, broad temperature coverage (97–402 K), and low density, making it particularly attractive for weight-sensitive applications such as aerospace and portable electronics. The work also carries a deeper message: the traditional thermodynamic rules used to judge elastocaloric materials are not absolute limits. By deliberately designing alloys whose internal stiffness and structural response change in unusual ways with temperature, it may be possible to routinely exceed classical expectations for solid-state cooling, opening a path to compact, efficient, and climate-friendly refrigeration technologies.
Citation: Song, Y., Xu, S., Omori, T. et al. Enhanced elastocaloric cooling beyond Clausius–Clapeyron limits. Nat Commun 17, 3747 (2026). https://doi.org/10.1038/s41467-026-72172-7
Keywords: elastocaloric cooling, shape memory alloys, solid-state refrigeration, titanium alloys, energy-efficient cooling