Clear Sky Science · en
Sliding friction over individual aromatic bonds correlates with bond order
Why the Tiny World of Friction Matters
Every time you brake a car, swipe a screen, or run an engine, friction is at work. Yet, despite its everyday familiarity, scientists still struggle to predict and control friction when things get very small—down to individual atoms. This study peels back that mystery by asking a deceptively simple question: when something slides over a single chemical bond, what exactly causes energy loss, and can we tune it by changing the bond itself?
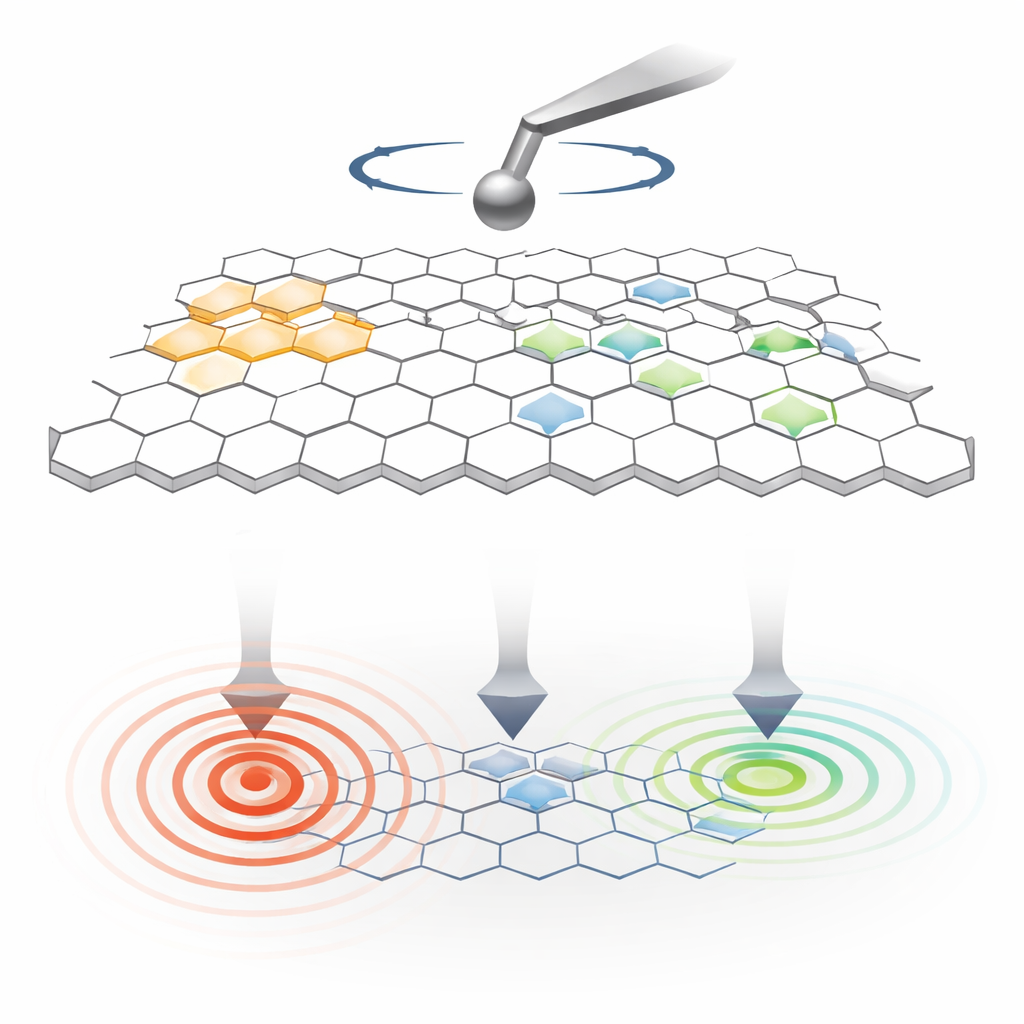
Looking at Friction One Bond at a Time
Most friction experiments involve rough surfaces with countless tiny contact points, making it nearly impossible to see what happens at each one. Here, the researchers instead used an ultra-sensitive microscope setup where a sharp tip ending effectively in a single atom is made to oscillate sideways over a surface. The surface is coated with carefully arranged organic molecules (PTCDA) on copper, which provide both strong covalent bonds and weaker hydrogen bonds in a well-defined pattern. By functionalizing the tip with a single carbon monoxide molecule, the team ensured that the sliding contact is chemically simple, stable, and reproducible over many measurements.
Measuring Invisible Energy Loss
As the tip swings back and forth over the bonds, it bends the CO molecule slightly like a tiny torsion spring. When the tip passes over a bond, the CO can snap from one side of the bond to the other. That snapping motion does mechanical work and converts ordered motion into vibrations and other excitations in the system—this is the energy lost to friction. The instrument tracks how much extra drive is needed to maintain the same oscillation, turning that into a direct measurement of energy dissipated per cycle. Importantly, this method senses only very short-range effects, probing the potential energy landscape within less than an angstrom above each bond.
Surprising Differences Between Similar Bonds
Intuition might suggest that sliding over similar aromatic carbon–carbon bonds would produce nearly identical friction. The experiments revealed otherwise: nominally similar C–C bonds showed almost a factor-of-two difference in maximum energy loss. Hydrogen bonds, often thought of as weaker and more diffuse, sometimes produced friction of similar magnitude to covalent bonds. To understand these variations, the team used density functional theory (DFT) combined with a machine-learning-based model of the tip–surface interaction. These advanced simulations reproduced the measured energy dissipation curves and allowed the researchers to connect friction directly to the electronic structure of each bond.
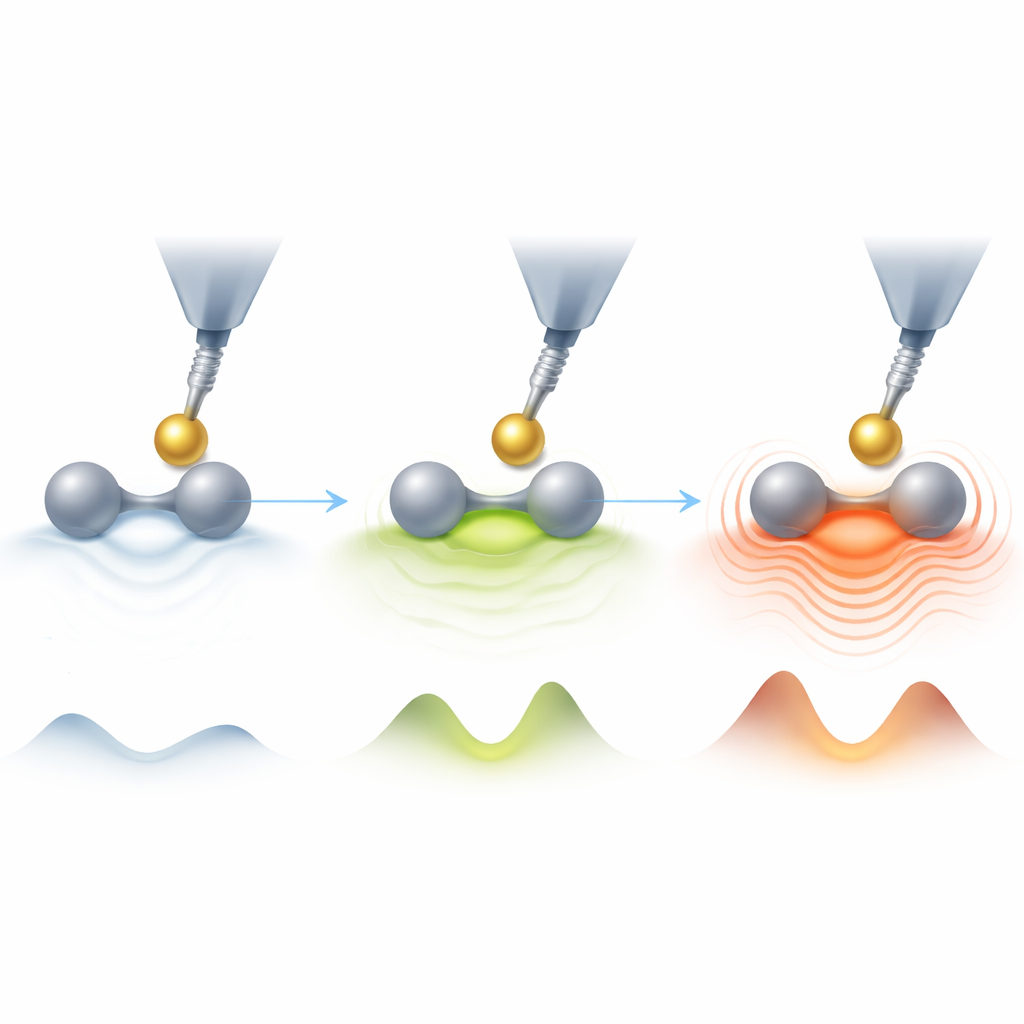
Bond Order as a Knob for Friction
For covalent aromatic bonds, the simulations showed a clear trend: bonds with higher bond order—meaning more shared electron density between the two atoms—produce more friction when the tip slides across them. In the language of chemistry, bond order reflects how strongly and how multiply two atoms are tied together; here, higher bond order also means a more “corrugated” energy landscape that the tip must climb over. This rougher landscape enhances repulsion as the tip passes, increasing the amount of energy lost per oscillation. Hydrogen bonds break this pattern. They have low bond order and little extra electron density between atoms, yet the friction they cause can rival that of aromatic bonds. In that case, the tip interacts more directly with the individual atoms forming the bond rather than with a concentrated electron cloud between them.
From Atomic Snap to Engineered Surfaces
By tying sliding friction to the detailed nature of individual bonds, this work offers a new way to think about designing materials with tailored friction. Instead of treating friction as an emergent property of rough surfaces, engineers could choose which types of bonds—and which bond orders—to place at a surface to raise or lower energy loss. The study shows that even subtle changes in how electrons are shared between atoms can dramatically alter friction at the smallest scale, opening a route toward atomically precise coatings and interfaces for future nanomachines, sensors, and low-wear materials.
Citation: Nam, S., Hörmann, L., Gretz, O. et al. Sliding friction over individual aromatic bonds correlates with bond order. Nat Commun 17, 3694 (2026). https://doi.org/10.1038/s41467-026-72128-x
Keywords: atomic-scale friction, chemical bonds, bond order, nanotribology, surface engineering